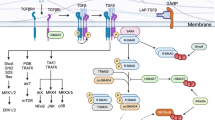
Abstract
Upon antigen stimulation, naive T helper cells differentiate into distinct lineages to attain specialized properties and effector functions. TH17 cells, a recently identified lineage of CD4+ effector T cells, play a key role in the immune defense against fungi and extracellular bacteria, but also contribute to the pathogenesis of many autoimmune conditions. The differentiation of TH17 cells is orchestrated by an intricate network of signaling pathways and transcriptional regulators in T cells. While the involvement of T cell-intrinsic pathways has been described extensively, we are just beginning to appreciate how TH17 cell development is shaped by extrinsic pathways, especially the innate immune signals. Dendritic cells (DCs), the most important cell type to bridge innate and adaptive immunity, drive TH17 cell differentiation by providing antigenic, costimulatory and cytokine signals. This is mediated by the recognition of innate and inflammatory signals by DCs via pattern recognition receptors, cytokine receptors and other immunomodulatory receptors that in turn activate the intracellular signaling network. In particular, p38α MAP kinase has emerged as a critical pathway to program DC-dependent TH17 cell differentiation by integrating multiple instructive signals in DCs. Here, we summarize the current knowledge on the mechanisms by which DC-derived innate immune signals drive TH17 cell differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mosmann TR, Coffman RL . TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. Annu Rev Immunol 1989; 7: 145-173.
Korn T, Bettelli E, Oukka M, Kuchroo VK . IL-17 and Th17 Cells. Annu Rev Immunol 2009; 27: 485-517.
Zhu J, Yamane H, Paul WE . Differentiation of effector CD4 T cell populations. Annu Rev Immunol 2010; 28: 445-489.
O'Shea JJ, Paul WE . Mechanisms underlying lineage commitment and plasticity of helper CD4+ T cells. Science 2010; 327: 1098-1102.
Littman DR, Rudensky AY . Th17 and regulatory T cells in mediating and restraining inflammation. Cell 2010; 140: 845-858.
Iwasaki A, Medzhitov R . Regulation of adaptive immunity by the innate immune system. Science 2010; 327: 291-295.
Joffre O, Nolte MA, Sporri R, Reis e Sousa C . Inflammatory signals in dendritic cell activation and the induction of adaptive immunity. Immunol Rev 2009; 227: 234-247.
Park H, Li Z, Yang XO, Chang SH, Nurieva R, Wang YH et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol 2005; 6: 1133-1141.
Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol 2005; 6: 1123-1132.
Dong C . TH17 cells in development: an updated view of their molecular identity and genetic programming. Nat Rev Immunol 2008; 8: 337-348.
Ivanov II . McKenzie BS . Zhou L . Tadokoro CE . Lepelley A . Lafaille JJ . et al The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 2006; 126: 1121-1133.
Yang XO, Pappu BP, Nurieva R, Akimzhanov A, Kang HS, Chung Y et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors ROR alpha and ROR gamma. Immunity 2008; 28: 29-39.
Chen Z, Laurence A, Kanno Y, Pacher-Zavisin M, Zhu BM, Tato C et al. Selective regulatory function of Socs3 in the formation of IL-17-secreting T cells. Proc Natl Acad Sci U S A 2006; 103: 8137-8142.
Yang XO, Panopoulos AD, Nurieva R, Chang SH, Wang D, Watowich SS et al. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J Biol Chem 2007; 282: 9358-9363.
Zhou L, Ivanov II, Spolski R, Min R, Shenderov K, Egawa T, et al. IL-6 programs TH-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol 2007; 8: 967-974.
Brustle A, Heink S, Huber M, Rosenplanter C, Stadelmann C, Yu P et al. The development of inflammatory TH-17 cells requires interferon-regulatory factor 4. Nat Immunol 2007; 8: 958-966.
