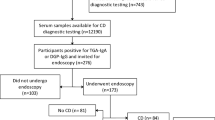
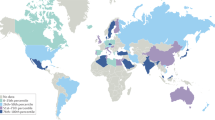
Abstract
Until a few years ago, celiac disease (CD) was thought to be a rare food intolerance that was confined to childhood and characterized by severe malabsorption and flat intestinal mucosa. Currently, CD is regarded as an autoimmune disorder that is common in the general population (affecting 1 in 100 individuals), with possible onset at any age and with many possible presentations. The identification of CD is challenging because it can begin not only with diarrhea and weight loss but also with atypical gastrointestinal (constipation and recurrent abdominal pain) and extra-intestinal symptoms (anemia, raised transaminases, osteoporosis, recurrent miscarriages, aphthous stomatitis and associated autoimmune disorders), or it could be completely symptomless. Over the last 20 years, the diagnostic accuracy of serology for CD has progressively increased with the development of highly reliable tests, such as the detection of IgA tissue transglutaminase and antiendomysial and IgG antideamidated gliadin peptide antibodies. The routine use of antibody markers has allowed researchers to discover a very high number of ‘borderline’ cases, characterized by positive serology and mild intestinal lesions or normal small intestine architecture, which can be classified as potential CD. Therefore, it is evident that the ‘old celiac disease’ with flat mucosa is only a part of the spectrum of CD. It is possible that serology could identify CD in its early stages, before the appearance of severe intestinal damage. In cases with a positive serology but with mild or absent intestinal lesions, the detection of HLA-DQ2 and HLA-DQ8 can help reinforce or exclude the diagnosis of gluten sensitivity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Di Sabatino A, Corazza GR . Coeliac disease. Lancet 2009; 373: 1480–1493.
Schuppan D . Current concepts of celiac disease pathogenesis. Gastroenterology 2000; 119: 234–242.
Shewry PR, Tatham AS, Kasarda DD . Cereal proteins and coeliac disease. In: Marsh MN (ed.) Coeliac Disease. Oxford: Blackwell Scientific, 1992: 305–348.
Janatuinen EK, Pikkarainen PH, Kemppainen TA, Kosma VM, Jarvinen RM, Uusitupa MI et al. A comparison of diets with and without oats in adults with coeliac disease. N Engl J Med 1995; 333: 1033–1037.
Hardman CM, Garioch JJ, Leonard JN, Thomas HJ, Walker MM, Lortan JE et al. Absence of toxicity of oats in patients with dermatitis herpetiformis. N Engl J Med 1997; 337: 1884–1887.
Sollid L, Thorsby E . HLA susceptibility genes in celiac disease: genetic mapping and role in pathogenesis. Gastroenterology 1993; 105: 910–922.
Partanen J . Major histocompatibility complex and coeliac disease. In: Maki M, Collin P, Visakorpi JK (eds.) Coeliac Disease. Tampere: University of Tampere, 1997: 253–264 .
Greco L, Corazza G, Babron MC, Clot F, Fulchignoni-Lataud MC, Percopo S et al. Genome search in celiac disease. Hum Genet 1998; 62: 669–675.
Holopainen P, Arvas M, Sistonen P, Mustalahti K, Collin P, Maki M et al. CD28/CTLA4 gene region on chromosome 2q33 confers genetic susceptibility to celiac disease. A linkage and family-based association study. Tissue Antigens 1999; 53: 470–475.
Dubois CA, Trynka G, Franke L, Hunt CA, Romanos J, Curtotti A et al. Multiple common variants for celiac disease influencing immune gene expression. Nat Genet 2010; 42: 295–303.
Godkin A, Jewell D . The pathogenesis of celiac disease. Gastroenterology 1998; 115: 206–210.
Dieterich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken E et al. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat Med 1997; 3: 797–801.
Molberg O, McAdam SM, Korner M, Quarsten H, Kristiansen C, Madsen L et al. Tissue transglutaminase selectively modifies gliadin peptides that are recognized by gut-derived T cells in celiac disease. Nat Med 1998; 4: 713–717.
