Abstract
The worldwide incidence of bone disorders and conditions has been increasing. Bone is a nanomaterials composed of organic (mainly collagen) and inorganic (mainly nano-hydroxyapatite) components, with a hierarchical structure ranging from nanoscale to macroscale. In consideration of the serious limitation in traditional therapies, nanomaterials provide some new strategy in bone regeneration. Nanostructured scaffolds provide a closer structural support approximation to native bone architecture for the cells and regulate cell proliferation, differentiation, and migration, which results in the formation of functional tissues. In this article, we focused on reviewing the classification and design of nanostructured materials and nanocarrier materials for bone regeneration, their cell interaction properties, and their application in bone tissue engineering and regeneration. Furthermore, some new challenges about the future research on the application of nanomaterials for bone regeneration are described in the conclusion and perspectives part.
Similar content being viewed by others
Introduction
Nowadays more and more bone diseases such as bone infections, bone tumors, and bone loss need for bone regeneration. Bone tissue engineering is a complex and dynamic process that initiates with migration and recruitment of osteoprogenitor cells followed by their proliferation, differentiation, matrix formation along with remodeling of the bone.1 Bone scaffold is typically made of porous biodegradable materials that provide the mechanical support during repair and regeneration of damaged or diseased bone. Researches on bone tissue engineering over the past decades have inspired innovation in novel materials, processing techniques, performance evaluation, and applications. Significant progress has been made toward scaffold materials for structural support for desired osteogenesis and angiogenesis abilities. Bioresorbable scaffolds with controlled porosity and tailored properties are possible today due to innovation in scaffold fabrication using advanced technologies.
Natural bone derives its unique combination of mechanical properties from an architectural design that spans nanoscale to macroscopic dimensions, with precisely and carefully engineered interfaces. Many different groups have tried to manipulate the mechanical properties (e.g., stiffness, strength, and toughness) of scaffolds through the design of nanostructures (e.g., the inclusion of nanoparticles or nanofiber reinforcements in polymer matrices) to mimic bone’s natural nanocomposite architecture.
Within the stem cell niche, micro-/nanoscale interactions with extracellular matrix (ECM) components constitute another source of passive mechanical forces that can influence stem cell behaviors. The ECM is composed of a wide spectrum of structural proteins and polysaccharides that span over different length scales, with strands of collagen fibrils dominating at the nanometer level, with a diameter between 35 and 60 nm and a length that can extend over the micron range.2 It is via such well-choreographed spatiotemporal dialog between stem cells and their micro-/nano-environment that long-term maintenance and control of stem cell behavior are achieved. The advent of sophisticated small-scale technologies has now made it possible for researchers to fabricate platforms that can be used to gain valuable insights into stem cell biomechanics.3 Furthermore, bio-inspired and mimicking substrates with micro-/nanofeatures have been employed to understand and control stem cell differentiation. Nonetheless, despite the significance of stem cell mechanobiology, how mechanical stimuli regulates the behaviors of stem cells both in vivo and ex vivo have yet to be fully understood.4
To better mimic the nanostructure in natural ECM, over the past decade, scaffolds manufactured from nanofibers, nanotubes, nanoparticles, and hydrogel have recently emerged as promising candidates in producing scaffolds that resemble the ECM and efficiently replace defective tissues.5 Because natural tissues or organs are nanometer in dimension and cells directly interact with (and create) nanostructured ECMs, the biomimetic features and excellent physiochemical properties of nanomaterials play a key role in stimulating cell growth as well as guiding tissue regeneration.6
Nanostructured scaffolds for bone tissue engineering
Hierarchical organization of native bone ECM
Following the approach of scaffolding as a way of temporarily mimicking the ECM of bone, it is necessary to look at the chemical, mechanical, and structural properties of bone.
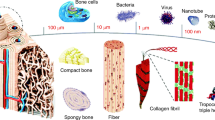
Bone is a sophisticated composite on different hierarchical levels, as shown in Figure 1. Bone tissue consists of two main parts, a compact shell called cortical bone and a porous core called spongiosa or trabecular bone (Figure 1a). Cortical bone is composed of repeating osteon units, whereas the cancellous bone is made of an interconnecting framework of trabeculae with bone marrow-filled free spaces. These trabeculae and osteon units are composed of collagen fibers and calcium phosphate crystals. The collagen fibrils include a 67 nm periodicity and 40 nm gaps between collagen molecules (Figure 1b).7,8 The HA crystals are embed in these gaps between collagen molecules and increase the rigidity of the bone (Figure 1c).9,10 The properties of bone tissues are strongly dependent on the structure and organization of the ECM and cells, where the organization of the ECM is hierarchical and spans several orders of magnitude (nm to cm).11 Thus, repair and reconstruction of bone defects require innovative strategies that account for the nanoscale to macroscale hierarchical assembly of tissue.
