Abstract
Circadian oscillation and cell cycle progression are the two most essential rhythmic events present in almost all organisms. Circadian rhythms keep track of time and provide temporal regulation with a period of about 24 h. The cell cycle is optimized for growth and division, but not for time keeping. Circadian gated cell divisions are observed in nearly all organisms. However, the implications of this coupling to the physiology of mammals are unknown. A mutation (S662G) in the clock protein PERIOD2 (PER2) is responsible for familial advanced sleep phase syndrome in which sleep onset occurs in the early evening and wakefulness occurs in the early morning. Here, we provide evidence that the PER2S662 mutation leads to enhanced resistance to X-ray-induced apoptosis and increased E1A- and RAS-mediated oncogenic transformation. Accordingly, the PER2S662 mutation affects tumorigenesis in cancer-sensitized p53R172H/+ mice. Finally, analyzing the clock-controlled cell cycle genes p21, c-Myc, Cyclin D1 and p27, we found that the relative phases between p21 and Cyclin D expression profiles have been changed significantly in these Per2 allele mutant mouse embryonic fibroblasts. This key role of the Per2-mediated phase alteration of p21 provides what we believe to be a novel mechanism in understanding cell cycle progression, its plasticity and its resistance to interference.
Similar content being viewed by others
Main
The circadian clock is essential for the maintenance of physiological homeostasis, as well as the coordination of responses to external signals. In mammals, the circadian clock is composed of interlocking positive and negative feedback loops. In the primary feedback loop, three basic helix–loop–helix (bHLH)/PAS domain-containing transcription factors, Clock, Npas2 and Bmal1, activate the transcription of two period genes (Per1 and Per2) and two cryptochrome (Cry) genes (Cry1 and Cry2) by binding to the E-box cis elements in the promoters of these genes. Subsequently, the PER/CRY complex inhibits the transcription of its own genes by blocking BMAL1/CLOCK activity. The second feedback loop is mediated by the orphan nuclear receptors RORα and Rev-erbα. Transcription of RORα and Rev-erba is activated during the day by the CLOCK/BMAL1 complex, after which they exert positive and negative transcriptional effects on the Bmal1 gene through Rev-erbα/RORα responsive elements (ROREs) in the Bmal1 promoter.1, 2, 3 The circadian system may be tightly connected to many physiological processes through clock-controlled nuclear receptor genes (e.g., PPARα and PPARγ) and cell cycle genes (e.g., c-Myc, cyclin D, Wee1 and p21).5, 6, 7, 8 The links between the circadian clock and a variety of physiological processes are just beginning to be unveiled; however, the underlying mechanisms remain unclear.
A major consequence of a modern lifestyle is the disruption of circadian rhythms. Importantly, epidemiological studies have shown that a disruption in circadian rhythms increases an individual's risk of developing colon, breast, prostate, ovarian, lung and liver cancer.9, 10 The International Agency for Research on Cancer has elevated the carcinogenicity of circadian disruption from Group 2B to Group 2A.11 Furthermore, several studies using mouse models have established convincing links between mutations in certain clock genes and tumorigenesis. Targeted disruption of PERIOD2 (Per2) leads to tumor susceptibility in the presence of γ radiation.6 Per1 also functions as a tumor suppressor by regulating cell cycle genes and by interacting with key checkpoint proteins that are activated by DNA damage.8 Given the strong links between circadian rhythms and the diverse physiological processes in these animal models, it is important to address whether human circadian variations influence cancer susceptibility.
Advanced sleep phase syndrome (ASPS) is a circadian rhythm variant in which sleep onset occurs in the early evening (i.e., 1800–2000 hours) and, as a consequence, wakefulness occurs in the early morning (i.e., 0100–0300 h).12, 13, 14 The discovery that the PER2S662G mutation causes familial ASPS (FASPS) was the first direct evidence that PER2 has an essential role in the human circadian clock.13, 15 Furthermore, given that this specific amino acid of the PER2 protein may represent an appealing therapeutic target, we investigated whether the S662 site is also crucial for its function as a tumor suppressor. Moreover, PER2S662G and PER2S662D transgenic mice are unique because single amino-acid changes at this site can either shorten or lengthen the circadian period in a mutation-dependent manner, thus providing a unique model to understand how circadian period length affects cell cycle progression and tumorigenesis.
In the present study, we focused on characterizing the link between PER2S662 mutations and tumorigenesis both in vitro and in vivo. We found that mutation of PER2S662 affects tumorigenesis in p53R172H/+ mice, which have a cancer-sensitized genetic background caused by a p53 knock-in mutation2H.16 We subsequently found that PER2 acts as a phase regulator, directly or indirectly, to affect the advanced or delayed cell cycle gene expression profiles, but not amplitude. These findings raise the possibility that alteration of p21 phase by Per2 allele mutant may interfere with cell phase locking to promote cell cycle progression.