Schraml BU, Hildner K, Ise W, Lee WL, Smith WA, Solomon B et al. The AP-1 transcription factor Batf controls TH17 differentiation. Nature 2009; 460: 405-409.
Zhang F, Meng G, Strober W . Interactions among the transcription factors Runx1, RORgammat and Foxp3 regulate the differentiation of interleukin 17-producing T cells. Nat Immunol 2008; 9: 1297-1306.
Bauquet AT, Jin H, Paterson AM, Mitsdoerffer M, Ho IC, Sharpe AH et al. The costimulatory molecule ICOS regulates the expression of c-Maf and IL-21 in the development of follicular T helper cells and TH-17 cells. Nat Immunol 2009; 10: 167-175.
Quintana FJ, Basso AS, Iglesias AH, Korn T, Farez MF, Bettelli E et al. Control of Treg and TH17 cell differentiation by the aryl hydrocarbon receptor. Nature 2008; 453: 65-71.
Veldhoen M, Hirota K, Westendorf AM, Buer J, Dumoutier L, Renauld JC et al. The aryl hydrocarbon receptor links TH17-cell-mediated autoimmunity to environmental toxins. Nature 2008; 453: 106-109.
Shi LZ, Wang R, Huang G, Vogel P, Neale G, Green DR et al. HIF1α-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J Exp Med 2011; 208: 1367-1376.
Dang EV, Barbi J, Yang HY, Jinasena D, Yu H, Zheng Y et al. Control of TH17/Treg balance by hypoxia-inducible factor 1. Cell 2011; 146: 772-784.
Hosken NA, Shibuya K, Heath AW, Murphy KM, O'Garra A . The effect of antigen dose on CD4+ T helper cell phenotype development in a T cell receptor-alpha beta-transgenic model. J Exp Med 1995; 182: 1579-1584.
Constant S, Pfeiffer C, Woodard A, Pasqualini T, Bottomly K . Extent of T cell receptor ligation can determine the functional differentiation of naive CD4+ T cells. J Exp Med 1995; 182: 1591-1596.
Ruedl C, Bachmann MF, Kopf M . The antigen dose determines T helper subset development by regulation of CD40 ligand. Eur J Immunol 2000; 30: 2056-2064.
Iezzi G, Sonderegger I, Ampenberger F, Schmitz N, Marsland BJ, Kopf M . CD40-CD40L cross-talk integrates strong antigenic signals and microbial stimuli to induce development of IL-17-producing CD4+ T cells. Proc Natl Acad Sci U S A 2009; 106: 876-881.
Perona-Wright G, Jenkins SJ, O'Connor RA, Zienkiewicz D, McSorley HJ, Maizels RM et al. A pivotal role for CD40-mediated IL-6 production by dendritic cells during IL-17 induction in vivo. J Immunol 2009; 182: 2808-2815.
Huang G, Wang Y, Vogel P, Kanneganti TD, Otsu K, Chi H . Signaling via the kinase p38alpha programs dendritic cells to drive TH17 differentiation and autoimmune inflammation. Nat Immunol 2012; 13: 152-161.
Odobasic D, Leech MT, Xue JR, Holdsworth SR . Distinct in vivo roles of CD80 and CD86 in the effector T-cell responses inducing antigen-induced arthritis. Immunology 2008; 124: 503-513.
Ying H, Yang L, Qiao G, Li Z, Zhang L, Yin F et al. Cutting edge: CTLA-4—B7 interaction suppresses Th17 cell differentiation. J Immunol 2010; 185: 1375-1378.
Paulos CM, Carpenito C, Plesa G, Suhoski MM, Varela-Rohena A, Golovina TN et al. The inducible costimulator (ICOS) is critical for the development of human TH17 cells. Sci Transl Med 2010; 2: 55ra78.
Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B . TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 2006; 24: 179-189.
Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 2006; 441: 235-238.
Mangan PR, Harrington LE, O'Quinn DB, Helms WS, Bullard DC, Elson CO et al. Transforming growth factor-beta induces development of the TH17 lineage. Nature 2006; 441: 231-234.