Pittschieler K, Ladinser B . Coeliac disease screened by a new strategy. Acta Paediatr Suppl 1996; 412: 42–45.
Corazza GR, Andreani ML, Biagi F, Corrao G, Pretolani S, Giulianelli G et al. The smaller size of the ‘coeliac iceberg’ in adults. Scand J Gastroenterol 1997; 32: 917–919.
Johnston SD, Watson RG, McMillan SA, Sloan J, Love AH . Prevalence of coeliac disease in Northern Ireland. Lancet 1997; 350: 1370.
Kolho KL, Farkkila MA, Savilahti E . Undiagnosed coeliac disease is common in Finnish adults. Scand J Gastroenterol 1998; 33: 1280–1283.
Ivarsson A, Persson LA, Juto P, Peltonen M, Suhr O, Hernell O . High prevalence of undiagnosed coeliac disease in adults: a Swedish population-based study. J Intern Med 1999; 245: 63–68.
Riestra S, Fernandez E, Rodrigo L, Garcia S, Ocio G . Prevalence of coeliac disease in the general population of Northern Spain. Scand J Gastroenterol 2000; 35: 398–402.
Volta U, Bellentani S, Bianchi FB, Brandi G, de Franceschi L, Miglioli L et al. High prevalence of celiac disease in Italian general population. Dig Dis Sci 2001; 46: 1500–1505.
Mäki M, Mustalahti K, Kokkonen J, Kulmala P, Haapalahti M, Karttunen T et al. Prevalence of celiac disease among children in Finland. N Engl J Med 2003; 348: 2517–2524.
Not T, Horvath K, Hill ID, Partanen J, Hammed A, Magazzu G et al. Coeliac disease risk in USA: high prevalence of antiendomysium antibodies in healthy blood donors. Scand J Gastroenterol 1998; 33: 494–498.
Gupta R, Reddy DN, Makharia GK, Sood A, Ramakrishna BS, Yachha SK et al. Indian task force for celiac disease: current status. World J Gastroenterol 2009; 15: 6028–6033.
Catassi C, Ratsch IM, Gandolfi L, Pratesi R, Fabiani E, El Asmar R et al. Why is coeliac disease endemic in the people of the Sahara? Lancet 1999; 354: 647–648.
Maki M, Collin P . Coeliac disease. Lancet 1997; 349: 1755–1759.
Fasano A, Catassi C . Current approaches to diagnosis and treatment of celiac disease: an evolving spectrum. Gastroenterology 2001; 120: 636–651.
Farrell RJ, Kelly CP . Celiac sprue. N Engl J Med 2002; 346: 180–188.
Howdle PD, Losowsky MS . Coeliac disease in adults. In: Marsh MN (ed.) Coeliac Disease. Oxford: Blackwell Scientific, 1992: 49–80.
Gasbarrini G, Ciccocioppo R, de Vitis I, Corazza GR; Club del Tenue Study Group. Coeliac disease in the elderly. A multicentre Italian study. Gerontology 2001; 47: 306–310.
Ciclitira PJ . AGA technical review on celiac sprue. Gastroenterology 2001; 120: 1526–1540.
Trier JS . Diagnosis of celiac sprue. Gastroenterology 1998; 115: 211–216.
Volta U . Coeliac disease: recent advances in pathogenesis, diagnosis and clinical signs. Rec Prog Med 1999; 1: 37–44.
McFarlane XA, Bhalla AK, Reeves DE, Morgan LM, Robertson DA . Osteoporosis in treated adult coeliac disease. Gut 1995; 36: 710–714.
Corazza GR, Valentini RA, Andreani ML, D'Anchino M, Leva MT, Ginaldi L et al. Subclinical coeliac disease is a frequent cause of iron-deficiency anaemia. Scand J Gastroenterol 1995; 30: 153–156.
Volta U . Dallo screening alla diagnosi. In: Corazza GR (ed.) Malattia Celiaca, Educazione Permanente in Malattie Digestive. Roma: Il Pensiero Scientifico Editore, 1996; 5: 29–35 .
Cacciari E, Salardi S, Volta U, Biasco G, Lazzari R, Corazza GR et al. Can antigliadin antibody detect symptomless coeliac disease in children with short stature. Lancet 1985; 1: 1469–1471.