Results
S662 is a key amino acid in PER2 that modulates X-ray-induced apoptosis
We first investigated whether S662 in PER2 is crucial for its function as a tumor suppressor by analyzing the apoptotic response. To avoid potential interference from the wild-type Per2 gene, we crossed both PER2S662G and PER2S662D (amino acid 662 changed from serine to aspartate) bacterial artificial chromosome (BAC) transgenic mice to a Per2−/− background.15, 17 The resultant compound mice, aged 8–10 weeks, were treated with 6-Gy X-ray irradiation at Zeitgeber Time (ZT) 0 and ZT 9. The percentage of apoptotic spleen cells was reduced significantly (approximately two fold) in Per2−/− spleens compared with those of wild-type littermates at ZT 0 and ZT 9, as shown by flow cytometry (Figures 1a and b). Interestingly, the cells from PER2S662G:Per2−/− mice displayed markedly attenuated levels of apoptosis that were similar to those of Per2−/− mice, and PER2S662D:Per2−/− mice were even more resistant to X-ray-induced apoptosis than Per2 knockout (KO) mice (Figure 1b).
The PER2S662 mutation leads to decreased levels of apoptosis. (a) Wild-type, Per2−/−, PER2S662G:Per2−/− and PER2S662D:Per2−/− mice were irradiated with 6 Gy X-rays at ZT 0 (0800 hours) and ZT 9 (1700 hours). Spleen cells were isolated 6 h after irradiation, stained with propidium iodide (PI) and analyzed by flow cytometry. One representative experiment is shown. Ap, apoptotic percentage. (b) Percentages of apoptotic cells measured by flow cytometry are shown in the histograms. Experiments were repeated at least three times with consistent results and at least two mice were used per group for each time (N=3 for each genotype; ***P<0.001; one-way ANOVA followed by Dunnett's test)
Generation of PER2 dose-dependent BAC transgenic mice
As there is a higher level of PER2 in PER2S662D mice compared with wild-type littermates,15 it is plausible that PER2 may affect apoptosis through multiple mechanisms. To determine whether increased expression of PER2 compared with wild-type levels is also responsible for the observed resistance to apoptosis, we generated PER2 dose-dependent BAC transgenic mice (Figure 2a). These transgenic mice were designed to express cis-acting genomic regulatory elements in a manner that faithfully recapitulates endogenous PER2 expression. We characterized these mice based on their copy number by Southern blot (Figure 2b) and quantitative-PCR (Q-PCR) (Figure 2c), and then we selected three transgenic lines as low (1–2 copies) (L), middle (2–3 copies) (M) and high copy (4–5 copies) (H). mRNA levels are much higher in the H line than in the PER2S662D2H in including liver (Figure 2d), spleen (Figure 2e) and mouse embryonic fibroblasts (MEFs) (Figure 2f), and PER2 protein in H line is more abundant than those from the PER2S662D mice (Figure 2g). These mice have a circadian period that lengthens as the copy number of Per2 increases (Figure 2h). Mice with four to five copies of the PER2 transgene showed an extreme elongation of the circadian period to 25 h, which is longer than that of the mice with the PER2S662D mutation (24 h).15 The PER2 dose dependence of period length suggests that PER2 has a critical role in circadian clock, which is also seen in dorosphila.18, 19 PER2S662D may elongate the circadian period through the increased PER2 protein level. We noticed that apoptosis rates also decreased in the high-copy mice (28%±0.9), but this decrease is less than that in the PER2S662G:Per2−/− mutant (16.5%±0.3), PER2S662D:Per2−/− mutant (9.9%±0.05) and Per2−/− mice (15.08%±0.2) (P<0.001) (Figures 3a and b). This suggests that PER2 homeostasis has a role in maintaining a proper apoptosis response. Furthermore, we crossed L-line mice to the Per2−/− background because all results are on a Per2−/− background. The cells from L-line mice displayed comparable levels of apoptosis to those from L line on a Per2−/− background (35.1±0.1 versus 35.2±0.5), to exclude a remote possibility that Per2–/– acts as a dominant negative role. These genetic observations suggest that both the mutational status of PER2S662 and the levels of PER2 protein may affect the apoptotic response to X-rays.