Korn T, Bettelli E, Gao W, Awasthi A, Jager A, Strom TB et al. IL-21 initiates an alternative pathway to induce proinflammatory TH17 cells. Nature 2007; 448: 484-487.
Nurieva R, Yang XO, Martinez G, Zhang Y, Panopoulos AD, Ma L et al. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 2007; 448: 480-483.
McGeachy MJ, Chen Y, Tato CM, Laurence A, Joyce-Shaikh B, Blumenschein WM et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol 2009; 10: 314-324.
Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 2003; 421: 744-748.
Duerr RH, Taylor KD, Brant SR, Rioux JD, Silverberg MS, Daly MJ et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science 2006; 314: 1461-1463.
Sutton C, Brereton C, Keogh B, Mills KH, Lavelle EC . A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J Exp Med 2006; 203: 1685-1691.
Chung Y, Chang SH, Martinez GJ, Yang XO, Nurieva R, Kang HS et al. Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 2009; 30: 576-587.
Li MO, Wan YY, Flavell RA . T cell-produced transforming growth factor-beta1 controls T cell tolerance and regulates Th1- and Th17-cell differentiation. Immunity 2007; 26: 579-591.
Torchinsky MB, Garaude J, Martin AP, Blander JM . Innate immune recognition of infected apoptotic cells directs TH17 cell differentiation. Nature 2009; 458: 78-82.
Zhou L, Lopes JE, Chong MM, Ivanov II, Min R, Victora GD, et al. TGF-beta-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORgammat function. Nature 2008; 453: 236-240.
Ghoreschi K, Laurence A, Yang XP, Tato CM, McGeachy MJ, Konkel JE et al. Generation of pathogenic TH17 cells in the absence of TGF-beta signalling. Nature 2010; 467: 967-971.
Gutcher I, Donkor MK, Ma Q, Rudensky AY, Flavell RA, Li MO . Autocrine transforming growth factor-beta1 promotes in vivo Th17 cell differentiation. Immunity 2011; 34: 396-408.
Veldhoen M, Uyttenhove C, van Snick J, Helmby H, Westendorf A, Buer J et al. Transforming growth factor-beta ‘reprograms’ the differentiation of T helper 2 cells and promotes an interleukin 9-producing subset. Nat Immunol 2008; 9: 1341-1346.
Melton AC, Bailey-Bucktrout SL, Travis MA, Fife BT, Bluestone JA, Sheppard D . Expression of alphavbeta8 integrin on dendritic cells regulates Th17 cell development and experimental autoimmune encephalomyelitis in mice. J Clin Invest 2010; 120: 4436-4444.
Acharya M, Mukhopadhyay S, Paidassi H, Jamil T, Chow C, Kissler S et al. alphav Integrin expression by DCs is required for Th17 cell differentiation and development of experimental autoimmune encephalomyelitis in mice. J Clin Invest 2010; 120: 4445-4452.
Stumhofer JS, Laurence A, Wilson EH, Huang E, Tato CM, Johnson LM et al. Interleukin 27 negatively regulates the development of interleukin 17-producing T helper cells during chronic inflammation of the central nervous system. Nat Immunol 2006; 7: 937-945.
Batten M, Li J, Yi S, Kljavin NM, Danilenko DM, Lucas S et al. Interleukin 27 limits autoimmune encephalomyelitis by suppressing the development of interleukin 17-producing T cells. Nat Immunol 2006; 7: 929-936.
Amadi-Obi A, Yu CR, Liu X, Mahdi RM, Clarke GL, Nussenblatt RB et al. TH17 cells contribute to uveitis and scleritis and are expanded by IL-2 and inhibited by IL-27/STAT1. Nat Med 2007; 13: 711-718.
Pot C, Apetoh L, Awasthi A, Kuchroo VK . Induction of regulatory Tr1 cells and inhibition of TH17 cells by IL-27. Semin Immunol 2011; 23: 438-445.