The microstructure and nanostructure of bone and the nanostructured material used in bone regeneration. (a) At the macroscopic level, bone consists of a dense shell of cortical bone with porous cancellous bone at both ends. (b) Repeating osteon units within cortical bone. In the osteons, 20–30 concentric layers of collagen fibers, called lamellae, are arranged at 90° surrounding the central canal, which contain blood vessels and nerves. (c) Collagen fibers (100–2 000 nm) are composed of collagen fibrils. The tertiary structure of collagen fibrils includes a 67 nm periodicity and 40 nm gaps between collagen molecules. The hydroxyapatite (HA) crystals are embedded in these gaps between collagen molecules and increase the rigidity of the bone. Nanostructures with features of nanopattern (d), nanofibers (e), nanotubers (f), nanopores (g), nanospheres (h), and nanocomposites (i) with structural components with a feature size in the nanoscale.
Bone regeneration requires four components: a morphogenetic signal, responsive host cells that will respond to the signal, a suitable carrier of this signal that can deliver it to specific sites then serve as a scaffold for the growth of the responsive host cells, and a viable, well-vascularized host bed.12 Bone tissue engineering, for the purpose of this review, is the use of a scaffolding material to either induce formation of bone from the surrounding tissue or to act as a carrier or template for implanted bone cells or other agents. Materials used as bone tissue-engineered scaffolds may be injectable or rigid, with the latter requiring an operative implantation procedure.
Biomimetic efforts in bone tissue engineering
As a bone tissue engineering, the ideal bone tissue scaffolds should be osteoconductive, osteoinductive, and osteogenic.13,14 Osteoconductivity requires these scaffolds to promote the attachment, survival, and migration of osteogenic cells. Osteoinductive scaffolds offer physical and biochemical factor to induce stem cells toward osteoblastic lineage. Osteogenic scaffolds contain osteogenic stem cells for bone regeneration. In a word, bone tissue engineering utilizes a biomimetic strategy which includes suitable scaffolds, biochemical and physical stimuli, stem cells, vascularization, and recapitulating the hierarchical organization of natural ECM to create functional bone tissues. These biomimetic efforts include choosing biomaterials that are present in native bone (e.g., HA and collagen), fabricating multiple scale architectures in scaffold especially with nanoscale components, and incorporating growth factors, e.g., bone morphogenetic proteins (BMPs), vascularization, and/or stem cells to provide a biomimetic niche for stimulating bone repair and regeneration.
Conventional tissue engineering scaffolds have used various pore-forming methods to recreate the macroscale and microscale properties of native tissues, but the nanoscale structures and properties were neglected. However, the nanoscale structures are crucial to regulating cell functions, such as proliferation, migration, differentiation, and the formation of ECM. To simulate the hierarchical organization of natural ECM, one important strategy is to build nanoscale and microscale features in the three-dimensional (3D) scaffolds design. The commonly accepted definition of nanomaterials refers to materials with clearly defined features between 1 and 100 nm, such as nanopattern,15 nanofibers,16 nanotubers,17 nanopores,18 nanospheres,19 and nanocomposites20,21 (Figure 1d–i).
Main types of nano-scaffolds for bone tissue engineering
Nanopattern
Stem cells are capable of differentiating into various types of cells, offering opportunities and alternatives not only for the treatment of diseases but also for the regeneration of tissues and organs beyond complex surgical treatments or tissue/organ transplantation. The construction of synthetic ECMs inspired by tissue-specific niches for programmed stem cell fate and response, such as proliferation and differentiation, is a topic of interest in the field of tissue regeneration.