Generation and characterization of PER2 BAC transgenic mice. (a) Generation of different copy BAC transgenic mice. (b) Transgene copy number was estimated by Southern blot by comparing with copy standards. Experiments were repeated two times with consistent results and one mouse was used per line. (c) The relative transgene copy number was further estimated by q-PCR. (d–f) The mRNA expression level was assessed by qRT-PCR in liver tissues (d), spleen tissues (e) and non-synchronized MEFs (f) of different BAC transgenic mice as indicated genotypes. Experiments were repeated independently at least three times with consistent results and at least two replicates were used per time point. ‘→’ denotes the band of PER2 and ‘*’ denotes a unspecific band. (g) PER2 protein levels in spleen tissues of H line and PER2S662D. Experiments were repeated three times with consistent results, and one mouse for each time point was used for western blot. (h) Voluntary locomotor activities were recorded as wheel-running activity. BAC transgenic mice were subjected first to LD (light and dark cycle) and then released into DD (dark and dark cycle). Representative actograms from low- (23.64±0.11, n=6), middle- (24.34±0.18, n=7) and high-copy BAC transgenic mice (25.07±0.17, n=9). Alternating white and dark bars indicate the LD cycles during entrainment before release in DD
High-dose PER2 results in resistance to apoptosis induced by X-rays. (a) Low-, middle- and high-copy PER2 mice were irradiated with 6 Gy X-rays at ZT 9. Spleen cells were isolated 6 h after irradiation and the DNA content was analyzed by flow cytometry. One representative experiment is shown. (b) Percentages of apoptotic cells detected by flow cytometry from low-, middle- and high-copy BAC transgenic spleens (***P<0.001; one-way ANOVA followed by Dunnett's test)
Mutation of S662 promotes oncogenic E1A–RAS-induced transformation in MEFs
A previous study has shown that primary embryo fibroblasts can be immortalized by the expression of at least two oncogenes; for example, E1A, myc and ras.20 This technique has been used to evaluate whether inactivation or deletion of a tumor suppressor gene can hasten the activation of oncogenes and promote oncogenic transformation.21 To determine whether the PER2S662 mutation results in a transformation-permissive phenotype in vitro, PER2S662G, PER2S662D, wild-type and Per2−/− MEFs were infected with a retroviral vector that co-expressed the oncogenes E1a and Ras. The ability of E1a and Ras to induce cellular transformation was far greater in PER2S662G mice than in wild-type MEFs, while the level of transformation was comparable to that of Per2 KO MEFs (Figures 4a and b). Furthermore, consistent with the phenotype shown for X-ray-induced apoptosis, PER2S662D MEFs formed more colonies than PER2S662G and Per2−/− MEFs (Figures 4a and b). Finally, we assessed whether overexpression of wild-type PER2 could trigger cellular transformation. As expected, the MEFs with a high-copy number of Per2 from H line grew more colonies than those with a low-copy number of Per2 or wild-type MEFs, but they grew fewer colonies than PER2S662D mutant MEFs (Figures 4c and d). The numbers for E1a- and Ras-induced transformation foci are 310±107 for PER2S662D, 205±52 for H line, 158±25 for Per2−/−, 150±36 for PER2S662G, 96±24 for L line, 74±35 for wild type and 8±5 for no-transfection control. These mice are on a wild-type background because of technical difficulty getting all embryos from seven genotype mice on a Per2−/− background at one time. These findings are consistent with the apoptotic responses observed after X-ray exposure. Furthermore, similar transformation rates between PER2S662G on wild-type background and Per2−/−MEFs suggest that PER2S662G is not just a loss of function mutation but has a dominant role. Altogether, these results illustrate the importance of the S662 site and of maintaining an appropriate level of PER2 to mediate the cellular response to environmental mutagens.
Dysregulation of PER2 increases formation of oncogenic E1A–RAS-mediated foci in MEFs. (a) and (c) Passage 0 MEFs with the indicated genotypes were infected with retroviruses encoding E1A–RAS. Transfections with empty virus vector were used as negative controls. Two representative culture plates are shown per genotype from at least two repeated independent experiments. (b) and (d) The numbers of foci per 60-mm plate in the assays are expressed as means±S.D. (N=6 for each group; one-way ANOVA followed by Dunnett's test)
Mutations in PER2S662 accelerate tumorigenesis in p53R172H/+ background mice
The influence of PER2S662 mutations on both X-ray-induced apoptosis and in vitro transformation led us to assess the relevance of the PER2S662 mutation with regard to tumorigenesis in vivo. It has been noted that a mutation in one or more core clock genes is not sufficient to elicit increased tumor incidence in the absence of a cancer-sensitized genetic background.22
As the PER2S662 mutation is found in a human circadian variant, we mated PER2S662 mutant mice with p53R172H knock-in mice. These knock-in mice have a mutation that corresponds to the p53R175H hot spot in human cancers.16 Through breeding, we established cohorts of p53R172H/+ (n=54), PER2S662G2H:p53R172H/+ (n=41), PER2S662D2H:p53R172H/+ (n=31) and ClockΔ19/+:p53R172H/+ (n=29) compound mutant mice. These mice all had the same genetic background of C57BL/6. We monitored these mice for tumor development and survival for up to 700 days. As shown in Figure 5a, Kaplan–Meier overall survival curves of these compound mutant mice showed slight but significant differences (log-rank test, P=0.049). A closer inspection of the data revealed that the survival of female p53R172H/+ and ClockΔ19/+:p53R172H/+ mice was decreased markedly compared with that of male mice (Supplementary Figures S1A and B). But the survival of PER2S662G2H:p53R172H/+ and PER2S662D2H:p53R172H/+ mice did not show any gender-based differences (Supplementary Figures S1C and 1D). Thus, to further dissociate the effects of the PER2S662 mutation on tumor development, we assessed male and female survival curves separately. The survival curve for males was significantly affected by circadian gene mutations (Figure 5b, log-rank test, P<0.001). The median life spans were also reduced for male PER2S662D2H:p53R172H/+ (15.0 months) and PER2S662G2H:p53R172H/+ (16.6 months) mice compared with those of male p53R172H/+ (17.9 months) and ClockΔ19/+:p53R172H/+ (19.8 months) mice (Figure 5b). As tumors in p53R172H/+ mice arise after a similar length of latency, these differences in the survival curves and median life spans reflected changes that contributed to tumorigenesis.