Aine L, Maki M, Collin P, Keyrilainen O . Dental enamel defects in celiac disease. J Oral Path Med 1990; 19: 241–245.
Ferguson R, Basu MK, Asquith P, Cooke WT . Jejunal mucosal abnormalities in patients with recurrent aphthous ulceration. BMJ 1975; 1: 11–13.
Giorgetti GM, Tursi A, Brandimarte G, Rubino C, Gasbarrini G . Dysmotility-like dyspeptic symptoms in coeliac patients: role of gluten and Helicobacter pylori infection. Dig Liv Dis 2000; 32: 73–74.
Sanders DS, Carter MJ, Hurlstone DP, Pearce A, Ward AM, McAlindon ME et al. Association of adult coeliac disease with irritable bowel syndrome: a case–control study in patients fulfilling ROME II criteria referred to secondary care. Lancet 2001; 358: 1504–1508.
Hallert C, Derefeldt T . Psychi disturbances in adult coeliac disease. Clinical observations. Scand J Gastroenterol 1982; 17: 17–19.
Gobbi G, Bouquet F, Greco L, Lambertini A, Tassinari CA, Ventura A et al. Coeliac disease, epilepsy, and cerebral calcifications. Lancet 1992; 340: 439–443. .
Hadjivassiliou M, Gibson A, Davies-Jones GA, Lobo AJ, Stephenson TJ, Milford-Ward A . Does cryptic gluten sensitivity play a part in neurological illness? Lancet 1996; 347: 369–371.
Luorostainen L, Pirttila T, Collin P . Coeliac disease presenting with neurological disorders. Eur Neurol 1999; 42: 132–135.
Fantelli FJ, Mitsumoto H, Sebek BA . Multiple sclerosis and malabsorption. Lancet 1978; 311: 1039–1040.
Volta U, de Giorgio R . Gluten sensitivity: an emerging issue behind neurological impairment? Lancet Neurol 2010; 9: 233–235.
Ferguson R, Holmes GK, Cooke WT . Coeliac disease, fertility and pregnancy. Scand J Gastroenterol 1982; 17: 65–68.
Sher KS, Mayberry JF . Female fertility, obstetric and gynecological history in coeliac disease: a case control study. Digestion 1994; 55: 243–246.
Farthing MJ, Edwards CR, Rees LH, Dawson AM . Male gonadal function in coeliac disease: 1 sexual dysfunction, infertility and semen quality. Gut 1982; 23: 608–614.
Farthing MJ, Rees LH, Edwards CR, Dawson AM . Male gonadal function in coeliac disease: 2 sexual dysfunction, infertility and semen quality. Gut 1982; 24: 127–135.
Volta U, de Franceschi L, Lari F, Molinaro N, Zoli M, Bianchi FB . Coeliac disease hidden by cryptogenic hypertransaminasemia. Lancet 1998; 352: 26–29.
Cooper BT, Holmes GK, Cooke WT . Coeliac disease and immunological disorders. BMJ 1978; 1: 537–539.
Collin P, Reunala T, Pukkala E, Laippala P, Keyrilainen O, Pasternack A . Coeliac disease, associated disorders and survival. Gut 1994; 35: 1215–1218.
Reunala T, Collin P . Diseases associated with dermatitis herpetiformis. Br J Dermatol 1997; 136: 315–318.
Cronin CC, Shanahan F . Insulin-dependent diabetes mellitus and coeliac disease. Lancet 1997; 349: 1096–1097.
Counsell CE, Taha A, Ruddell WS . Coeliac disease and autoimmune thyroid disease. Gut 1994; 35: 844–846.
Volta U, de Franceschi L, Molinaro N, Cassani F, Muratori L, Lenzi M et al. Frequency and significance of anti-gliadin and anti-endomysial antibodies in autoimmune hepatitis. Dig Dis Sci 1998; 43: 2190–2195.
Volta U, Rodrigo L, Granito L, Petrolini L, Muratori P, Muratori P et al. Celiac disease in autoimmune cholestatic liver disorders. Am J Gastroenterol 2002; 97: 2609–2613.