Awasthi A, Carrier Y, Peron JP, Bettelli E, Kamanaka M, Flavell RA et al. A dominant function for interleukin 27 in generating interleukin 10-producing anti-inflammatory T cells. Nat Immunol 2007; 8: 1380-1389.
Fitzgerald DC, Zhang GX, El-Behi M, Fonseca-Kelly Z, Li H, Yu S et al. Suppression of autoimmune inflammation of the central nervous system by interleukin 10 secreted by interleukin 27-stimulated T cells. Nat Immunol 2007; 8: 1372-1379.
Stumhofer JS, Silver JS, Laurence A, Porrett PM, Harris TH, Turka LA et al. Interleukins 27 and 6 induce STAT3-mediated T cell production of interleukin 10. Nat Immunol 2007; 8: 1363-1371.
Wilke CM, Wang L, Wei S, Kryczek I, Huang E, Kao J et al. Endogenous interleukin-10 constrains Th17 cells in patients with inflammatory bowel disease J Transl Med. 2011; 9: 217.
Laurence A, Tato CM, Davidson TS, Kanno Y, Chen Z, Yao Z et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 2007; 26: 371-381.
McGeachy MJ, Bak-Jensen KS, Chen Y, Tato CM, Blumenschein W, McClanahan T et al. TGF-beta and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell-mediated pathology. Nat Immunol 2007; 8: 1390-1397.
Mucida D, Park Y, Kim G, Turovskaya O, Scott I, Kronenberg M et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 2007; 317: 256-260.
Sun CM, Hall JA, Blank RB, Bouladoux N, Oukka M, Mora JR et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J Exp Med 2007; 204: 1775-1785.
Coombes JL, Siddiqui KR, Arancibia-Carcamo CV, Hall J, Sun CM, Belkaid Y et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-beta and retinoic acid-dependent mechanism. J Exp Med 2007; 204: 1757-1764.
Sutton CE, Lalor SJ, Sweeney CM, Brereton CF, Lavelle EC, Mills KH . Interleukin-1 and IL-23 induce innate IL-17 production from gammadelta T cells, amplifying Th17 responses and autoimmunity. Immunity 2009; 31: 331-341.
van de Veerdonk FL, Marijnissen RJ, Kullberg BJ, Koenen HJ, Cheng SC, Joosten I et al. The macrophage mannose receptor induces IL-17 in response to Candida albicans. Cell Host Microbe 2009; 5: 329-340.
Ivanov II, Atarashi K, Manel N, Brodie EL, Shima T, Karaoz U . et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 2009; 139: 485-498.
Schnare M, Barton GM, Holt AC, Takeda K, Akira S, Medzhitov R . Toll-like receptors control activation of adaptive immune responses. Nat Immunol 2001; 2: 947-950.
Evans HG, Suddason T, Jackson I, Taams LS, Lord GM . Optimal induction of T helper 17 cells in humans requires T cell receptor ligation in the context of Toll-like receptor-activated monocytes. Proc Natl Acad Sci U S A 2007; 104: 17034-17039.
Abdollahi-Roodsaz S, Joosten LA, Koenders MI, Devesa I, Roelofs MF, Radstake TR et al. Stimulation of TLR2 and TLR4 differentially skews the balance of T cells in a mouse model of arthritis. J Clin Invest 2008; 118: 205-216.
LeibundGut-Landmann S, Gross O, Robinson MJ, Osorio F, Slack EC, Tsoni SV et al. Syk- and CARD9-dependent coupling of innate immunity to the induction of T helper cells that produce interleukin 17. Nat Immunol 2007; 8: 630-638.
Huang G, Wang Y, Shi LZ, Kanneganti TD, Chi H . Signaling by the phosphatase MKP-1 in dendritic cells imprints distinct effector and regulatory T cell fates. Immunity 2011; 35: 45-58.