PER2S662 mutations increased tumor incidence in the p53R172H/+ background. (a) Overall survival. (b) Male survival. Survival curves (Kaplan–Meier representation) of the indicated mouse cohorts. n=number of mice per genotype. The survival curves are plotted with respect to the number of weeks using SPSS software. Statistical significance was assessed using the log-rank test and is indicated (P-value). Alterations in median life span of the indicated genotypes relative to control (increase as plus or decrease as minus) are labeled
This raises the question of why female and male on a p53R172H/+ background have different survival rates, and why we did not observe a difference between PER2S6622H:p53R172H/+2 and p53R172H/+ female mice, in contrast to the male mice. Of note, we did not obtain any p53R172H/R172H female homozygous mice from 75 pups (Supplementary Table 1), and we found that they all died by 13.5 days as a result of fatal embryonic exencephaly (Figure 6). These findings are consistent with a previous reporting that lack of the tumor suppressor protein p53 causes a female-specific disruption of neural tube closure associated with embryonic or neonatal death.23 This suggests that female p53R172H/+ mice may have a penetrant phenotype that could have masked the effects of PER2S662 mutations on female tumorigenesis. These data demonstrate collectively that the PER2S662 mutation has an impact on tumor development in the p53R172H/+ mutant background.
Female-specific neural tube defects in p53R172H/R172H mice. (a) p53R172H/R172H female embryos develop exencephaly, which results from a failure of neural tube defects. This is a represent from 13.5-day mouse embryo from p53R172H/+ and p53R172H/+ crosses. Two p53R172H/R172H females all develop exencephaly (arrow). (b) HE staining for female embryo neural tubes with exencephaly (arrow)
Circadian cycle length is not tightly associated with tumorigenesis
Then, we tested whether the accelerated tumorigenesis caused by PER2S662 mutation was related to the alteration in circadian period. As shown in Supplementary Figures 2A and B, the p53R172H/+ mutation did not affect the circadian period or the expression of core clock genes. Furthermore, in the PER2S662G2H:p53R172H/+, PER2S662D2H:p53R172H/+ and ClockΔ19/+:p53R172H/+ mice, circadian period was not closely related to tumor development (Figure 5a and Supplementary Figure S2C).
The most common tumor types observed in mice of the above genotypes are lymphomas and sarcomas, consistent with the reported tumor ratio of p53R172H/+ mice (Supplementary Figure S3).16 It is noteworthy that the incidence of primary hepatic malignancy and metastatic tumors was increased in PER2S662G2H:p53R172H/+ (27.3%) and PER2S662D2H:p53R172H/+ mice (19.4%) compared with that in p53R172H/+ mice (8.7%), although it did not reach statistical significance (P=0.17) (Table 1). We observed that circadian cycle length was also not associated with certain types of tumors (Table 1). These results support the notion that mutation of PER2 at S662 promotes tumorigenesis, independent of the acceleration or deceleration of the circadian clock.
PER2S662 affects changes in the relative phases of clock-controlled cell cycle gene
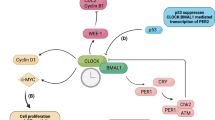
To understand the mechanism underlying tumorigenesis, the clock-related cell cycle genes p21, c-Myc, Cyclin D1 and p27 were assayed in Per2 allele mutant MEFs. Unexpectedly, in several independent experiments, the expression levels of these circadian-related cell cycle regulators are not significantly different in Per2 allele mutant MEFs compared with the wild-type (Figure 7 and Supplementary Figure S4). We noticed that p21 transcripts show peak levels when Cyclin D attains trough levels in the wild-type MEFs. However, the relative phases of p21 and Cyclin D appear to be highly similar in these Per2 allele mutant MEFs in contrast to the wild-type MEFs. It is important to note that p21 transcript phases are delayed in Per2 allele mutant MEFs, which may bring changes in the relative phases between p21 and Cyclin D (Figure 7).
