Abstract
It is generally believed that some inflammatory antigens can recognize Toll-like receptors on synovial fibroblasts (SFs) and then activate downstream signals, leading to the formation of RASFs and inducing rheumatoid arthritis (RA). The objective of the current work was to study on the hypothesis that outer PAMP (LPS) binds to the inner DAMP (HMGB1) and becomes a complex that recognizes TLRs/RAGE on SFs, thus initiating a signaling cascade that leads to the secretion of inflammatory cytokines and chemokines, production of tissue-destructive enzymes, and formation of RASFs, finally resulting in RA. Osteoarthritis synovial fibroblasts (OASFs) were co-cultured with HMGB1–LPS complex in vitro for five generations to induce the transformation of human SFs to RA-like SFs (tOASFs). Then, changes of tOASFs in cell cycle and apoptosis–autophagy balance were investigated in vitro, and the pathogenicity of tOASFs was evaluated in a SCID mouse model in vivo. In vitro cell cycle analysis showed more tOASFs passing through the G1/S checkpoint and moving to S or G2 phase. Flow cytometry and confocal microscopy showed that apoptosis was reduced and autophagy was enhanced significantly in tOASFs as compared with those in OASFs. The expression of certain receptors and adhesion molecules in tOASFs was upregulated. In vivo experiments showed that tOASFs attached to, invaded, and degraded the co-implanted cartilage. In addition, histochemistry showed excessive proliferation of tOASFs and the expression of matrix metalloproteinases (MMPs). Based on the above findings, we conclude that HMGB1–LPS complex could promote the formation of RASFs.
Similar content being viewed by others
Main
Rheumatoid arthritis (RA) is a common autoimmune disease causing progressive disability, systemic complications, early death, and socioeconomic hardship. The etiology and specific pathogenesis of RA remain unclear. It is generally believed that some inflammatory antigens and their metabolites recognize Toll-like receptors on synovial fibroblasts (SFs) and activate downstream signals, which induces the secretion of inflammatory cytokines and chemokines, production of tissue-destructive enzymes, and formation of RASFs, finally leading to RA.1, 2, 3 The transformation of normal SFs to RASFs is crucial in the pathogenesis of RA, because RASFs are recognized as both the propagator of immune response and the engine of joint damage in RA. SFs from RA patients exhibit characteristics of transformed cells, including anchorage-independent growth, insensitivity to apoptosis, and increased proliferation.1, 3
However, the mechanism underlying the change from normal SFs to the aggressive behavior remains unclear. Infections may contribute to the onset of RA, for example, patients with periodontal disease have been shown to have a higher prevalence of RA than patients without periodontitis, and several virulence factors, including the bacterial lipopolysaccharides (LPS), toxins, and hemagglutinins of the main pathogen—P. gingivalis, may have a role in the pathogenesis of RA.4, 5 Group A streptococcal cell wall peptidoglycan–polysaccharide complexes can induce acute inflammation of the peripheral joints resulting in chronic erosive arthritis in susceptible rats,6, 7 which indicates that microorganism components can induce the onset of RA. Our previous studies8, 9 showed that the high mobility group box 1 (HMGB1)–LPS complex directly induced experimental arthritis in DBA/1 mice. In addition, pathological study8 showed synovial thickening and excessive proliferation of SFs, suggesting that the HMGB1–pathogen-associated molecular pattern (PAMP) complex might induce the formation of RASFs. In the current study, osteoarthritis synovial fibroblasts (OASFs) were isolated from the tissue obtained during reconstructive surgery and co-cultured with HMGB1–LPS complex in vitro for 5–7 generations to induce the transformation of normal SFs to RA-like SFs (tOASFs). Then, pathogenicity of tOASFs was evaluated both in vitro and in vivo. The result showed that tOASFs attached to, invaded, and degraded the co-implanted cartilage in a SCID mouse model. Histochemistry further showed excessive proliferation of tOASFs and the expression of MMPs. In vitro cell cycle analysis showed more tOASFs passing through the G1/S checkpoint and moving to S or G2 phase. Flow cytometry and confocal microscopy showed that apoptosis was reduced and autophagy was enhanced significantly in tOASFs as compared with those in OASFs. We further showed that the expression of certain receptors was upregulated in tOASFs, including TLR2, TLR4, and receptor for advanced glycation end products (RAGE), and the adhesion molecules ICAM-1 and VCAM-1. In conclusion, we have proved that HMGB1–LPS complex could promote the formation of RA-like transformed SFs.
Results
Cell cycle, apoptosis, and autophagy balance disorders in tOASFs
Many studies have demonstrated that excessive proliferation and invasive growth of RASFs are attributed to impaired regulation of the cell cycle and imbalance between survival (autophagy) and death (apoptosis).10, 11 To evaluate the change of cell cycle in tOASFs, cells first underwent serum starvation to synchronize cell cycle at G0/1 phage (Supplementary Figure S1) and then were further cultured for 24 h with DMEM containing 10% FBS. Cells were collected, fixed, stained with propidium iodide (PI), and then analyzed by flow cytometry. As shown in Figure 1a, more tOASF cells passed through the G1/S checkpoint and moved into S or G2 phase as compared with OASFs, suggesting that accelerated proliferation occurred in tOASFs.