Manicassamy S, Ravindran R, Deng J, Oluoch H, Denning TL, Kasturi SP et al. Toll-like receptor 2-dependent induction of vitamin A-metabolizing enzymes in dendritic cells promotes T regulatory responses and inhibits autoimmunity. Nat Med 2009; 15: 401-409.
Loures FV, Pina A, Felonato M, Calich VL . TLR2 is a negative regulator of Th17 cells and tissue pathology in a pulmonary model of fungal infection. J Immunol 2009; 183: 1279-1290.
Moreira AP, Cavassani KA, Ismailoglu UB, Hullinger R, Dunleavy MP, Knight DA et al. The protective role of TLR6 in a mouse model of asthma is mediated by IL-23 and IL-17A. J Clin Invest 2011; 121: 4420-4432.
Lukacs NW, Smit JJ, Mukherjee S, Morris SB, Nunez G, Lindell DM . Respiratory virus-induced TLR7 activation controls IL-17-associated increased mucus via IL-23 regulation. J Immunol 2010; 185: 2231-2239.
Reynolds JM, Pappu BP, Peng J, Martinez GJ, Zhang Y, Chung Y et al. Toll-like receptor 2 signaling in CD4+ T lymphocytes promotes T helper 17 responses and regulates the pathogenesis of autoimmune disease. Immunity 2010; 32: 692-702.
Kerrigan AM, Brown GD . Syk-coupled C-type lectins in immunity. Trends Immunol 2011; 32: 151-156.
Robinson MJ, Osorio F, Rosas M, Freitas RP, Schweighoffer E, Gross O et al. Dectin-2 is a Syk-coupled pattern recognition receptor crucial for Th17 responses to fungal infection. J Exp Med 2009; 206: 2037-2051.
Saijo S, Ikeda S, Yamabe K, Kakuta S, Ishigame H, Akitsu A et al. Dectin-2 recognition of alpha-mannans and induction of Th17 cell differentiation is essential for host defense against Candida albicans. Immunity 2010; 32: 681-691.
Fritz JH, Le Bourhis L, Sellge G, Magalhaes JG, Fsihi H, Kufer TA et al. Nod1-mediated innate immune recognition of peptidoglycan contributes to the onset of adaptive immunity. Immunity 2007; 26: 445-459.
Elinav E, Strowig T, Henao-Mejia J, Flavell RA . Regulation of the antimicrobial response by NLR proteins. Immunity 2011; 34: 665-679.
van Beelen AJ, Zelinkova Z, Taanman-Kueter EW, Muller FJ, Hommes DW, Zaat SA et al. Stimulation of the intracellular bacterial sensor NOD2 programs dendritic cells to promote interleukin-17 production in human memory T cells. Immunity 2007; 27: 660-669.
Shaw PJ, Barr MJ, Lukens JR, McGargill MA, Chi H, Mak TW et al. Signaling via the RIP2 adaptor protein in central nervous system-infiltrating dendritic cells promotes inflammation and autoimmunity. Immunity 2011; 34: 75-84.
Shaw PJ, Lukens JR, Burns S, Chi H, McGargill MA, Kanneganti TD . Cutting edge: critical role for PYCARD/ASC in the development of experimental autoimmune encephalomyelitis. J Immunol 2010; 184: 4610-4614.
Gris D, Ye Z, Iocca HA, Wen H, Craven RR, Gris P et al. NLRP3 plays a critical role in the development of experimental autoimmune encephalomyelitis by mediating Th1 and Th17 responses. J Immunol 2010; 185: 974-981.
Lalor SJ, Dungan LS, Sutton CE, Basdeo SA, Fletcher JM, Mills KH . Caspase-1-processed cytokines IL-1beta and IL-18 promote IL-17 production by gammadelta and CD4 T cells that mediate autoimmunity. J Immunol 2011; 186: 5738-5748.