The relative phases of cell cycle genes are shifted by PER2 mutations. Wild-type and different PER2 mutant MEFs were synchronized by a dexamethasone treatment, and RNA was extracted at 3 h intervals stating 10 h after the shock. p21, Cyclin D1 and Nr1d1 expression analysis were done by q-PCR. Experiments were repeated independently at least three times with consistent results. Plotted values are the mean values±S.D. Cosinor analysis was performed on mRNA profiles using SPSS 17.0. The amplitude and acrophase obtained from cosinor analysis were further analyzed by Students t-test. The null hypothesis was rejected at P<0.05
For the p21 gene, it has been described previously that its regulatory region contains a RORE binding site and is regulated by the antagonistic activities of the orphan nuclear receptors NR1D1/2 and NR1F1.24 The question then arises as to whether NR1D1 mediates the p21 phase shifting. Therefore, we examined the transcription profiles of Nr1d1 to compare the results obtained from p21 and Cyclin D. We found the accumulation of Nr1d11 cycles with an antiphase to that of p21 transcription in the wild-type MEFs, indicating that NR1D1 inhibits the transcription of p21 expression as described previously.24 While in these mutant MEFs, p21 expression profiles are uncoupled at least in part from Nr1d1 expression (Figure 7), thereby offering the possibility that NR1D1 needs wild-type PER2 to regulate the phase of p21 expression. In addition, as shown in Figure 7, the cycles of Nr1d1 transcripts respond to intrinsic circadian period in these mutant MEFs, while the periods of p21 and Cyclin D seem to be fixed 24 h in these mutant MEFs no matter whether they own short or long circadian period. Moreover, the phases of p21 transcripts delay to the same extend in Per2 allele mutant MEFs, suggesting that the ability of the circadian control of p21 expression has been abolished when PER2 is mutated, which is mediated mainly through NR1D1. This assumption is supported by a recent study by Isabelle Schmutz et al.5 demonstrating that PER2 coordinates circadian output by interacting with NR1D1.
The p21 protein inhibits the activity of cyclin–CDK2 or –CDK4 complexes, and functions as a regulator of cell cycle progression at G1.25 Although the p21 expression levels in these PER2 mutant MEFs remains comparable to that of wild-type MEFs, the peak of p21 is locked to the abnormal region that overlaps with the peak of the cyclin D phase. It is conceivable that the p21/cyclin D interval variations may trigger cell cycle progression.
Discussion
The physiological relevance of the PER2S662 mutation to human cancer
In this study, we provided evidence that the human PER2S662G mutation, which causes the FASPS circadian variant, is also responsible for increased cancer risk. To our knowledge, this represents the first instance of experimental evidence that links a human circadian mutation to cancer risk. Although the cancer incidence data in FASPS individuals are unavailable owing to scarcity of sampling (one family), a missense mutation in the evolutionarily conserved residue of hPER2 (L823V) has been identified in some human breast cancer samples26 and altered PER2 expression is common in human breast malignancies.27, 28 By comparing the X-ray-induced apoptosis level and transformation rate among Per2−/−, PER2S662G, PER2S662D and PER2 dose-dependent BAC transgenic mice, we suggested that PER2S662G is a dominant negative mutation and PER2S662D gains extra function to prevent apoptosis and increase transformation rate. Interestingly, the functional alternations of PER2S662G and PER2S662D displayed in the aspect of cell cycle are different from that in circadian clock, because PER2S662G mice show shorter circadian period length than Per2−/− mice and PER2S662D might elongate the circadian period through increasing PER2 protein level. Furthermore, our data that overexpressing and knockout PER2 both trigger more transformation foci and affect normal apoptosis function suggest that maintaining an optimal level of PER2 is critical to reduce the risk of tumorigenesis. Our functional studies of human variants of PER2, together with unbiased analyses in human cancer by other groups, indicate the importance of PER2 in human tumorigenesis. But the hypothesis that the FASPS mutation increases cancer risk in humans remains to be tested in future.
Impaired gene expression or impaired circadian rhythms?
Previous work has demonstrated that the PER2S662G and PER2S662D mutations give rise to completely contrary circadian phenotypes in terms of circadian period, phase and amplitude;15 thus, we predicted that the PER2S662G and PER2S662D mutant mice should have contrary cancer risks if impaired rhythm is the culprit. However, the enhanced cancer risk in both PER2S662G2H:p53R172H/+ and PER2S662D2H:p53R172H/+ mice strongly suggested that the impaired PER2 gene, not the impaired circadian rhythm, is responsible for the increased cancer risk in these mice. This observation is further supported by survival data from the PER2S662D2H:p53R172H/+ and ClockΔ19/+:p53R172H/+ mice, both of which exhibited lengthened circadian periods, but showed highly contrary cancer risks. Interestingly, the ‘impaired gene’ theory is consistent with the observation from cancer risks in Cry1−/−:Cry2−/− mice22, 29 and ClockΔ19 mutant mice.30 In addition, we wish to point out that our experiments share an important similarity with the above-mentioned investigations; all of these experimental systems retain the circadian mutant mice on normal light/dark cycles and subject these mice to entrainment from external time cues. Accordingly, the ‘impaired gene’ theory may only apply to circadian mutant mice kept in normal light/dark cycles. Circadian disruption is not as intense or detrimental in normal cycles as the disruption experienced by shift workers. In this regard, an important future direction may focus on both the respective contribution and the composite consequence of disrupted circadian genes, as well as disrupted circadian rhythm to cancer risk in persistent phase-shifted schedules.