Volta U, Bardazzi F, Zauli D, de Franceschi L, Tosti A, Molinaro N et al. Serological screening for coeliac disease in vitiligo and alopecia areata. Br J Dermatol 1997; 136: 801–802.
O'Leary C, Walsh CH, Wieneke P, O'Regan P, Buckley B, O'Halloran DJ et al. Coeliac disease and autoimmune Addison's disease: a clinical pitfall. QJM 2002; 95: 79–82.
Henriksson KG, Hallert C, Walan A . Gluten-sensitive polymyositis and enteropathy. Lancet 1976; 2: 317.
Teppo AM, Maury CP . Antibodies to gliadin, gluten and reticulin glycoprotein in rheumatic diseases: elevated levels in Sjogren's disease. Clin Exp Immunol 1984; 57: 73–78.
O'Farrelly C, Marten M, Melcher D, McDougall B, Price R, Goldstein AJ et al. Association between villous atrophy in rheumatoid arthritis and a rheumatoid factor and gliadin-specific IgG. Lancet 1988; 2: 819–822.
Fornasieri A, Sinco RA, Maldifassi P, Bernasconi P, Vegni M, D'Amico G . IgA-antigliadin antibodies in IgA mesangial nephropathy (Berger's disease). BMJ 1987; 295: 78–80.
Cataldo F, Marino V, Ventura A, Bottaro G, Corazza G . Prevalence and clinical features of selective immunoglobulin A deficiency in coeliac disease: an Italian multicentre study. Gut 1998; 42: 362–365.
Curione M, Barbato M, de Biase L, Viola F, Lo Russo L, Cardi E . Prevalence of coeliac disease in idiopathic dilated cardiomiopathy. Lancet 1999; 354: 222–223.
Zauli D, Grassi A, Granito A, Foderaro S, de Franceschi L, Ballardini G et al. Prevalence of silent coeliac disease in atopics. Dig Dis Sci 2000; 32: 775–779.
Kitis G, Holmes GK, Cooper BT, Thompson H, Allan RN . Association of coeliac disease and inflammatory bowel disease. Gut 1980; 21: 636–641.
Gale L, Wimalaratna H, Brotodiharjo A, Duggan JM . Down's syndrome is strongly associated with coeliac disease. Gut 1997; 40: 492–496.
Bonamico M . Celiac disease and Turner syndrome. J Pediatr Gastroenterol 1999; 29: 107–108.
Giannotti A, Tiberio G, Castro M, Virgilii F, Colistro F, Ferretti F et al. Coeliac disease in Williams syndrome. J Med Genet 2001; 38: 767–768.
Corazza GR, Valentini RA, Frisoni M, Volta U, Corrao G, Bianchi FB et al. Gliadin immune reactivity is associated with overt and latent enteropathy in relatives of celiac patients. Gastroenterology 1992; 103: 1517–1522.
Maki M, Holm K, Collin P, Savilahti E . Increase in γ/δ T cell receptor bearing lymphocytes in normal small bowel mucosa in latent coeliac disease. Gut 1991; 32: 1412–1414.
Arranz E, Bode J, Kingstone K, Ferguson A . Intestinal antibody pattern of coeliac disease: association with γ/δ T cell receptor expression by intraepithelial lymphocytes, and other indices of potential coeliac disease. Gut 1994; 35: 476–482.
Shiner M . Duodenal biopsy. Lancet 1956; 1: 17–19.
Upton MP . ‘Give us this day our daily bread’. Evolving concepts in celiac sprue. Arch Pathol Lab Med 2008; 132: 1594–1599.
Villanacci V . The problem of biopsies in the diagnosis of celiac disease. Gastrointest Endosc 2009; 69: 983–984.
Brown I, Mino-Keduson M, Deshpande V, Lauwers GY . Intraepithelial lymphocytosis in architecturally preserved proximal small intestinal mucosa. Arch Pathol Lab Med 2006; 130: 1020–1025.
Corazza GR, Villanacci V, Zambelli C, Milione M, Luinetti O, Vindigni C et al. Comparison of the interobserver reproducibility with different histologic criteria used in celiac disease. Clin Gastroenterol Hepatol 2007; 5: 838–843.