Transformed OASFs showed a RASF-like response in cell cycle, apoptosis and autophagy. (a) Cells were cultured with DMEM containing 1% heat-activated FBS for 24 h, then media were replaced with DMEM containing 10% heat-activated FBS and cultured for another 4 h, followed by collection and staining with PI for cell cycle analysis. (b) Cells were exposed to apoptosis-inducer agents for 8 h, and then the cells were collected for apoptosis analysis using a BD FACSCalibur cytometer. (c) Cells were grown on polylysine-treated coverslips in a six-well plate and cultured with DMEM containing 10% heat-activated FBS for 24 h. Then media were replaced with PBS for 8 h to induce autophagy. The coverslips were then stained and observed for LC3II under a confocal microscope (LEICA TCS SP2). Nuclei were visualized with DAPI. (d) Cells of panels (b) and (c) were lysed, and key proteins were studied with immunoblotting. Data are representative of three independent experiments
A characteristic feature of RASFs is their decreased susceptibility to apoptosis and the increased ability to survive upon certain stimulations.12, 13, 14 To further characterize the survival–apoptosis imbalance in tOASFs, cells were exposed to an apoptosis-inducing agent for 8 h, followed by staining with Annexin V-FITC/PI and analysis of cell apoptosis using flow cytometry. As shown in Figure 1b, fewer tOASFs underwent apoptosis as compared with OASFs, indicating that the survival–apoptosis balance was impaired in tOASFs and that more tOASFs could survive a challenge by harmful stimuli. Immunoblotting also showed that pro-survival protein (BCL-2) was upregulated, whereas pro-apoptosis protein (Bax) was downregulated in tOASFs (Figure 1c).
Much research has indicated that HMGB1 is a critical regulator of autophagy, as HMGB1 translocation induces autophagy after prolonged cellular stress.15, 16 Also, RASFs were proved to go through autophagy to avoid cell death upon certain stimulations, such as starvation and rapamycin treatment.17, 18 To investigate the ability of tOASFs to induce autophagy under certain stimuli, OASFs, tOASFs, and RASFs were put in PBS for 4 h, and then detected for LC3II expression with a confocal microscope. As shown in Figure 1d, upon 4 h of culture in the absence of serum and nutrients (starvation), more autophagy-punctuated fluorescent spots were observed in tOASFs and RASFs as compared with those in OASFs. Immunoblotting also showed that the amount of LC3II in tOASFs was significantly higher than that in OASFs (Figure 1c), indicating that autophagy was induced in tOASFs upon certain harmful stimulations as a means of survival.
HMGB1–LPS complex-associated receptors are upregulated in tOASFs
HMGB1 was reported to bind to and signal through RAGE,19 as well as TLR2 and TLR4,20 and upregulated Toll-like receptors were observed on the surface of RASFs. RAGE is a multiligand receptor that binds structurally to diverse molecules, not only including HMGB1 but also S100 family members and amyloid-β. RAGE activation has been implicated in sterile inflammation, cancer, diabetes, and Alzheimer’s disease. To evaluate the expression of HMGB1–LPS complex-associated receptors in tOASFs, cells were stained with fluorescent-labeled antibodies and analyzed by flow cytometry. As shown in Figure 2, the expression levels of RAGE, TLR2, and TLR4 were significantly upregulated in tOASFs as compared with OASFs.
The expressions of RAGE, TLR 2, and TLR4 were upregulated in tOASFs. Cells were collected and stained with PE-antiTLR2, APC-antiTLR4, or PE-antiRAGE, and then the expressions of RAGE, TLR2, and TLR4 on the cell surface were studied using a BD FACSCalibur cytometer. (a) Representative histograms and (b) the percentage of positive cells are shown. Data are expressed as mean±S.D. (n=3). *P<0.05 compared with OASFs (one-way analysis of variance and Newman–Keuls multiple comparison test)
Chemokines and adhesion molecules are upregulated in tOASFs
Chemokines are of special interest in RASFs activation and function. In particular, chemokines expressed and secreted by SFs such as CCL2 and CXCL12 have a key role in driving chronic inflammation by attracting monocytes and lymphocytes into the joint and by stimulating SFs to produce proinflammatory cytokines in RA.21, 22, 23, 24 To evaluate the expression of chemokines in tOASFs, cells were fixed and stained with fluorescent-labeled antibodies and analyzed by flow cytometry. As shown in Figure 3a, the expression of CCL2 and CXCL12 in tOASFs was upregulated as compared with OASFs, indicating that tOASFs express chemokines to recruit immune cells such as lymphocytes or macrophages to the joint.
Chemokines and adhesion molecules were upregulated in tOASFs. The expression levels of chemokines (CCL2 and CXCL12) and adhesion molecules (ICAM-1 and VCAM-1) were assessed by flow cytometry. (a) Representative histograms and (b) the percentage of the positive cells were shown. Data are expressed as mean±S.D. (n=3). *P<0.05 compared with OASFs (one-way analysis of variance and Newman–Keuls multiple comparison test)
About two decades ago, researchers found that adhesion molecules are constitutively expressed on SFs and are significantly upregulated in RASFs.25, 26 Upregulation of adhesion molecules on the surface of RASFs has a key role in recruitment and infiltration of lymphocytes at the sites of inflammation, and the activation of OASFs often requires direct cell–cell interactions mediated by adhesion molecules, such as ICAM-1 or VCAM-1.27, 28 To evaluate the expression of the adhesion molecules ICAM-1 and VCAM-1 in tOASFs, cells were stained with fluorescent-labeled antibodies and analyzed by flow cytometry. As shown in Figure 3b, the expression levels of these adhesion molecules were significantly upregulated in tOASFs as compared with OASFs, indicating that the HMGB1–LPS complex mediated the upregulation of adhesion molecules in tOASFs.
Changes in activation levels of inflammatory-associated transcription factors in tOASFs
Transcription factors have critical roles in the function of RASFs, including cytokine/chemokine expression, MMPs expression, and the control of synovial cell apoptosis/autophagy.29 It has been well established that the altered morphology and the aggressive behavior of RASFs mirror specific alterations in the activation levels of several transcription factors and intracellular signaling proteins, such as NF-κB and p38 mitogen-activated protein kinase (MAPK).27, 30, 31 Using a commercial pathway scan kit (Figure 4) and western blotting (Supplementary Figure S2), the activation levels of inflammatory-associated key intracellular signaling proteins in tOASFs were evaluated. The results showed that the activation levels of NF-κB, SAPK/JNK, and p38 MAPK were significantly increased in tOASFs as compared with OASFs. However, the activation level of STAT3 was significantly decreased in tOASFs as compared with OASFs or RASFs.