Using nanogrooved matrices mimicking the native tissues, Kim et al. found that the body and nucleus of human mesenchymal stem cells (hMSCs) with the sparser nanogrooved pattern elongated and orientated more along the direction of nanogrooves than those with the relatively denser nanogroove patterns.15 In contrast, the perimeter of the cells was lower at the 1:3 pattern as compared to the 1:1 and 1:5 patterns spacing ratio (width:spacing, width = 550 nm) (Figure 2a). The effect of nano-topographical density on the osteo- or neurogenesis of hMSCs was significant at the 1:1 and 1:3 nanogrooved patterns, but not significant at 1:5 nanogrooved pattern compared to that at the flat substrate. It is demonstrated the effects of nano-topographical density on the morphology and differentiation of mesenchymal stem cells. The formation of cytoskeleton is necessary for the shape effect on the stem cell differentiation and that the Rho-associated protein kinase (ROCK)-pathway-related cell tension is responsible for this effect in the case of osteogenesis even in growth medium. The adipogenic differentiation does not seem to be simply negatively related to cell tension, and otherwise the adipogenic fraction might be minimum in the case of large aspect ratio of cells (with the highest cell tension).
Schematic depictions of representative nanotopography geometries. Three basic nanotopography geometries include nanogrooves (a), nanopost array (b), and nanopit array (c). The speculative pathways (d) for cell-shape-directed osteogenic and adipogenic differentiations of MSCs were examined in growth medium. RhoA, Ras homolog gene family member A; ROCK, Rho-associated protein kinase.
Jangho Kim supports the notion that multiscale hierarchical topography can be used as an efficient strategy for the design and manipulation of synthetic ECMs for stem cell-based bone regeneration.22 Nanopatterned hierarchical transplantable patches with precisely defined architectures and sizes were fabricated via capillary force lithography in combination with a micro-wrinkling method using poly(lactic-co-glycolic acid) (PLGA). A type of synthetic ECM comprised of hierarchically multiscale structures could provide native ECM-like topographical cues for controlling the adhesion and differentiation of hMSCs. Interestingly, the platform that integrates hMSCs into the multiscale hierarchical PLGA patch showed the potential to regenerate the bone tissues without complex surgical treatments. They work provides insight into the design and manipulation of functional engineered constructs using multi-scale hierarchical topography-based substrates for various biomedical applications, including stem cell therapy and tissue engineering.
Cell shape, in particular, the degree of cell spreading reflected in the cell area, is known to influence cell fate decisions of hMSC. Since cell area was shown to be regulated by the density of nanoposts, Ahn et al. explored whether the density of the nanoposts could also influence the cell fate decision of hMSC (Figure 2b).23 To investigate the influence of nanoposts density on actin cytoskeleton signaling, they determined the F-actin structure in hMSC cultured on different nanoposts density surfaces. The finding suggested that the nanoposts density might be capable of directly regulating cytoskeletal stiffness and the dynamic changes in stiffness correlated with the differentiation of hMSC into osteogenic or adipogenic lineages. Using spatially ordered and disordered arrays of nanopatterned c-RGDfK peptide with well-defined interpattern distances that ranged from 55 to 100 nm against a non-adhesive background, Huang et al. further characterized the influence of nanospacing on adhesion and spreading.24 When osteoblasts were cultured on nano-engineered substrates, it was found that a critical lateral inter-distance of 70 nm was required for effective clustering of integrins, beyond which (>70 nm) the cells were unable to spread effectively. It was proposed that an inability to cross-link the integrins at the cytoplasmic end by proteins such as talin and α-actin to a comparable size (60 nm) might be the reason for this limitation.
As is known, synthetically nanofabricated topography can also influence cell morphology, alignment, adhesion, migration, proliferation, and cytoskeleton organization.25 The symmetry and order of the nanopits was found to significantly affect the expression of osteopontin and osteocalcin, two bone-specific ECM proteins, in both cell types (Figure 2c).26 While hMSCs cultured on completely ordered or completely random nanopits did not lead to expression of these two proteins, hMSCs cultured on slightly irregular substrates did exhibit significant amounts of these proteins of interest. Increased bone nodule formation was also evident in hMSCs cultured on these substrates relative to substrates with either completely ordered or completely random features. The results from the studies demonstrated the potential of nanotopography to direct cell fate. Furthermore, the complementary findings of hMSCs cultured on nanogratings and ordered-disordered nanopits suggested the potential for selective, controllable differentiation based solely on the geometry of the nanotopographic substrate.