Dunne A, Ross PJ, Pospisilova E, Masin J, Meaney A, Sutton CE et al. Inflammasome activation by adenylate cyclase toxin directs Th17 responses and protection against Bordetella pertussis. J Immunol 2010; 185: 1711-1719.
van de Veerdonk FL, Joosten LA, Shaw PJ, Smeekens SP, Malireddi RK, van der Meer JW et al. The inflammasome drives protective Th1 and Th17 cellular responses in disseminated candidiasis. Eur J Immunol 2011; 41: 2260-2268.
Meng G, Zhang F, Fuss I, Kitani A, Strober W . A mutation in the Nlrp3 gene causing inflammasome hyperactivation potentiates Th17 cell-dominant immune responses. Immunity 2009; 30: 860-874.
Zaba LC, Cardinale I, Gilleaudeau P, Sullivan-Whalen M, Suarez-Farinas M, Fuentes-Duculan J et al. Amelioration of epidermal hyperplasia by TNF inhibition is associated with reduced Th17 responses. J Exp Med 2007; 204: 3183-3194.
Krishnamoorthy N, Oriss TB, Paglia M, Fei M, Yarlagadda M, Vanhaesebroeck B et al. Activation of c-Kit in dendritic cells regulates T helper cell differentiation and allergic asthma. Nat Med 2008; 14: 565-573.
Guo B, Chang EY, Cheng G . The type I IFN induction pathway constrains Th17-mediated autoimmune inflammation in mice. J Clin Invest 2008; 118: 1680-1690.
Shinohara ML, Kim JH, Garcia VA, Cantor H . Engagement of the type I interferon receptor on dendritic cells inhibits T helper 17 cell development: role of intracellular osteopontin. Immunity 2008; 29: 68-78.
Ramgolam VS, Sha Y, Jin J, Zhang X, Markovic-Plese S . IFN-beta inhibits human Th17 cell differentiation. J Immunol 2009; 183: 5418-5427.
Axtell RC, de Jong BA, Boniface K, van der Voort LF, Bhat R, De Sarno P et al. T helper type 1 and 17 cells determine efficacy of interferon-beta in multiple sclerosis and experimental encephalomyelitis. Nat Med 2010; 16: 406-412.
Axtell RC, Raman C, Steinman L . Interferon-beta exacerbates Th17-mediated inflammatory disease. Trends Immunol 2011; 32: 272-277.
Murugaiyan G, Mittal A, Weiner HL . Increased osteopontin expression in dendritic cells amplifies IL-17 production by CD4+ T cells in experimental autoimmune encephalomyelitis and in multiple sclerosis. J Immunol 2008; 181: 7480-7488.
Murugaiyan G, Mittal A, Weiner HL . Identification of an IL-27/osteopontin axis in dendritic cells and its modulation by IFN-gamma limits IL-17-mediated autoimmune inflammation. Proc Natl Acad Sci U S A 2010; 107: 11495-11500.
Kuwabara T, Ishikawa F, Yasuda T, Aritomi K, Nakano H, Tanaka Y et al. CCR7 ligands are required for development of experimental autoimmune encephalomyelitis through generating IL-23-dependent Th17 cells. J Immunol 2009; 183: 2513-2521.
Wilson JM, Kurtz CC, Black SG, Ross WG, Alam MS, Linden J et al. The A2B adenosine receptor promotes Th17 differentiation via stimulation of dendritic cell IL-6. J Immunol 2011; 186: 6746-6752.
Sheibanie AF, Yen JH, Khayrullina T, Emig F, Zhang M, Tuma R et al. The proinflammatory effect of prostaglandin E2 in experimental inflammatory bowel disease is mediated through the IL-23—>IL-17 axis. J Immunol 2007; 178: 8138-8147.
Esaki Y, Li Y, Sakata D, Yao C, Segi-Nishida E, Matsuoka T et al. Dual roles of PGE2-EP4 signaling in mouse experimental autoimmune encephalomyelitis. Proc Natl Acad Sci U S A 2010; 107: 12233-12238.