Activation levels of inflammation-associated transcription factors. The fifth-passage cells were harvested and lysed with lysis buffer, and then the phosphorylation levels of key transcription factors (NF-κB, JNK, MAPK p38, Stat3, and IκB) were detected with a CST's PathScan Inflammation Multi-Target Sandwich ELISA Kit. **P<0.01, compared with OASFs (one-way analysis of variance and Newman–Keuls multiple comparison test). Data are representative of three independent experiments. NS, not significant
Inflammatory cytokines and MMPs are upregulated in tOASFs
RASFs are shown to express a high level of inflammatory cytokines, chemokines, and MMPs, which, in turn, results in stabilizing the activated phenotype of RASFs. Inflammatory factors, such as TNF-α, IL-1β, and IL-6, can strongly enhance the proinflammatory and matrix-destructive properties of RASFs.2, 32 To evaluate the activation level of tOASFs, cells were collected, and the expression of proinflammatory cytokines and MMPs was measured with real-time RT-PCR. Meanwhile, the culture supernatants were collected, and the secretion of proinflammatory cytokines and MMPs was measured with ELISA. The results in Figure 5 show that tOASFs exhibited a RASFs-like phenotype that expressed and secreted a high level of proinflammatory cytokines (TNF-α, IL-1β, and IL-6) and MMPs (MMP-3 and MMP-13).
Expression and secretion of proinflammatory cytokines, MMPs, and chemokines were upregulated in tOASFs. The fifth-passage cells were cultured in triplicate in 24-well plates for 4 h (mRNA analysis) or 24 h (ELISA analysis). (a) Then cells were collected for detection of expression of proinflammatory cytokines (TNF-α, IL-1β, and IL-6), MMPs (MMP-3 and MMP-13), and a chemokine (CCL2) with real-time RT-PCR; (b) the supernatants were collected for detection of secretion of proinflammatory cytokines (TNF-α, IL-1β, and IL-6) and MMPs (MMP-3 and MMP-13) with ELISA. Data are representative of three independent experiments. *P<0.05, **P<0.01, compared with tOASFs (one-way analysis of variance and Newman–Keuls multiple comparison test)
In vitro-transformed OASFs induce the degradation of cartilage in a SCID mouse model
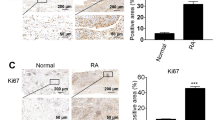
RASFs are producers of matrix-degrading enzymes that mediate articular cartilage destruction. These cells have been shown to attach to, invade, and degrade cartilage and bone in vitro. To evaluate the pathogenicity of tOASFs relative to their ability to attach to and destroy cartilage, a cartilage–sponge complex that contained tOASFs was implanted subcutaneously in SCID mice. Cartilage–sponge complexes containing OASFs or RASFs were also implanted subcutaneously in SCID mice as controls. The implants were removed after 60 days and then cut, sectioned, and stained for histological evaluation (Figures 6a and b). The pathological index of the co-implants with tOASFs was significantly higher than that of the co-implants with OASFs (P<0.01). Similar to RASFs, tOASFs showed excessive proliferation with attachment to and degradation of co-implanted cartilage (Figures 6b and c). Alcian blue special and Masson’s trichrome staining showed that chondrocytes and perichondrocytic cartilage degraded in the co-implanted cartilage of both RASFs and tOASFs. Immunohistochemistry detecting human-specific pro-matrix metalloproteinase-13 (proMMP-13) showed that both tOASFs and RASFs expressed excessive proMMP-13, causing degradation of the co-implanted cartilage (Figure 6c).
Transformed OASFs invading the cartilage in an inverse wrap SCID mouse model. (a) Schematic showing the inverse wrap SCID mouse model of RA. Following embedding of the normal cartilage into the inert sponge and soaking the sponge in different synovial fibroblasts, the implants were then inserted under the skin of a SCID mouse. After 60 days, the implants were removed and stained for histological evaluation of cartilage degradation. (b) Invasion scores show that tOASFs induce co-implantation cartilage degradation in a manner similar to RASFs. **P<0.01, error bars represent S.D. (c) Histology (H&E, Alcian blue, and Masson’s trichrome staining) showing tOASFs invasion and chondrocytic degradation. Immunohistochemistry used human-specific antibodies to detect proMMP-13 in the co-implantation cartilage. Data are representative of two independent experiments, with two mice/group
Discussion
HMGB1 that was first discovered as a nuclear protein with rapid electrophoretic migration is a highly conserved 30-kDa nonhistone DNA-binding molecule.33, 34 It acts on various cells and interacts with many different types of ligands, including RAGE and TLR4, to modulate pleiotropic functions of these entities in various physiological and pathological situations.35, 36 In particular, an interaction between HMGB1 and TLR2 or TLR4 postulates that it may mediate the proinflammatory actions of HMGB1.20, 37, 38, 39 Knowing that HMGB1 has pleiotropic functions, many researchers have suggested that it may have a crucial role in various pathophysiological processes, including RA,40, 41 liver injury,42 and tumors.43 RA is characterized by persistent synovitis, systemic inflammation, and autoantibodies, particularly to rheumatoid factor and citrullinated peptide. In industrialized countries, RA affects 0·5–1·0% of adults, with 5–50 per 100 000 new cases annually. The disorder is most typical in women and elderly people. Uncontrolled active RA causes joint damage, disability, decreased quality of life, and cardiovascular and other co-morbidities.1
Autoimmune disease can develop as a result of a breakdown in immunological tolerance, leading to the activation of self-reactive immune response. An established link exists between infection and human autoimmune diseases. In addition, experimental autoimmune diseases can be induced by autoantigens that are administered together with complete Freund’s adjuvant, which contains killed Mycobacterium tuberculosis. In some cases, these bacteria can be replaced by individual PAMPs.44 Although the exact cause for RA remains elusive, infectious agents, such as viruses, bacteria, and fungi, have long been suspected. Our previous studies8 showed that HMGB1–LPS complex directly induced experimental synovitis in DBA/1 mice and that HMGB1 is a well-verified adjuvant-like damage-associated molecular pattern (DAMP) protein.45, 46, 47 Taking these observations into consideration, we hypothesized a signaling cascade in which outer PAMP (LPS) bound to the inner DAMP (HMGB1) becomes a complex that recognizes the TLRs/RAGE on SFs, leading to the activation of downstream signals, which, in turn, causes the secretion of inflammatory cytokines and chemokines, the production of tissue-destructive enzymes, and the formation of RASFs, finally inducing RA (Figure 7). In the present study, we have demonstrated that HMGB1–LPS complex mediated the transformation of OASFs to RASFs-like phenotypes, including alterations in morphology and behavior, molecular changes in signal transduction, apoptosis/autophagy responses, and the expression of adhesion molecules, as well as matrix-degrading enzymes.
