Abstract
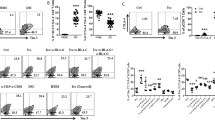
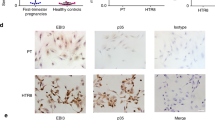
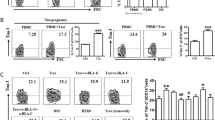
Successful human pregnancy requires the maternal immune system to recognize and tolerate the semi-allogeneic fetus. Myeloid-derived suppressor cells (MDSCs), which are capable of inhibiting T-cell responses, are highly increased in the early stages of pregnancy. Although recent reports indicate a role for MDSCs in fetal–maternal tolerance, little is known about the expansion of MDSCs during pregnancy. In the present study, we demonstrated that the trophoblast cell line HTR8/SVneo could instruct peripheral CD14+ myelomonocytic cells toward a novel subpopulation of MDSCs, denoted as CD14+HLA-DR−/low cells, with suppressive activity and increased expression of IDO1, ARG-1, and COX2. After interaction with HTR8/SVneo cells, CD14+ myelomonocytic cells secrete high levels of CCL2, promoting the expression of signal transducer and activator of transcription 3. We utilized a neutralizing monoclonal antibody to reveal the prominent role of CCL2 in the induction of CD14+HLA-DR−/low MDSCs. In combination, the results of the present study support a novel role for the cross-talk between the trophoblast cell line HTR8/SVneo and maternal CD14+ myelomonocytic cells in initiating MDSCs induction, prompting a tolerogenic immune response to ensure a successful pregnancy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Cha J, Sun X, Dey SK . Mechanisms of implantation: strategies for successful pregnancy. Nat Med 2012; 18: 1754–67.
Vacca P, Cantoni C, Vitale M, Prato C, Canegallo F, Fenoglio D et al. Crosstalk between decidual NK and CD14+ myelomonocytic cells results in induction of Tregs and immunosuppression. Proc Natl Acad Sci 2010; 107: 11918–11923.
McIntire RH, Ganacias KG, Hunt JS . Programming of human monocytes by the uteroplacental environment. Reprod Sci 2008; 15: 437–447.
Houser BL, Tilburgs T, Hill J, Nicotra ML, Strominger JL . Two unique human decidual macrophage populations. J Immunol 2011; 186: 2633–2642.
Nagamatsu T, Schust DJ . The contribution of macrophages to normal and pathological pregnancies. Am J Reprod Immunol 2010; 63: 460–471.
Poschke I, Mougiakakos D, Hansson J, Masucci GV, Kiessling R . Immature immunosuppressive CD14+HLA-DR-/low cells in melanoma patients are Stat3hi and overexpress CD80, CD83, and DC-sign. Cancer Res 2010; 70: 4335–4345.
Höchst B, Schildberg FA, Sauerborn P, Gäbel YA, Gevensleben H, Goltz D et al. Activated human hepatic stellate cells induce myeloid derived suppressor cells from peripheral blood monocytes in a CD44-dependent fashion. J Hepatol 2013; 59: 528–535.
Hoechst B, Ormandy LA, Ballmaier M, Lehner F, Krüger C, Manns MP et al. A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4+CD25+Foxp3+ T cells. Gastroenterol 2008; 135: 234–243.
Le Bitoux M-A, Waeber S, Stamenkovic I . Myeloid cells display immunosuppression activities during pregnancy, participating to the establishment of pre-metastatic niches. Cancer Res 2013; 73: 4980.
Köstlin N, Kugel H, Spring B, Leiber A, Marmé A, Henes M et al. Granulocytic myeloid‐derived suppressor cells expand in human pregnancy and modulate T‐cell responses. Eur J Immunol 2014; 44: 2582–2591.
Red-Horse K, Drake PM, Gunn MD, Fisher SJ . Chemokine ligand and receptor expression in the pregnant uterus: reciprocal patterns in complementary cell subsets suggest functional roles. American J Pathol. 2001; 159: 2199–2213.
Kayisli UA, Mahutte NG, Arici A . Uterine chemokines in reproductive physiology and pathology. Am J Reprod Immunol. 2002; 47: 213–221.
