Abstract
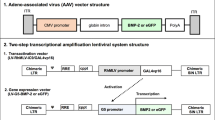
A number of hormonal factors have been shown to be instrumental in the calcification process. This study represents an attempt at using one of these factors to specifically induce calcification of tumors to arrest tumor growth. The gene encoding bone morphogenetic protein 2 (BMP-2) was placed under transcriptional control of the promoter for carcinoembryonic antigen (CEA). This gene cassette was cloned into a herpes simplex virus (HSV) amplicon vector (HSV–CEA–BMP2). This vector was used to induce local BMP-2 production in CEA-expressing tumor cells to retard cell growth. Lysates of tumor cells treated with HSV–CEA–BMP2 were applied to stem cells to determine if BMP-2 expression promotes differentiation to bone lineage. pHSV–CEA–BMP2 efficiently transduced both CEA-expressing and non-expressing cells. BMP-2 was only expressed in CEA-positive cells. BMP-2 expression led to an inhibition of tumor cell growth. BMP-2 released by these CEA-expressing tumors also drove the differentiation of mesenchymal stem cells to bone lineage. This proof-of-concept study demonstrates that tumor cells can be specifically engineered to produce BMP-2, which leads to growth retardation and to the differentiation of non-committed stem cells to bone. This ‘Medusa’ effect can theoretically be exploited to retard tumor growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kypson AP, Morphew E, Jones R, Gottfried MR, Seigler HF . Heterotopic ossification in rectal cancer: rare finding with a novel proposed mechanism. J Surg Oncol 2003; 82: 132–136.
Al-Maghrabi H, Jamison BM, Veinot JP . Calcified colonic mass. Arch Pathol Lab Med 2005; 129: 1347–1348.
Randall JC, Morris DC, Tomita T, Anderson HC . Heterotopic ossification: a case report and immunohistochemical observations. Hum Pathol 1989; 20: 86–88.
Hruska KA, Mathew S, Saab G . Bone morphogenetic proteins in vascular calcification. Circ Res 2005; 97: 105–114.
Ryoo HM, Lee MH, Kim YJ . Critical molecular switches involved in BMP-2-induced osteogenic differentiation of mesenchymal cells. Gene 2006; 366: 51–57.
Hanada K, Dennis JE, Caplan AI . Stimulatory effects of basic fibroblast growth factor and bone morphogenetic protein-2 on osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells. J Bone Miner Res 1997; 12: 1606–1614.
Ghosh-Choudhury N, Ghosh-Choudhury G, Celeste A, Ghosh PM, Moyer M, Abboud SL et al. Bone morphogenetic protein-2 induces cyclin kinase inhibitor p21 and hypophosphorylation of retinoblastoma protein in estradiol-treated MCF-7 human breast cancer cells. Biochim Biophys Acta 2000; 1497: 186–196.
Waite KA, Eng C . BMP2 exposure results in decreased PTEN protein degradation and increased PTEN levels. Hum Mol Genet 2003; 12: 679–684.
Pouliot F, Labrie C . Role of Smad1 and Smad4 proteins in the induction of p21WAF1, Cip1 during bone morphogenetic protein-induced growth arrest in human breast cancer cells. J Endocrinol 2002; 172: 187–198.
Epstein AL, Marconi P, Argnani R, Manservigi R . HSV-1-derived recombinant and amplicon vectors for gene transfer and gene therapy. Curr Gene Ther 2005; 5: 445–458.
Epstein AL . Present and future of herpes simplex virus type 1 (HSV-1)-based amplicon vectors. Pathol Biol (Paris) 2005; 53: 1–3.
Reinblatt M, Pin RH, Fong Y . Carcinoembryonic antigen directed herpes viral oncolysis improves selectivity and activity in colorectal cancer. Surgery 2004; 136: 579–584.
Mesri EA, Federoff HJ, Brownlee M . Expression of vascular endothelial growth factor from a defective herpes simplex virus type 1 amplicon vector induces angiogenesis in mice. Cir Res 1995; 76: 161–167.