RASFs are one of the key players in the destructive process of RA. RASFs contribute primarily to the progression of the disease by attaching to, invading, and degrading cartilage and bone.10, 48 As there was a limited source to get normal SFs, we used relatively normal OASFs to perform our experiments. We first isolated OASFs from the tissue obtained during reconstructive surgery and co-cultured it with HMGB1–LPS complex in vitro for five generations, expecting that the HMGB1–LPS complex would mediate the transformation of OASFs. RASFs are tumor-like cells with an enhanced proliferation.32 We then investigated the cell cycle, apoptosis, and autophagy of tOASFs in vitro. The results of our experiment confirmed that HMGB1–LPS complex accelerated cell proliferation and prompted OASFs to undergo autophagy and thus survive starvation. The activation of TLRs and RAGE was found to be involved in the pathogenesis of RA, resulting in the production of inflammatory cytokines, chemokines, and tissue-destructive enzymes.36, 49 HMGB1 interacts with several seemingly unrelated receptors that had been previously identified for their capacity to transduce activation signals from exogenous (TLR2, TLR4, and TLR9) and endogenous (RAGE) ligands.19, 50 Our previous work8 found that HMGB1–LPS complex upregulated TLRs and RAGE in macrophages. Similarly, our present work revealed that HMGB1–LPS complex mediated the upregulation of receptors in tOASFs, whereas the TLR4 expression in RASFs was the lowest, which is not consistent with the finding in the literature. We think that the low expression of TLR4 in RASFs is due to receptor shedding. Receptor shedding is a process by which cells cleave off the extracellular domain of a cytokine receptor and release it into the circulation as a soluble product. TLR4 shedding was observed after exposure of cells to certain cytokines or after long-term culture in vitro.51 We next investigated the intracellular TLRs expression in RASFs, and the result confirmed our speculation (Supplementary Figure S3).
Adhesion molecules such as ICAM-1 and chemokines such as CCL2 are crucial for the pathogenicity of SFs.12, 28 Excessive expression of adhesion molecules by RASFs facilitates the attachment of RASFs to the adjacent cartilage and bone, leading to joint destruction, and excessive expression of chemokines by RASFs facilitates the influx of inflammatory cells. Therefore, we next investigated the expression of adhesion molecules and chemokines in tOASFs by flow cytometry. As expected, the expression of adhesion molecules ICAM-1 and VCAM-1 and chemokines CCL2 and CXCL12 was significantly upregulated in tOASFs.
Many inflammatory intercellular signal pathways, such as NF-κB, MAPK pathways, and Jak-Stat3 pathway, are involved in the activation of RASFs. The ubiquitously expressed transcription factor NF-κB is highly activated in RASFs. NF-κB containing DNA-binding heterodimers is normally retained in the cytoplasm by its natural counterpart IκB. The activation of NF-κB results in the nuclear translocation of NF-κB, enabling it to bind to the promoters of target genes, such as proinflammatory genes such as IL-6 and IL-8, adhesion molecules, and MMPs. In addition, it has been suggested that NF-κB negatively regulates the tumor-suppressor PTEN, thus promoting cell survival.10 The activation of MAPKs, especially MAPK p38, is thought to regulate processes involved in apoptosis and proliferation, and p38 can also induce the production of MMPs, IL-6, and IL-8.27 The Jak-Stat signal transduction pathway is used by many cytokines and growth factors that regulate gene expression and cellular activation, proliferation, and differentiation. The Stat3 transcription factor is an important signaling molecule for many cytokines and growth factor receptors in RASFs. Stat3 has been shown to be active in synovial lining cells in adjuvant arthritis and RA and in freshly isolated RASFs. Stat3 has been strongly implicated in promoting cell survival and growth in many cell types, and it contributes to cellular transformation.52 The underlying mechanisms described so far include conferring resistance to apoptosis and the transcriptional activation of genes important in cell cycle progression, such as cyclins. We evaluated the phosphorylation levels of key transcription factors in tOASFs using a commercial pathway scan kit. The results proved that the phosphorylation level of NF-κB p65, MAPK p38, and JNK was increased in tOASFs. Ample evidence supports the role of Stat3 in cellular transformation and oncogenesis, and the phosphorylation level of Stat3 in RASFs was higher as compared with OASFs.52 Our results showed that the phosphorylation level of Stat3 was not increased in tOASFs as expected, possibly because the Jak-Stat signal was activated by growth factors, such as epidermal growth factor and platelet-derived growth factor (PDGF). Further study should be taken to investigate why Stat3 is downregulated in tOASFs.