Collectively, a few common observations can be drawn from the before mentioned studies of the mechano-sensitivity of stem cells. All the studies have explicitly or implicitly suggested the involvement of cytoskeleton contractility in regulating the mechanosensitivity of stem cells, suggesting the importance of the force balance along the mechanical axis of the ECM–integrin–cytoskeleton linkage and their regulation by the mechanical signals in the stem cell niche (Figure 2d).27 Moreover, strong evidence suggested that the differentiation potentials of stem cells toward distinct lineages could be maximized if the cells were cultured in the mechanical microenvironment mimicking their tissue elasticity in vivo.28 In addition, nanoscale manipulation of surface features such as surface texture, geometry, spatial position, and height might potentially alter clustering of the integrins, the development of focal adhesions, and cytoskeletal structure, thus influencing the osteogenic differentiation to the surface.29
Electrospinning
The principle of electrospinning is that an electric field is used to overcome the surface tension of a polymer solution to shoot a jet of liquid out of a needle toward a conducting collector.30–32 The volatile solvent evaporates in the air leaving behind, under the right conditions, a polymer fiber with a diameter that can range from tens of nanometers to microns. Many parameters affect this process including polymer properties, solvent properties, solution flow rate, voltage, distance from the needle to the collector, and polymer concentration, among others.33,34 The wide range of polymers capable of being electrospun is appealing to bone tissue engineering and gives researchers flexibility in designing nanofibrous scaffolds. Generally, there are two types of polymers that are chosen: synthetic polymers or natural polymers. Synthetic polymers, such as poly(L-lactic acid) (PLLA), poly(glycolic acid) (PGA), and polycaprolactone (PCL), among others, provide great flexibility in synthesis, processing, and modification. However, these polymers lack bioactivity and special care needs to be taken to ensure that newly synthesized polymers are biocompatible. Many natural polymers, on the other hand, have inherent bioactivity with peptide sequences that affect cell adhesion, proliferation, and differentiation. Collagen, gelatin, silk, and chitosan, among others, are commonly used natural polymers for scaffold fabrication, but care must be taken to prevent denaturation when proteins are used.35
Since both synthetic and natural polymers have advantages and disadvantages, research has progressed to fabricate hybrid scaffolds in an effort to maximize the benefits of both. Yang et al. combined PCL with various amounts of chitosan to create bioactive nanofibers.36 Pure electrospun chitosan was too weak to be mechanically tested and pure PCL had reduced cell adhesion, but nanofibers produced from a 9.1% chitosan in PCL solution had the maximum Young’s modulus while significantly increasing cell adhesion compared to pure PCL. This novel hybrid scaffold takes advantage of the physical properties of the synthetic polymer and the bioactivity of the natural polymer while minimizing the disadvantages of both.
To develop biomimetic bone tissue engineering scaffolds for the repair of critical-sized calvarial defect, and growth factors can be incorporated into the polymer to create a controlled delivery system (Figure 3). Li et al. developed a new nanoparticle-embedded electrospun nanofiber scaffold for the controlled dual delivery of BMP-2 and dexamethasone (DEX).16 The scaffold was achieved by (1) the encapsulation of BMP-2 into bovine serum albumin (BSA) nanoparticles to maintain the bioactivity of BMP-2 and (2) the co-electrospinning of the blending solution composed of the BSA nanoparticles, DEX, and the poly(ε-caprolactone)-co-poly(ethylene glycol) copolymer. The in vitro studies showed that the bioactivity of DEX and BMP-2 was preserved in the dual-drug-loaded nanofiber scaffold, and a sequential release pattern in which most of the DEX was released in the original 8 days and the BMP-2 release lasted up to 35 days was achieved. The in vitro osteogenesis study demonstrated that the drug-loaded groups exhibited a strong ability to induce differentiation toward osteoblasts. In vivo osteogenesis studies also revealed that the degrees of repair of rat calvarial defect achieved with the drug-loaded nanofiber scaffolds were significantly better than those obtained with the blank materials; in particular, the dual-drug-loaded nanofiber scaffold manifested the best repair efficacy due to a synergistic effect of BMP-2 and DEX.
With electrospinning’s clear benefits, there are also some obstacles that need to be overcome. It remains difficult to create clinically relevant 3D constructs beyond a relatively 2D mat. For bone tissue engineering, a large 3D scaffold may be required. While new processing techniques have shown promise to increase the size and porosity of electrospun scaffolds. More work needs to be done to further help the architectural control. Having pores large enough for not only cell penetration, but also vascular in growth is imperative for a vascularized tissue such as bone.