Shynlova O, Tsui P, Dorogin A, Lye SJ . Monocyte chemoattractant protein-1 (CCL-2) integrates mechanical and endocrine signals that mediate term and preterm labor. J Immunol 2008; 181: 1470–1479.
Lockwood CJ, Matta P, Krikun G, Koopman LA, Masch R, Toti P et al. Regulation of monocyte chemoattractant protein-1 expression by tumor necrosis factor-α and interleukin-1β in first trimester human decidual cells: implications for preeclampsia. Am J Pathol 2006; 168: 445–452.
Gu L, Tseng S, Horner RM, Tam C, Loda M, Rollins BJ . Control of TH2 polarization by the chemokine monocyte chemoattractant protein-1. Nature 2000; 404: 407–411.
Karpus WJ, Kennedy KJ, Kunkel SL, Lukacs NW . Monocyte chemotactic protein 1 regulates oral tolerance induction by inhibition of T helper cell 1-related cytokines. J Exp Med 1998; 187: 733–741.
Fest S, Aldo PB, Abrahams VM, Visintin I, Alvero A, Chen R et al. Trophoblast-macrophage interactions: a regulatory network for the protection of pregnancy. Am J Reprod Immunol 2007; 57: 55–66.
Du M-R, Guo P-F, Piao H-L, Wang S-C, Sun C, Jin L-P et al. Embryonic trophoblasts induce decidual regulatory T cell differentiation and maternal–fetal tolerance through thymic stromal lymphopoietin instructing dendritic cells. J Immunol 2014; 192: 1502–1511.
Guo P-F, Du M-R, Wu H-X, Lin Y, Jin L-P, Li D-J . Thymic stromal lymphopoietin from trophoblasts induces dendritic cell-mediated regulatory TH2 bias in the decidua during early gestation in humans. Blood 2010; 116: 2061–2069.
McKinnon T, Chakraborty C, Gleeson LM, Chidiac P, Lala PK . Stimulation of human extravillous trophoblast migration by IGF-II is mediated by IGF type 2 receptor involving inhibitory G protein (s) and phosphorylation of MAPK. J Clin Endocrinol Metab 2001; 86: 3665–3674.
Johnsen G, Basak S, Weedon-Fekjaer M, Staff A, Duttaroy A . Docosahexaenoic acid stimulates tube formation in first trimester trophoblast cells, HTR8/SVneo. Placenta 2011; 32: 626–632.
Irving J, Lysiak J, Graham C, Hearn S, Han V, Lala P . Characteristics of trophoblast cells migrating from first trimester chorionic villus explants and propagated in culture. Placenta 1995; 16: 413–433.
Zhang J, Patel L, Pienta KJ . CC chemokine ligand 2 (CCL2) promotes prostate cancer tumorigenesis and metastasis. Cytokine Growth Factor Rev 2010; 21: 41–48.
Vasquez-Dunddel D, Pan F, Zeng Q, Gorbounov M, Albesiano E, Fu J et al. STAT3 regulates arginase-I in myeloid-derived suppressor cells from cancer patients. J Clin Investig 2013; 123: 1580–1589.
Kämmerer U, Eggert AO, Kapp M, McLellan AD, Geijtenbeek TB, Dietl J et al. Unique appearance of proliferating antigen-presenting cells expressing DC-SIGN (CD209) in the decidua of early human pregnancy. Am J Pathol 2003; 162: 887–896.
Croxatto D, Vacca P, Canegallo F, Conte R, Venturini PL, Moretta L et al. Stromal cells from human decidua exert a strong inhibitory effect on NK cell function and dendritic cell differentiation. PLoS One 2014; 9: e89006.
Ziegler-Heitbrock L, Ancuta P, Crowe S, Dalod M, Grau V, Hart DN et al. Nomenclature of monocytes and dendritic cells in blood. Blood 2010; 116: e74–e80.
McIntire RH, Ganacias KG, Hunt JS . Programming of human monocytes by the uteroplacental environment. Reprod Sci 2008; 15: 437–447.
Miwa N . IDO expression on decidual and peripheral blood dendritic cells and monocytes/macrophages after treatment with CTLA-4 or interferon-γ increase in normal pregnancy but decrease in spontaneous abortion. Mol Hum Reprod 2006; 11: 865–870.