Corazza GR, Villanacci V . Coeliac disease. J Clin Pathol 2005; 58: 573–574.
Kakar S, Nehra V, Murray JA, Dayharsh GA, Burgart LJ . Significance of intraepithelial lymphocytosis in small bowel biopsy samples with normal mucosa architecture. Am J Gastroenterol 2003; 98: 2027–2033.
Paparo F, Petrone E, Tosco A, Maglio M, Borrelli M, Salvati VM et al. Clinical, HLA and small boel immunohistochemical features of children with positive serum antiendomysium antibodies and architecturally normal small intestinal mucosa. Am J Gastroeneterol 2005; 100: 2294–2298.
Oberhuber G, Grandtisch G, Vogelsang H . The histopathology of celiac disease: time for standardized report scheme for pathologists. Eur J Gastroenterol Hepatol 1999; 11: 1185–1194.
Villanacci V, Catassi C, Rostami K, Volta U . Celiac disease: changing dogma on historical diagnosis. GastroHep 2010 ; January 13. http://www.gastrohep.com/freespeech/freespeech.asp?id=128
Dieterich W, Laag E, Schopper H, Volta U, Ferguson A, Gillet H et al. Autoantibodies to tissue transglutaminase as predictors of celiac disease. Gastroenterology 1998; 115: 1317–1321.
Volta U, Fabbri A, Parisi C, Piscaglia M, Caio G, Tovoli F et al. Old and new serological tests for celiac disease screening. Expert Rev Gastroenterol Hepatol 2010; 4: 31–35.
Stern M, Working Group on Serologic Screening for Celiac Disease. Comparative evaluation of serologic tests for celiac disease: a European iniative toward standardization. J Pediatr Gastroenterol Nutr 2000; 31: 513–519.
Lagerqvist C, Dahlbom I, Hansson T, Jidell E, Juto P, Olcén P et al. Antigliadin immunoglobulin A best in finding celiac disease in children younger than 18 months of age. J Pediatr Gastroenterol Nutr 2008; 47: 428–435.
Cataldo F, Marino V, Ventura A, Bottaro G, Corazza GR . Prevalence and clinical features of selective immunoglobulin A deficiency in coeliac disease: an Italian multicentre study. Gut 1998; 42: 362–365.
Volta U, Granito A, Parisi C, Fabbri A, Fiorini E, Piscaglia M et al. Deamidated gliadin peptide antibodies as a routine test for celiac disease: a prospective analysis. J Clin Gastroenterol 2010; 44: 186–190.
Salmi TT, Collin P, Järvinen O, Haimila K, Partanen J, Laurila K et al. Immunoglobulin A autoanttibodies against transglutaminase 2 in the small intestinal mucosa predict forthcoming celiac disease. Aliment Pharmacol Ther 2006; 24: 541–552.
Maglio M, Florian F, Vecchiet M, Auricchio R, Paparo F, Spadaro R et al. Majority of children with type 1 diabetes produce and deposit anti-tissue transglutaminase antibodies in the small intestine. Diabetes 2009; 58: 1578–1584.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volta, U., Villanacci, V. Celiac disease: diagnostic criteria in progress. Cell Mol Immunol 8, 96–102 (2011). https://doi.org/10.1038/cmi.2010.64
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2010.64
Keywords
This article is cited by
-
Genetic, Immunological, Dietary, Gut Microbiota, and Environmental Determinants of Osteoporosis in the Course of Celiac Disease: Which Factor Plays the First Violin in This Orchestra?
Calcified Tissue International (2023)
-
29/w mit Bauchschmerzen, geblähtem Abdomen und gelegentlichen Diarrhöen
Der Internist (2022)
-
Diagnosing and Monitoring Celiac Patients with Selective IgA Deficiency: Still an Open Issue
Digestive Diseases and Sciences (2021)
-
Value of biopsy in a cohort of children with high-titer celiac serologies: observation of dynamic policy differences between Europe and North America
BMC Health Services Research (2020)
-
Back to the roots: dental calculus analysis of the first documented case of coeliac disease
Archaeological and Anthropological Sciences (2020)