Nanocomposites
Bone tissue itself represents a biological nanocomposite composed of organic (predominantly collagen type I) and inorganic (nanocrystal-line HA) components, with a hierarchical structure ranging from the microscale to the nanoscale.37 Nanobiomaterials and nanocomposites represent promising platforms in bone tissue engineering with a capacity to recapitulate the organization of natural ECM and the generation of functional bone tissues through osteo-mimetic architecture. The inherent properties of nanocomposites, such as increased wettability, roughness, and surface area, can also promote biomaterial-driven bone regeneration through increased protein adsorption, nutrient exchange, and porosity relative to macroscale biomaterials.
Mehta et al. have demonstrated an association between in vitro and in vivo studies of the PDLLA scaffolds with different incorporated VACNT-O:nHAp nanoparticles produced by electrode position and immersion into simulated body fluid solution.38 Our in vitro results showed that the produced scaffolds simultaneously promote bioactivity without any cytotoxic effects. In vivo study showed that the PDLLA/VACNT-O:nHAp2 scaffolds mimicked the immature bone and induced bone remodeling. The PDLLA/VACNT-O:nHAp scaffolds presented superior morphology and porosity properties compared to the PDLLA alone. These findings indicated surface improvement and the applicability of this new nano-biomaterial for bone regenerative medicine.
Nanocomposite scaffolds provide structural support for the cells, while changes to the nanoscale level of tissue hierarchy may have significant effects on cell-scaffold adhesion, integrin-triggered signaling pathways and cellular function; indeed, nanoscale features have been shown to have regulatory effects over multiple aspects of osteoblast and bone derived stem-cell behavior including adhesion, migration, proliferation, cell signaling, genetic expression, and stem cell fate. Consequentially, biomaterial design has focused on the introduction of nanoscale elements that elicit directed cellular behavior while imparting structural and mechanical advantages to the bone construct to induce the formation of functional tissues. Current methodologies employed in the fabrication of nanocomposites include electrospinning and molecular self-assembly.
Conclusions
The purpose of this article was to give a general description of studies of nanostructured materials for bone tissue engineering.
Nanophase ceramics, especially nano-HA, are popular bone substitutes, coatings and other filler materials due to their documented ability to promote mineralization. The nanometer grain sizes and high surface fraction of grain boundaries in nanoceramics increase osteoblast functions (such as adhesion, proliferation, and differentiation). Similar tendencies have been reported for other nanoceramics including alumina, zinc oxide, and titania; thus, providing evidence that, to some extent, it may not matter what implant chemistry is fabricated to have nanometer surface features to promote bone growth. However, this need further studies. For applications, synthetic and natural polymers, e.g., PGA, PLGA, PLLA, PLA, gelatin, collagen, chitosan, are regarded as excellent candidates for bone tissue engineering applications due to their biodegradability and ease of fabrication. Nanoporous or nanofibrous polymer matrices can be fabricated via electrospinning, phase separation, particulate leaching, chemical etching, and 3D printing techniques.
As mentioned in this article, features or ingredients of these scaffolds are nanoscale so that a variety of interactions can be stimulated at the cellular level. In some instances, some of those interactions also lead to toxicity, which can be of serious concerns. In particular, toxic responses to nanoparticles generated from the degradation of implanted nanomaterials, via wear debris from artificial joints with nano-features, and heavy metals (iron, nickel, and cobalt catalysts) remaining in carbon nanotubes, have all been reported. Sometimes nanoparticle interactions with biomolecules in vivo or their aggregation states may change their toxicity to humans. But the often contradictory results of current studies are clearly not enough to provide the final answer concerning nanomaterial toxicity. In-depth investigations of nanomaterials on human health and the environment are necessary to fully elucidate whether nanoparticles should be used in biomedical applications.
New frontiers of research should be directed toward better biomimicing the natural process of bone tissue regeneration such as coupling between angiogenesis and osteogenesis which may require progenitor cell recruitment and differentiation. Although it is difficult to mimic nature, recent scientific and technological findings show potential to achieve bone scaffolds that would encourage local and systemic biological functions. Proper selection of scaffold materials, their geometry, pore size, and size distribution, and ability to release biomolecules at a desired rate will play critical roles in future development of bone scaffolds.