Once activated, RASFs produce a variety of cytokines, chemokines, and matrix-degrading enzymes that mediate interaction with neighboring inflammatory and endothelial cells and are responsible for the progressive destruction of articular cartilage and bone. Finally, we detected the expression and secretion of pathogenic factors in tOASFs by real-time RT-PCR and ELISA. The results are consistent with our expectations in that the expression and secretion of proinflammatory cytokines (TNF-α, IL-1β, and IL-6) and matrix metalloproteinases (MMPs; MMP-3, and MMP-13) were significantly enhanced in tOASFs.
Then, we used an ‘Inverse wrap’ mouse model to study the pathogenicity of in vitro-transformed OASFs (tOASFs), and the results proved that tOASFs showed excessive proliferation, followed by attachment to and degradation of co-implant cartilage.53 The in vivo model showed that in vitro-transformed OASFs had a RASF-like phenotype.
In summary, the HMGB1–LPS complex promotes the transformation of normal SFs towards a RASF-like phenotype. Our previous work8 found that HMGB1–LPS complex induced experimental arthritis in DBA/1 mice with significant synovitis, suggesting that HMGB1–LPS complex might mediate the formation of RASFs, causing the pathogenesis and leading to RA. In the current study, we proved that the transformation of OASFs was mediated by HMGB1–LPS complex both in vivo and in vitro. We therefore suggest that the outer PAMP (LPS) binds to the inner DAMP (HMGB1) to become a complex that recognizes TLRs/RAGE on SFs, leading to the activation of downstream signaling and causing the secretion of inflammatory cytokines and chemokines, the production of tissue-destructive enzymes, and the formation of RASFs, finally inducing the onset of RA. RASFs are transformed and pseudo-tumoral phenotype cells with many different characteristics as compared with normal SFs. We have only shown limited aspects of transformed tOASFs, and more work is needed to further evaluate the role of HMGB1–LPS complex in the pathogenesis of RA.
Materials and Methods
Tissues and cells
On approval of the Ethics Committee of the Second Military Medical University (Shanghai, China), RA (n=3) and osteoarthritis (n=8) synovial tissues were obtained during arthroplastic surgery, with informed consent obtained from the subjects involved and in accordance with the criteria of the American College of Rheumatology.54 OASFs (n=8) and RASFs (n=3) were isolated and cultured with DMEM (HyClone, Beijing, China) with 10% heat-inactivated FBS (HyClone) from various subjects (maximum seven passages). OASFs were cultured with 1 μg/ml HMGB1 plus 10 ng/ml LPS for five passages (media were replaced every 3 days with fresh HMGB1–LPS complex) to get the RASF-like transformed OASFs (tOASFs). Healthy human cartilages (n=5) were obtained from non-arthritic knee joints during arthroplastic surgery and were used in in vivo experiments.
Mice
Male SCID BALB/c mice aged 4–6 weeks (Model Animal Research Center of Nanjing University, Nanjing, China) were bred and housed in the animal facility at the Second Military Medical University under standard conditions of temperature and light and fed with laboratory chow and water ad libitum. The study was approved by the Ethics Committee of the Second Military Medical University.
Severe combined immunodeficient mouse model and histochemistry
The severe combined immunodeficient mouse model was prepared as described previously.48, 53 Sponge–cartilage complex with RASFs, OASFs, or tOASFs was implanted with the inverse wrap technique under the skin of the SCID mice (Figure 6a). After 60 days, the mice were killed, and implants were removed for histological evaluation of cartilage degradation. The implants were fixed in 4% formaldehyde, decalcified, embedded in paraffin, sectioned, and then stained using hematoxylin and eosin (HE) stain, Alcian blue special stain, and Masson’s trichrome stain. The stained sections of the implants were evaluated to determine fibroblast invasion and perichondrocytic cartilage degradation. The invasion scores were classified as: 0, no or minimal invasion; 1, visible invasion with 1–4 cell depth; 2, invasion with 5–9 cell depth; and 3, deep invasion with ≥10 cell depth. Perichondrocytic cartilage degradation scores (Masson’s trichrome staining) were classified as: 0, no degradation (sharp, intact halo of the chondrons compared with the cartilage stored before implantation); 1, visible degradation (one diameter of the chondron); 2, degradation (between one and two diameters of a chondron); and 3, intensive degradation (more than two diameters of a chondron). All single implants from all mice were scored by three trained researchers independently. Paraffin sections with antibodies against human proMMP-13 (R&D Systems, Minneapolis, MN, USA) were assayed by immunohistochemistry.
Flow cytometry
OASFs, tOASFs, and RASFs were cultured with DMEM containing 1% heat-inactivated FBS for 24 h, and then the media were replaced with DMEM containing 10% heat-inactivated FBS and cultured for additional 24 h. Cells were collected, fixed, and stained with PI (Keygentec, Nanjing, China), and cell cycle was analyzed using a FACSCalibur with ModFit software (Becton Dickinson, Sparks, MD, USA).
OASFs, tOASFs, and RASFs were pretreated with apoptosis-inducers A (Apopisa) and B (Apobid) (1 : 1000, Beyotime, Nantong, China) for 8 h, harvested, stained with Annexin V-FITC/PI (Bender MedSystems, Vienna, Austria), and analyzed by flow cytometry.
OASFs, tOASFs, and RASFs were stained with PE-antiTLR2 (eBioScience, San Diego, CA, USA), APC-antiTLR4 (eBioScience), and anti-RAGE (Millipore, Billerica, MA, USA). The expression of HMGB1-associcated receptors was analyzed by flow cytometry.
OASFs, tOASFs, and RASFs were stained with PerCP-antiCXCL12 (R&D), APC-antiCCL2, PE-antiVCAM-1, and APC-antiICAM-1 (eBioscience). The expression of RA-associated chemokines and intercellular adhesion molecules was analyzed by flow cytometry.