The cell cycle, circadian clock and relative phase
Recent studies have shown that the circadian clock is coupled to the cell cycle processes through clock-controlled genes, which may contain E-box or ROR elements in their promoters. Somewhat surprisingly, although we found that deregulation of PER2 has a strong effect on the induced transformation of MEFs and tumorigenesis; the gene expression changes in these clock-controlled genes are less profound in Per2 allele mutant MEFs. This leads us to think about their phase implication. Although the Nr1d1 transcripts displayed obvious phase shift and period length changes in these mutant MEFS, p21 transcripts displayed fixed 24-h-period oscillation with phase delay to the same extent, leading to a similar phase to that of Cyclin D. It indicates that in the wide-type MEFs, the opposite phases of p21 and Cyclin D are a result of coordination between circadian clock and cell cycle, with circadian clock adjusting transcription phase of p21 and the main driver of cell cycle. With the interaction of PER2, NR1D1 may be a critical circadian factor in exerting the phase adjusting function. At present, the physiological significance of the altered phases of these genes is unclear, and the phase shifting cannot explain all the phenotypes that are seen in these mutant mice. It is likely that modifier relative phases between the cycle genes influence features such as the number of transformation foci and the development of the tumor-associated fibroblasts. One of the possibilities comes from interfering with phase locking. Oscillation-based synchrony is the most energy-efficient physical mechanism for temporal coordination. It occurs in many biological systems, including two oscillators of cell division and the circadian phase31 or within a cell cycle oscillator through multiple regulatory interactions.32 It is conceivable that, if phase-coupled p21 appears at a later phase of the inhibitory cycle while Cyclin D remains unchanged, the corresponding inhibition of the CDK complex becomes weak. In other words, if the threshold is reached, CDK activation can initialize unexpected cell cycle progression. It would be interesting, therefore, to investigate the extent to which relative phase changes might affect cell cycle progression, transformation and tumorigenesis.
Materials and Methods
Animals
Mice were observed daily and killed when they showed signs of morbidity or tumor growth in accordance to the Guidelines for Human Endpoints for Animals Used in Biomedical Research. Mice were housed in an Assessment And Accreditation Of Laboratory Animal Care credited SPF animal facility. PER2S662G5H:p53R175H/+, PER2S662D5H: p53R175H/+, ClockΔ19/+:p53R175H/+ mice and their littermates were derived from PER2S662 mutants and ClockΔ19 mutants33 by crossing with p53R175H/+ mice. All mutant animals have been backcrossed for more than 10 generations onto a C57BL/6 inbred background. Low-, middle- and high-copy PER2 BAC transgenic mice were generated, as described previously.15 Locomotor activity and activity pattern analyses were performed, as described previously.34
X-ray and flow cytometer
Mice were radiated at the indicated ZT times with a single dose of 6 Gy (100 cGy/min) with a XHA600C Medical Electron Linear Accelerator (Shinva Medical Instrument Co., Ltd, Zibo, Shandong, China). Splenic lymphocytes were isolated at 6 h after irradiation, and red cells were removed using Gey's solution. For flow cytometer analyses, splenic lymphocytes were centrifuged at 1000 r.p.m., and the supernatants were thoroughly removed by aspiration and fixed in 70% ethanol. After incubation with 40 μg/ml propidium iodide and 100 μg/ml RNase at 37 °C for 30 min, the samples were analyzed with a Becton Dickinson FACScan flow cytometer using CellQuest software (Becton Dickinson, Auckland, New Zealand).
Retroviral transfection and focus formation assays
The Plate-E virus packaging cells were transfected with the retroviral vector pMXs carrying the cDNA encoding E1A and Ras or pMXs with GFP as the control using PEI as the transfection reagent.35 Culture media collected 2 days after transfection were used for infection. MEFs were isolated from 13.5-day embryos as described. P1 MEFs were incubated with fresh virus for 6 h. One day after infection, the medium was replaced with fresh DMEM supplemented with 5% fetal bovine serum and purimycin, and the cells were selected after another 48 h. The infection efficiency was >95%, as estimated by a control experiment. After selection, the cells were plated at 1 × 106 cells/6 cm2 for 14 days. The medium was changed every 2 days. Confluent monolayer cultures with foci were rinsed with phosphate buffered saline (PBS), fixed in 10% formalin and stained with 0.1% crystal violet for 20 min; subsequently, the colonies were scored.
Tumorigenesis
Cohorts of mice were monitored carefully for signs of morbidity or obvious tumor burden. Mice with visible neoplasms around 2 cm in diameter were killed and necropsied. Mice with ill health but no visible neoplasms were monitored carefully for signs of morbidity. Moribund mice or mice that died without signs of morbidity were also necropsied. In most cases, tumor-burdened mice could be identified through necropsy and all tissues with suspected gross lesions or neoplasms were further examined by histopathological examination. Samples were fixed in neutral buffered formalin. Tissues with osseous structure were decalcified with 10% hydrochloric acid. Paraffin sections were prepared and stained with hematoxylin and eosin following standard procedures. Tumor spectrum analysis was performed blindly with the assistance of a clinical pathologist.