Nguyen NT, Kimura A, Nakahama T, Chinen I, Masuda K, Nohara K et al. Aryl hydrocarbon receptor negatively regulates dendritic cell immunogenicity via a kynurenine-dependent mechanism. Proc Natl Acad Sci U S A 2010; 107: 19961-19966.
Quintana FJ, Murugaiyan G, Farez MF, Mitsdoerffer M, Tukpah AM, Burns EJ et al. An endogenous aryl hydrocarbon receptor ligand acts on dendritic cells and T cells to suppress experimental autoimmune encephalomyelitis. Proc Natl Acad Sci U S A 2010; 107: 20768-20773.
Manicassamy S, Reizis B, Ravindran R, Nakaya H, Salazar-Gonzalez RM, Wang YC et al. Activation of beta-catenin in dendritic cells regulates immunity versus tolerance in the intestine. Science 2010; 329: 849-853.
Huang G, Shi LZ, Chi H . Regulation of JNK and p38 MAPK in the immune system: signal integration, propagation and termination. Cytokine 2009; 48: 161-169.
Cohen P . Targeting protein kinases for the development of anti-inflammatory drugs. Curr Opin Cell Biol 2009; 21: 317-324.
Chi H, Barry SP, Roth RJ, Wu JJ, Jones EA, Bennett AM et al. Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proc Natl Acad Sci U S A 2006; 103: 2274-2279.
Kim C, Sano Y, Todorova K, Carlson BA, Arpa L, Celada A et al. The kinase p38 alpha serves cell type-specific inflammatory functions in skin injury and coordinates pro- and anti-inflammatory gene expression. Nat Immunol 2008; 9: 1019-1027.
Hu JH, Chen T, Zhuang ZH, Kong L, Yu MC, Liu Y et al. Feedback control of MKP-1 expression by p38. Cell Signal 2007; 19: 393-400.
Janeway CA Jr, . Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb Symp Quant Biol 1989; 54 Pt 1: 1-13.
Marks BR, Nowyhed HN, Choi JY, Poholek AC, Odegard JM, Flavell RA et al. Thymic self-reactivity selects natural interleukin 17-producing T cells that can regulate peripheral inflammation. Nat Immunol 2009; 10: 1125-1132.
Spits H, Di Santo JP . The expanding family of innate lymphoid cells: regulators and effectors of immunity and tissue remodeling. Nat Immunol 2011; 12: 21-27.
Caton ML, Smith-Raska MR, Reizis B . Notch-RBP-J signaling controls the homeostasis of CD8- dendritic cells in the spleen. J Exp Med 2007; 204: 1653-1664.
Acknowledgements
I acknowledge the large number of researchers who have contributed to this field whose work was not cited owing to space limitations. I thank members of my laboratory for helpful discussions, and Dr John Lukens for critical reading of the manuscript. The authors' research is supported by US National Institutes of Health (K01 AR053573 and R01 NS064599), National Multiple Sclerosis Society (RG4180-A-1), Lupus Research Institute, Cancer Research Institute, and the American Lebanese Syrian Associated Charities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, G., Wang, Y. & Chi, H. Regulation of TH17 cell differentiation by innate immune signals. Cell Mol Immunol 9, 287–295 (2012). https://doi.org/10.1038/cmi.2012.10
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2012.10
Keywords
This article is cited by
-
A systematic review of dysregulated microRNAs in Hashimoto’s thyroiditis
Endocrine (2024)
-
Ginsenoside from ginseng: a promising treatment for inflammatory bowel disease
Pharmacological Reports (2021)
-
A perfect storm: fetal inflammation and the developing immune system
Pediatric Research (2020)
-
p38α has an important role in antigen cross-presentation by dendritic cells
Cellular & Molecular Immunology (2018)
-
Circulating Mycobacterium tuberculosis DosR latency antigen-specific, polyfunctional, regulatory IL10+ Th17 CD4 T-cells differentiate latent from active tuberculosis
Scientific Reports (2017)