Confocal microscopy
Cells were grown on polylysine-treated coverslips in a six-well plate and cultured with DMEM containing 10% heat-inactivated FBS for 24 h to 70% confluence. The media were then replaced with PBS for 8 h to induce autophagy. The coverslips were fixed with 4% formaldehyde for 15 min and permeabilized with ice-cold 100% methanol for 10 min. The slides were then blocked with 5% goat serum for 30 min and incubated overnight with the rabbit anti-human LC3II primary antibody (Cell Signaling, Danvers, MA, USA). Following washes with PBS, PE-labeled goat anti-rabbit IgG secondary antibody (Cell Signaling) was added to the slides for 1 h. The slides were then counterstained with DAPI (Sigma-Aldrich, St. Louis, MO, USA) before observation for LC3II under a confocal microscope (LEICA TCS SP2, Am Friedensplatz, Germany).
Immunoblot analysis
Cells were lysed in a lysis buffer containing 1.0% (vol/vol) Nonidet-P40, 150 mM NaCl, 20 mM Tris-HCl, pH 7.5, 1 mM EDTA, and a protease inhibitor ‘cocktail’ (Roche, Branchburg, NJ, USA) for 5 min. Cell lysates were separated by SDS-PAGE and transferred to polyvinylidene difluoride membranes. The membranes were blotted with the appropriate antibodies (Cell Signaling) and visualized with an ECL Western Blotting System (Pierce Protein Research Products, Rockford, IL, USA).
Real-time RT-PCR
Total RNA was isolated from cells using TRIzol reagent (Invitrogen, Shanghai, China), and cDNAs were prepared and analyzed for the expression of the gene of interest by real-time PCR using a PrimeScript RT Master Mix kit (TaKaRa, Dalian, China). The primers for IL-1β, IL-6, TNF-α, ICAM, VCAM, CXCL12, MMP-3, and β-actin were purchased from Invitrogen. Transcript levels were calculated relative to controls and are expressed as −ΔΔCt. The gene-specific primers used are listed in Supplementary Table S1.
Enzyme-linked immunosorbent assay
Human TNF-α, IL-1β, IL-6, MMP-3, and MMP-13 were measured by enzyme-linked immunosorbent assay with the kits from eBioscience (TNF-α, IL-1β, and IL-6) or R&D Systems (MMP-3 and MMP-13). The phosphorylation levels of inflammatory-associated key intracellular signaling proteins in the three cell groups were assessed using a PathScan Inflammation Multi-Target Sandwich ELISA Kit (Cell Signaling). Cells were washed with ice-cold PBS, followed by addition of the 1X Cell Lysis Buffer (Cell Signaling). Then, proteins were extracted and analyzed according to the manufacturer’s instructions.
Statistics
Each experiment was repeated at least three times, and one representative result was shown. One-way ANOVA and Newman–Keuls multiple comparison test were used for the analysis with GraphPad Prism version 4.0 (GraphPad Software, La Jolla, CA, USA).
Abbreviations
- RA:
-
rheumatoid arthritis
- SF:
-
synovial fibroblast
- LPS:
-
lipopolysaccharides
- HMGB1:
-
high mobility group box 1
- OASF:
-
osteoarthritis synovial fibroblast
- RAGE:
-
receptor for advanced glycation end products
- DAMP:
-
damage-associated molecular pattern
- MAPK:
-
mitogen-activated protein kinase
- MMP:
-
matrix metalloproteinase
References
Scott DL, Wolfe F, Huizinga TW . Rheumatoid arthritis. Lancet 2010; 376: 1094–1108.
McInnes IB, Schett G . The pathogenesis of rheumatoid arthritis. N Engl J Med 2011; 365: 2205–2219.
Neumann E, Lefevre S, Zimmermann B, Gay S, Muller-Ladner U . Rheumatoid arthritis progression mediated by activated synovial fibroblasts. Trends Mol Med 2010; 16: 458–468.
Hitchon CA, El-Gabalawy HS . Infection and rheumatoid arthritis: still an open question. Curr Opin Rheumatol 2011; 23: 352–357.
Ogrendik M . Does periodontopathic bacterial infection contribute to the etiopathogenesis of the autoimmune disease rheumatoid arthritis? Discov Med 2012; 13: 349–355.
Perruche S, Saas P, Chen W . Apoptotic cell-mediated suppression of streptococcal cell wall-induced arthritis is associated with alteration of macrophage function and local regulatory T-cell increase: a potential cell-based therapy? Arthritis Res Ther 2009; 11: R104.
Marijnissen RJ, Roeleveld DM, Young D, Nickerson-Nutter C, Abdollahi-Roodsaz S, Garcia de Aquino S et al. IL-21R-deficiency increases the initial TLR2 response but protects against joint pathology by reducing Th1/17 cells during SCW-arthritis. Arthritis Rheum 2013 e-pub ahead of print 10 December 2013 doi:10.1002/art.38312.
Qin YH, Dai SM, Tang GS, Zhang J, Ren D, Wang ZW et al. HMGB1 enhances the proinflammatory activity of lipopolysaccharide by promoting the phosphorylation of MAPK p38 through receptor for advanced glycation end products. J Immunol 2009; 183: 6244–6250.
He Z-W, Qin Y-H, Wang Z-W, Chen Y, Shen Q, Dai S-M . HMGB1 acts in synergy with lipopolysaccharide in activating rheumatoid synovial fibroblasts via p38 MAPK and NF-κB signaling pathways. Mediators Inflamm 2013; 2013: 10.
Huber LC, Distler O, Tarner I, Gay RE, Gay S, Pap T . Synovial fibroblasts: key players in rheumatoid arthritis. Rheumatology (Oxford) 2006; 45: 669–675.
Matsumoto S, Muller-Ladner U, Gay RE, Nishioka K, Gay S . Ultrastructural demonstration of apoptosis, Fas and Bcl-2 expression of rheumatoid synovial fibroblasts. J Rheumatol 1996; 23: 1345–1352.