Q-PCR and reverse transcription-PCR (RT-PCR)
Primers are listed in Supplementary Table 2. All procedures were carried out as previously described.34
Western blotting
Tissue proteins were prepared as described previously using a nuclear extraction kit at the indicated circadian time (Active Motif, Carlsbad, CA, USA). Rabbit anticyclin E and antiPPARα (Santa Cruz Biotechnology, Santa Cruz, CA, USA) were utilized for western blotting according to the manufacturer's suggested protocol.
Statistical analysis
Groups of data are presented as mean±S.D. One-way analysis of variance (ANOVA) followed by Dunnett's test or Students t-test was used to determine the statistical significance of difference in measured parameters. Difference was considered significant at P<0.05. Cosinor analysis was performed on mRNA profiles using SPSS 17.0. The amplitude and acrophase obtained from cosinor analysis were further analyzed by Students t-test. The null hypothesis was rejected at P<0.05.
Abbreviations
- PER2:
-
PERIOD2
- FASPS:
-
familial advanced sleep phase syndrome
- ASPS:
-
advanced sleep phase syndrome
- ZT:
-
Zeitgeber Time
- MEF:
-
mouse embryonic fibroblast
- KO:
-
knockout
- BAC:
-
bacterial artificial chromosome
- ANOVA:
-
one-way analysis of variance
- RT-PCR:
-
reverse transcription PCR
- RORE:
-
Rev-erbα/RORα responsive elements
- Cry:
-
cryptochrome
References
Lowrey PL, Takahashi JS . Mammalian circadian biology: elucidating genome-wide levels of temporal organization. Annu Rev Genomics Hum Genet 2004; 5: 407–441.
Reppert SM, Weaver DR . Coordination of circadian timing in mammals. Nature 2002; 418: 935–941.
Young MW, Kay SA . Time zones: a comparative genetics of circadian clocks. Nat Rev Genet 2001; 2: 702–715.
Panda S, Antoch MP, Miller BH, Su AI, Schook AB, Straume M et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell 2002; 109: 307–320.
Schmutz I, Ripperger JA, Baeriswyl-Aebischer S, Albrecht U . The mammalian clock component PERIOD2 coordinates circadian output by interaction with nuclear receptors. Genes Dev 2010; 24: 345–357.
Fu L, Pelicano H, Liu J, Huang P, Lee C . The circadian gene Period2 plays an important role in tumor suppression and DNA damage response in vivo. Cell 2002; 111: 41–50.
Yang X . A wheel of time: the circadian clock, nuclear receptors, and physiology. Genes Dev 2010; 24: 741–747.
Gery S, Komatsu N, Baldjyan L, Yu A, Koo D, Koeffler HP . The circadian gene per1 plays an important role in cell growth and DNA damage control in human cancer cells. Mol Cell 2006; 22: 375–382.
Davis S, Mirick DK, Stevens RG . Night shift work, light at night, and risk of breast cancer. J Natl Cancer Inst 2001; 93: 1557–1562.
Baan R, Grosse Y, Straif K, Secretan B, El Ghissassi F, Bouvard V et al. A review of human carcinogens–Part F: chemical agents and related occupations. Lancet Oncol 2009; 10: 1143–1144.
Straif K, Baan R, Grosse Y, Secretan B, El Ghissassi F, Bouvard V et al. Carcinogenicity of shift-work, painting, and fire-fighting. Lancet Oncol 2007; 8: 1065–1066.
Jones CR, Campbell SS, Zone SE, Cooper F, DeSano A, Murphy PJ et al. Familial advanced sleep-phase syndrome: a short-period circadian rhythm variant in humans. Nat Med 1999; 5: 1062–1065.
Toh KL, Jones CR, He Y, Eide EJ, Hinz WA, Virshup DM et al. An hPer2 phosphorylation site mutation in familial advanced sleep phase syndrome. Science (New York, NY) 2001; 291: 1040–1043.
Xu Y, Padiath QS, Shapiro RE, Jones CR, Wu SC, Saigoh N et al. Functional consequences of a CKIdelta mutation causing familial advanced sleep phase syndrome. Nature 2005; 434: 640–644.
Xu Y, Toh KL, Jones CR, Shin JY, Fu YH, Ptacek LJ . Modeling of a human circadian mutation yields insights into clock regulation by PER2. Cell 2007; 128: 59–70.
Lang GA, Iwakuma T, Suh YA, Liu G, Rao VA, Parant JM et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell 2004; 119: 861–872.