Bowers WJ, Howard DF, Brooks AI, Halterman MW, Federoff HJ . Expression of vhs and VP16 during HSV-1 helper virus-free amplicon packaging enhances titers. Gene Therapy 2001; 8: 111–120.
Bowers WJ, Howard DF, Federoff HJ . Discordance between expression and genome transfer titering of HSV amplicon vectors: recommendation for standardized enumeration. Mol Ther 2000; 1: 294–299.
Geller AI, Breakefield XO . A defective HSV-1 vector expresses Escherichia coli beta-galactosidase in cultured peripheral neurons. Science 1988; 241: 1667–1669.
Ackerman GA . Substituted naphthol AS phosphate derivatives for the localization of leukocyte alkaline phosphatase activity. Lab Invest 1962; 11: 563–567.
Huggins CB . Nobel Lecture: Endocrine-Induced Regression of Cancers. Science 1967; 156: 1050–1054.
Reddi AH, Ma SS, Cunningham NS . Induction and maintenance of new bone formation by growth and differentiation factors. Ann Chir Gynaecol 1988; 77: 189–192.
Reddi AH, Muthukumaran N, Ma S, Carrington JL, Luyten FP, Paralkar VM et al. Initiation of bone development by osteogenin and promotion by growth factors. Connect Tissue Res 1989; 20: 303–312.
Reddi AH, Cunningham NS . Bone induction by osteogenin and bone morphogenetic proteins. Biomaterials 1990; 11: 33–34.
Federoff H . Growth of replication defective herpes virus amplicon vectors and their use for gene transfer. In: Spector (ed). Cell Biology: A Laboratory Manual. CSH Press: Woodbury, NY, 1997.
Advani SJ, Weichselbaum RR, Whitley RJ, Roizman B . Friendly fire: redirecting herpes simplex virus-1 for therapeutic applications. Clin Microbiol Infect 2002; 8: 551–563.
Herrlinger U, Kramm CM, Aboody-Guterman KS, Silver JS, Ikeda K, Johnston KM et al. Pre-existing herpes simplex virus 1 (HSV-1) immunity decreases, but does not abolish, gene transfer to experimental brain tumors by a HSV-1 vector. Gene Therapy 1998; 5: 809–819.
Delman KA, Bennett JJ, Zager JS, Burt BM, McAuliffe PF, Petrowsky H et al. Effects of preexisting immunity on the response to herpes simplex-based oncolytic viral therapy. Human Gene Therapy 2000; 11: 2465–2472.
Li Y, Chen Y, Dilley J, Arroyo T, Ko D, Working P et al. Carcinoembryonic antigen-producing cell-specific oncolytic adenovirus, OV798, for colorectal cancer therapy. Mol Cancer Ther 2003; 2: 1003–1009.
Albores-Saavedra J, Nadji M, Morales AR, Henson DE . Carcinoembryonic antigen in normal, preneoplastic and neoplastic gallbladder epithelium. Cancer 1983; 52: 1069–1072.
Khoo S, Warner N, Lie J et al. CEA activity in tissue extracts: a quantitative study of malignant and benign neoplasms, cirrhotic liver, normal adults and fetal organs. Int J Cancer 1973; 11: 681–687.
Xiang R, Silletti S, Lode HN, Dolman CS, Ruehlmann JM, Niethammer AG et al. Protective immunity against human carcinoembryonic antigen (CEA) induced by an oral DNA vaccine in CEA-transgenic mice. Clin Cancer Res 2001; 7 (3 Suppl): 856s–864s.
Acknowledgements
We thank Ann Casey, Clark Burris and Louis Lotta (University of Rochester) for helper virus-free amplicon packaging. We would also like to thank Meryl Greenblatt (MSKCC) for her editorial expertise.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fong, S., Chan, MK., Fong, A. et al. Viral vector-induced expression of bone morphogenetic protein 2 produces inhibition of tumor growth and bone differentiation of stem cells. Cancer Gene Ther 17, 80–85 (2010). https://doi.org/10.1038/cgt.2009.56
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2009.56
Keywords
This article is cited by
-
BMP-2 inhibits tumor-initiating ability in human renal cancer stem cells and induces bone formation
Journal of Cancer Research and Clinical Oncology (2015)