Cupurdija K, Azzola D, Hainz U, Gratchev A, Heitger A, Takikawa O et al. Macrophages of human first trimester decidua express markers associated to alternative activation. Am J Reprod Immunol 2004; 51: 117–122.
Atay S, Gercel‐Taylor C, Suttles J, Mor G, Taylor DD . Trophoblast‐derived exosomes mediate monocyte recruitment and differentiation. Am J Reprod Immunol 2011; 65: 65–77.
Aldo PB, Racicot K, Craviero V, Guller S, Romero R, Mor G . Trophoblast induces monocyte differentiation into CD14+/CD16+ macrophages. Am J Reprod Immunol 2014; 72: 270–284.
Talmadge JE, Gabrilovich DI . History of myeloid-derived suppressor cells. Nat Rev Cancer 2013; 13: 739–752.
Highfill SL, Rodriguez PC, Zhou Q, Goetz CA, Koehn BH, Veenstra R et al. Bone marrow myeloid-derived suppressor cells (MDSCs) inhibit graft-versus-host disease (GVHD) via an arginase-1-dependent mechanism that is up-regulated by interleukin-13. Blood 2010; 116: 5738–5747.
Luther SA, Cyster JG . Chemokines as regulators of T cell differentiation. Nature Immunol 2001; 2: 102–107.
He Y-Y, He X-J, Guo P-F, Du M-R, Shao J, Li M-Q et al. The decidual stromal cells-secreted CCL2 induces and maintains decidual leukocytes into Th2 bias in human early pregnancy. Clin Immunol 2012; 145: 161–173.
Nefedova Y, Huang M, Kusmartsev S, Bhattacharya R, Cheng P, Salup R et al. Hyperactivation of STAT3 is involved in abnormal differentiation of dendritic cells in cancer. J Immu: 464–474.
Nefedova Y, Nagaraj S, Rosenbauer A, Muro-Cacho C, Sebti SM, Gabrilovich DI . Regulation of dendritic cell differentiation and antitumor immune response in cancer by pharmacologic-selective inhibition of the janus-activated kinase 2/signal transducers and activators of transcription 3 pathway. Cancer Res 2005; 65: 9525–9535.
Xin H, Zhang C, Herrmann A, Du Y, Figlin R, Yu H . Sunitinib inhibition of Stat3 induces renal cell carcinoma tumor cell apoptosis and reduces immunosuppressive cells. Cancer Res 2009; 69: 2506–2513.
Acknowledgements
We are very grateful to Dr Charles H. Graham (Department of Anatomy and Cell Biology, Queen's University, Kingston, ON, Canada) for providing the HTR8/SVneo cell line. This study was supported by grants from the National Natural Science Foundation of China (grant nos. 31470885, 31270971, 81300510, 31300752, and 31100650).
Author information
Authors and Affiliations
Additional information
Supplementary Information accompanies the paper on Cellular & Molecular Immunology’s website (http://www.nature.com/cmi).
Rights and permissions
About this article
Cite this article
Zhang, Y., Qu, D., Sun, J. et al. Human trophoblast cells induced MDSCs from peripheral blood CD14+ myelomonocytic cells via elevated levels of CCL2. Cell Mol Immunol 13, 615–627 (2016). https://doi.org/10.1038/cmi.2015.41
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2015.41
Keywords
This article is cited by
-
Trophoblast-derived miR-410-5p induces M2 macrophage polarization and mediates immunotolerance at the fetal-maternal interface by targeting the STAT1 signaling pathway
Journal of Translational Medicine (2024)
-
Mesenchymal stem cells, as glioma exosomal immunosuppressive signal multipliers, enhance MDSCs immunosuppressive activity through the miR-21/SP1/DNMT1 positive feedback loop
Journal of Nanobiotechnology (2023)
-
Human placental exosomes induce maternal systemic immune tolerance by reprogramming circulating monocytes
Journal of Nanobiotechnology (2022)
-
Continuous activation of polymorphonuclear myeloid-derived suppressor cells during pregnancy is critical for fetal development
Cellular & Molecular Immunology (2021)
-
Non-invasive Embryo Assessment: Altered Individual Protein Profile in Spent Culture Media from Embryos Transferred at Day 5
Reproductive Sciences (2021)