Tsou HK, Chen HT, Chang CH, Yang WY, Tang CH . Apoptosis signal-regulating kinase 1 is mediated in TNF-alpha-induced CCL2 expression in human synovial fibroblasts. J Cell Biochem 2012; 113: 3509–3519.
Niederer F, Trenkmann M, Ospelt C, Karouzakis E, Neidhart M, Stanczyk J et al. Down-regulation of microRNA-34a* in rheumatoid arthritis synovial fibroblasts promotes apoptosis resistance. Arthritis Rheum 2012; 64: 1771–1779.
Pundt N, Peters MA, Wunrau C, Strietholt S, Fehrmann C, Neugebauer K et al. Susceptibility of rheumatoid arthritis synovial fibroblasts to FasL- and TRAIL-induced apoptosis is cell cycle-dependent. Arthritis Res Ther 2009; 11: R16.
Tang D, Kang R, Livesey KM, Cheh CW, Farkas A, Loughran P et al. Endogenous HMGB1 regulates autophagy. J Cell Biol 2010; 190: 881–892.
Tang D, Kang R, Cheh CW, Livesey KM, Liang X, Schapiro NE et al. HMGB1 release and redox regulates autophagy and apoptosis in cancer cells. Oncogene 2010; 29: 5299–5310.
Shin YJ, Han SH, Kim DS, Lee GH, Yoo WH, Kang YM et al. Autophagy induction and CHOP under-expression promotes survival of fibroblasts from rheumatoid arthritis patients under endoplasmic reticulum stress. Arthritis Res Ther 2010; 12: R19.
Connor AM, Mahomed N, Gandhi R, Keystone EC, Berger SA . TNFalpha modulates protein degradation pathways in rheumatoid arthritis synovial fibroblasts. Arthritis Res Ther 2012; 14: R62.
Tian J, Avalos AM, Mao SY, Chen B, Senthil K, Wu H et al. Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE. Nat Immunol 2007; 8: 487–496.
Nadatani Y, Watanabe T, Tanigawa T, Machida H, Okazaki H, Yamagami H et al. High mobility group box 1 promotes small intestinal damage induced by nonsteroidal anti-inflammatory drugs through Toll-like receptor 4. Am J Pathol 2012; 181: 98–110.
Karouzakis E, Rengel Y, Jungel A, Kolling C, Gay RE, Michel BA et al. DNA methylation regulates the expression of CXCL12 in rheumatoid arthritis synovial fibroblasts. Genes Immun 2011; 12: 643–652.
Andreas K, Lubke C, Haupl T, Dehne T, Morawietz L, Ringe J et al. Key regulatory molecules of cartilage destruction in rheumatoid arthritis: an in vitro study. Arthritis Res Ther 2008; 10: R9.
Pavkova Goldbergova M, Lipkova J, Pavek N, Gatterova J, Vasku A, Soucek M et al. RANTES, MCP-1 chemokines and factors describing rheumatoid arthritis. Mol Immunol 2012; 52: 273–278.
Santiago B, Izquierdo E, Rueda P, Del Rey MJ, Criado G, Usategui A et al. CXCL12gamma isoform is expressed on endothelial and dendritic cells in rheumatoid arthritis synovium and regulates T cell activation. Arthritis Rheum 2012; 64: 409–417.
Morales-Ducret J, Wayner E, Elices MJ, Alvaro-Gracia JM, Zvaifler NJ, Firestein GS . Alpha 4/beta 1 integrin (VLA-4) ligands in arthritis. Vascular cell adhesion molecule-1 expression in synovium and on fibroblast-like synoviocytes. J Immunol 1992; 149: 1424–1431.
Johnson BA, Haines GK, Harlow LA, Koch AE . Adhesion molecule expression in human synovial tissue. Arthritis Rheum 1993; 36: 137–146.
Luo SF, Fang RY, Hsieh HL, Chi PL, Lin CC, Hsiao LD et al. Involvement of MAPKs and NF-kappaB in tumor necrosis factor alpha-induced vascular cell adhesion molecule 1 expression in human rheumatoid arthritis synovial fibroblasts. Arthritis Rheum 2010; 62: 105–116.
Yang CM, Luo SF, Hsieh HL, Chi PL, Lin CC, Wu CC et al. Interleukin-1beta induces ICAM-1 expression enhancing leukocyte adhesion in human rheumatoid arthritis synovial fibroblasts: involvement of ERK, JNK, AP-1, and NF-kappaB. J Cell Physiol 2010; 224: 516–526.
Okamoto H, Cujec TP, Yamanaka H, Kamatani N . Molecular aspects of rheumatoid arthritis: role of transcription factors. FEBS J 2008; 275: 4463–4470.
Bondeson J, Foxwell B, Brennan F, Feldmann M . Defining therapeutic targets by using adenovirus: blocking NF-kappaB inhibits both inflammatory and destructive mechanisms in rheumatoid synovium but spares anti-inflammatory mediators. Proc Natl Acad Sci USA 1999; 96: 5668–5673.
Kew RR, Penzo M, Habiel DM, Marcu KB . The IKKalpha-dependent NF-kappaB p52/RelB noncanonical pathway is essential to sustain a CXCL12 autocrine loop in cells migrating in response to HMGB1. J Immunol 2012; 188: 2380–2386.
Seemayer CA, Kuchen S, Kuenzler P, Rihoskova V, Rethage J, Aicher WK et al. Cartilage destruction mediated by synovial fibroblasts does not depend on proliferation in rheumatoid arthritis. Am J Pathol 2003; 162: 1549–1557.
Smerdon MJ, Isenberg I . Interactions between the subfractons of calf thymus H1 and nonhistone chromosomal proteins HMG1 and HMG2. Biochemistry 1976; 15: 4242–4247.