Bae K, Jin X, Maywood ES, Hastings MH, Reppert SM, Weaver DR . Differential functions of mPer1, mPer2, and mPer3 in the SCN circadian clock. Neuron 2001; 30: 525–536.
Baylies MK, Bargiello TA, Jackson FR, Young MW . Changes in abundance or structure of the per gene product can alter periodicity of the Drosophila clock. Nature 1987; 326: 390–392.
Yang Z, Sehgal A . Role of molecular oscillations in generating behavioral rhythms in Drosophila. Neuron 2001; 29: 453–467.
Cook JL, May DL, Wilson BA, Walker TA . Differential induction of cytolytic susceptibility by E1A, myc, and ras oncogenes in immortalized cells. J Virol 1989; 63: 3408–3415.
Sherr CJ . Principles of tumor suppression. Cell 2004; 116: 235–246.
Ozturk N, Lee JH, Gaddameedhi S, Sancar A . Loss of cryptochrome reduces cancer risk in p53 mutant mice. Proc Natl Acad Sci USA 2009; 106: 2841–2846.
Embree-Ku M, Boekelheide K . Absence of p53 and FasL has sexually dimorphic effects on both development and reproduction. Exp Biol Med (Maywood) 2002; 227: 545–553.
Grechez-Cassiau A, Rayet B, Guillaumond F, Teboul M, Delaunay F . The circadian clock component BMAL1 is a critical regulator of p21WAF1/CIP1 expression and hepatocyte proliferation. J Biol Chem 2008; 283: 4535–4542.
Harper JW, Adami GR, Wei N, Keyomarsi K, Elledge SJ . The p21 Cdk-interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 1993; 75: 805–816.
Sjoblom T, Jones S, Wood LD, Parsons DW, Lin J, Barber TD et al. The consensus coding sequences of human breast and colorectal cancers. Science (New York, NY) 2006; 314: 268–274.
Winter SL, Bosnoyan-Collins L, Pinnaduwage D, Andrulis IL . Expression of the circadian clock genes Per1 and Per2 in sporadic and familial breast tumors. Neoplasia 2007; 9: 797–800.
Chen ST, Choo KB, Hou MF, Yeh KT, Kuo SJ, Chang JG . Deregulated expression of the PER1, PER2 and PER3 genes in breast cancers. Carcinogenesis 2005; 26: 1241–1246.
Gauger MA, Sancar A . Cryptochrome, circadian cycle, cell cycle checkpoints, and cancer. Cancer Res 2005; 65: 6828–6834.
Miller BH, McDearmon EL, Panda S, Hayes KR, Zhang J, Andrews JL et al. Circadian and CLOCK-controlled regulation of the mouse transcriptome and cell proliferation. Proc Natl Acad Sci US A 2007; 104: 3342–3347.
Yang Q, Pando BF, Dong G, Golden SS, van Oudenaarden A . Circadian gating of the cell cycle revealed in single cyanobacterial cells. Science (New York, NY) 2010; 327: 1522–1526.
Lu Y, Cross FR . Periodic cyclin-Cdk activity entrains an autonomous Cdc14 release oscillator. Cell 2010; 141: 268–279.
King DP, Zhao Y, Sangoram AM, Wilsbacher LD, Tanaka M, Antoch MP et al. Positional cloning of the mouse circadian clock gene. Cell 1997; 89: 641–653.
Wang X, Tang J, Xing L, Shi G, Ruan H, Gu X et al. Interaction of MAGED1 with nuclear receptors affects circadian clock function. Embo J 2010; 29: 1389–1400.
Morita S, Kojima T, Kitamura T . Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther 2000; 7: 1063–1066.
Acknowledgements
We thank David Weaver for reading the paper and giving insightful comments. We appreciate JS Takahashi for Clock mice and Fu & Ptáèek labs for PER2S662 transgenic mice. This work was funded by the National Science Foundation of China, the Distinguished Young Scholar Foundation (30725011 to YX), (10971152 to LY) and Ministry of Science and Technology (2010CB945102 to YX).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by C Borner
Supplementary Information accompanies the paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Gu, X., Xing, L., Shi, G. et al. The circadian mutation PER2S662G is linked to cell cycle progression and tumorigenesis. Cell Death Differ 19, 397–405 (2012). https://doi.org/10.1038/cdd.2011.103
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cdd.2011.103
Keywords
This article is cited by
-
Circadian disruption: from mouse models to molecular mechanisms and cancer therapeutic targets
Cancer and Metastasis Reviews (2023)
-
Circadian rhythms and cancers: the intrinsic links and therapeutic potentials
Journal of Hematology & Oncology (2022)
-
Molecular regulations of circadian rhythm and implications for physiology and diseases
Signal Transduction and Targeted Therapy (2022)
-
Metabolic rivalry: circadian homeostasis and tumorigenesis
Nature Reviews Cancer (2020)
-
Curcumin: a therapeutic strategy in cancers by inhibiting the canonical WNT/β-catenin pathway
Journal of Experimental & Clinical Cancer Research (2019)