- NEWS AND VIEWS
Ageing-related receptors resolved
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 553, 409-410 (2018)
doi: https://doi.org/10.1038/d41586-017-09032-4
References
Kuro-o, M. et al. Nature 390, 45–51 (1997).
Kurosu, H. et al. J. Biol. Chem. 281, 6120–6123 (2006).
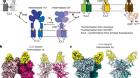
Chen, G. et al. Nature 553, 461–466 (2018).
Lee, S. et al. Nature 553, 501–505 (2018).
Hu, M. C., Shiizake, K., Kuro-o, M. & Moe, O. W. Annu. Rev. Physiol. 75, 503–533 (2013).
Shimada, T. et al. J. Clin. Invest. 113, 561–568 (2004).
Ogawa, Y. et al. Proc. Natl Acad. Sci. USA 104, 7432–7437 (2007).
Kurosu, H. et al. J. Biol. Chem. 282, 26687–26695 (2007).
Wright, P. E. & Dyson, H. J. J. Mol. Biol. 293, 321–331 (1999).
Kuro-o, M. Nature Rev. Nephrol. 9, 650–660 (2013).
Zhang, Y. et al. eLife 1, e00065 (2012).

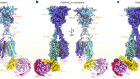
 Read the paper: α-Klotho is a non-enzymatic molecular scaffold for FGF23 hormone signalling
Read the paper: α-Klotho is a non-enzymatic molecular scaffold for FGF23 hormone signalling
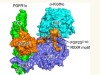
 Read the paper: Structures of β-klotho reveal a ‘zip code’-like mechanism for endocrine FGF signalling
Read the paper: Structures of β-klotho reveal a ‘zip code’-like mechanism for endocrine FGF signalling
 Longer life through an odd Pol enzyme
Longer life through an odd Pol enzyme
 Mapping the path to a longer life
Mapping the path to a longer life