Bustin M, Hopkins RB, Isenberg I . Immunological relatedness of high mobility group chromosomal proteins from calf thymus. J Biol Chem 1978; 253: 1694–1699.
Andersson U, Tracey KJ . HMGB1 is a therapeutic target for sterile inflammation and infection. Annu Rev Immunol 2011; 29: 139–162.
Sims GP, Rowe DC, Rietdijk ST, Herbst R, Coyle AJ . HMGB1 and RAGE in inflammation and cancer. Annu Rev Immunol 2010; 28: 367–388.
Ding HS, Yang J, Gong FL, Ding JW, Li S, Jiang YR . High mobility box 1 mediates neutrophil recruitment in myocardial ischemia-reperfusion injury through toll like receptor 4-related pathway. Gene 2012; 509: 149–153.
Li L, Chen L, Hu L, Liu Y, Sun HY, Tang J et al. Nuclear factor high-mobility group box1 mediating the activation of Toll-like receptor 4 signaling in hepatocytes in the early stage of nonalcoholic fatty liver disease in mice. Hepatology 2011; 54: 1620–1630.
Wang X, Sun R, Wei H, Tian Z . HMGB1-TLR4-IL-23-IL-17A axis in drug-induced damage-associated lethal hepatitis: Interaction of gammadelta T cells with macrophages. Hepatology 2012; 57: 373–384.
Kokkola R, Sundberg E, Ulfgren AK, Palmblad K, Li J, Wang H et al. High mobility group box chromosomal protein 1: a novel proinflammatory mediator in synovitis. Arthritis Rheum 2002; 46: 2598–2603.
Andersson U, Harris HE . The role of HMGB1 in the pathogenesis of rheumatic disease. Biochim Biophys Acta 2010; 1799: 141–148.
Gong Q, Zhang H, Li JH, Duan LH, Zhong S, Kong XL et al. High-mobility group box 1 exacerbates concanavalin A-induced hepatic injury in mice. J Mol Med (Berl) 2010; 88: 1289–1298.
Apetoh L, Ghiringhelli F, Tesniere A, Criollo A, Ortiz C, Lidereau R et al. The interaction between HMGB1 and TLR4 dictates the outcome of anticancer chemotherapy and radiotherapy. Immunol Rev 2007; 220: 47–59.
Mills KH . TLR-dependent T cell activation in autoimmunity. Nat Rev Immunol 2011; 11: 807–822.
Ciucci A, Gabriele I, Percario ZA, Affabris E, Colizzi V, Mancino G . HMGB1 and cord blood: its role as immuno-adjuvant factor in innate immunity. PLoS One 2011; 6: e23766.
Steer SA, Scarim AL, Chambers KT, Corbett JA . Interleukin-1 stimulates beta-cell necrosis and release of the immunological adjuvant HMGB1. PLoS Med 2006; 3: e17.
Fagone P, Shedlock DJ, Bao H, Kawalekar OU, Yan J, Gupta D et al. Molecular adjuvant HMGB1 enhances anti-influenza immunity during DNA vaccination. Gene Ther 2011; 18: 1070–1077.
Lefevre S, Knedla A, Tennie C, Kampmann A, Wunrau C, Dinser R et al. Synovial fibroblasts spread rheumatoid arthritis to unaffected joints. Nat Med 2009; 15: 1414–1420.
Brentano F, Kyburz D, Schorr O, Gay R, Gay S . The role of Toll-like receptor signalling in the pathogenesis of arthritis. Cell Immunol 2005; 233: 90–96.
Apetoh L, Ghiringhelli F, Tesniere A, Obeid M, Ortiz C, Criollo A et al. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med 2007; 13: 1050–1059.
Zager RA, Johnson AC, Lund S, Randolph-Habecker J . Toll-like receptor (TLR4) shedding and depletion: acute proximal tubular cell responses to hypoxic and toxic injury. Am J Physiol Renal Physiol 2007; 292: F304–F312.
Krause A, Scaletta N, Ji JD, Ivashkiv LB . Rheumatoid arthritis synoviocyte survival is dependent on Stat3. J Immunol 2002; 169: 6610–6616.
Judex M, Neumann E, Fleck M, Pap T, Mountz JD, Gay RE et al. "Inverse wrap": an improved implantation technique for virus-transduced synovial fibroblasts in the SCID mouse model for rheumatoid arthritis. Mod Rheumatol 2001; 11: 145–150.
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988; 31: 315–324.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (Nos. 31000395, 30972729). We thank Professor Minghua Zhu (Department of Pathology, Changhai Hospital, Shanghai, China) for his kind help in the pathological evaluation of the experimental arthritis model and Professor Shibo Jiang for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by A Stephanou
Supplementary Information accompanies this paper on Cell Death and Disease website
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Qin, Y., Chen, Y., Wang, W. et al. HMGB1–LPS complex promotes transformation of osteoarthritis synovial fibroblasts to a rheumatoid arthritis synovial fibroblast-like phenotype. Cell Death Dis 5, e1077 (2014). https://doi.org/10.1038/cddis.2014.48
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cddis.2014.48
Keywords
This article is cited by
-
The role of the Beclin-1: a gene related to autophagy in rheumatoid arthritis
Egyptian Rheumatology and Rehabilitation (2023)
-
Polyene phosphatidylcholine ameliorates synovial inflammation: involvement of PTEN elevation and glycolysis suppression
Molecular Biology Reports (2023)
-
Anti-HMGB1 antibody is a potential characteristic autoantibody for Sjögren's syndrome
Scientific Reports (2022)
-
HMGB1, anti-HMGB1 antibodies, and ratio of HMGB1/anti-HMGB1 antibodies as diagnosis indicator in fever of unknown origin
Scientific Reports (2021)
-
Blockade of HMGB1 signaling pathway by ethyl pyruvate inhibits tumor growth in diffuse large B-cell lymphoma
Cell Death & Disease (2019)