To better mimic the nanostructure in natural ECM, over the past decade, scaffolds manufactured from nanofibers, nanotubes, nanoparticles, and hydrogel, have recently emerged as promising candidates in producing scaffolds that resemble the ECM and efficiently replace defective tissues. Even so, the combination of these materials in the form of nano-scaffolds is an under explored arena. The design of stronger and tougher scaffold materials requires incorporation of a hierarchical design encompassing many length scales from the nanolevel to generate strength (i.e., to mimic composite deformation of nanocrystals of HA and collagen) as well as micro-level structures to influence the crack path and generate toughness (e.g., to mimic osteons and cement lines). However, nanotechnology alone may not be the answer to improving the mechanical properties of scaffolds. The limitations in processing techniques, in part, have hampered the progress in the development of new scaffolds to form structures with a multidimensional architecture. The challenge is to use these technologies in combination with nanomaterials. It is possible that at the end an optimum scaffold combining several materials and techniques (e.g., a complex polymer structure can be created by ice-templating or computer-assisted fabrication that can subsequently be mineralized to achieve the desired mechanical and biodegradation responses) will become reality.
References
Giannoudis PV, Dinopoulos H, Tsiridis E . Bone substitutes: an update. Injury 2005; 36: S20–S27.
Li X, Wang L, Fan Y, Feng Q, Cui FZ, Watari F . Nanostructured scaffolds for bone tissue engineering. J Biomed Mater Res A 2013; 101: 2424–2435.
Stevens MM . Biomaterials for bone tissue engineering. Mater Today 2008; 11: 18–25.
Stevens MM, George JH . Exploring and engineering the cell surface interface. Science 2005; 310: 1135–1138.
McMahon RE, Wang L, Skoracki R, Mathur AB . Development of nanomaterials for bone repair and regeneration. J Biomed Mater Res B Appl Biomater 2013; 101: 387–397.
Chen CS, Mrksich M, Huang S, Whitesides GM, Ingber DE . Geometric control of cell life and death. Science 1997; 276: 1425–1428.
Robling AG, Stout SD . Morphology of the drifting osteon. Cells Tissues Organs 1999; 164: 192–204.
Faingold A, Cohen SR, Reznikov N, Wagner HD . Osteonal lamellae elementary units: lamellar microstructure, curvature and mechanical properties. Acta Biomater 2013; 9: 5956–5962.
Liu H, Yazici H, Ergun C, Webster TJ, Bermek H . An in vitro evaluation of the Ca/P ratio for the cytocompatibility of nano-to-micron particulate calcium phosphates for bone regeneration. Acta Biomater 2008; 4: 1472–1479.
Discher DE, Mooney DJ, Zandstra PW . Growth factors, matrices, and forces combine and control stem cells. Science 2009; 324: 1673–1677.
Odde D . Getting cells and tissues into shape. Cell 2011; 144: 325–326.
Gibson LJ . Biomechanics of cellular solids. J Biomech 2005; 38: 377–399.
Petite H, Viateau V, Bensaïd Wet al . Tissue-engineered bone regeneration. Nat Biotechnol 2000; 18: 959–963.
Vo TN, Kasper FK, Mikos AG . Strategies for controlled delivery of growth factors and cells for bone regeneration. Adv Drug Deliv Rev 2012; 64: 1292–1309.
Kim J, Kim HN, Lim KT et al. Designing nanotopographical density of extracellular matrix for controlled morphology and function of human mesenchymal stem cells. Sci Rep 2013; 3: 3552.
Li L, Zhou G, Wang Y, Yang G, Ding S, Zhou S . Controlled dual delivery of BMP-2 and dexamethasone by nanoparticle-embedded electrospun nanofibers for the efficient repair of critical-sized rat calvarial defect. Biomaterials 2015; 37: 218–229.
Xin Y, Jiang J, Huo K, Hu T, Chu PK . Bioactive SrTiO(3) nanotube arrays: strontium delivery platform on Ti-based osteoporotic bone implants. ACS Nano 2009; 3: 3228–3234.
Lemaire T, Pham TT, Capiez-Lernout E, de Leeuw NH, Naili S . Water in hydroxyapatite nanopores: possible implications for interstitial bone fluid flow. J Biomech 2015; 48: 3066–3071.
Wang H, Leeuwenburgh SC, Li Y, Jansen JA . The use of micro- and nanospheres as functional components for bone tissue regeneration. Tissue Eng Part B Rev 2012; 18: 24–39.
Kotela I, Podporska J, Soltysiak E, Konsztowicz KJ, Blazewicz M . Polymer nanocomposites for bone tissue substitutes. Ceram Int 2009; 35: 2475–2480.
Rogel MR, Qiu H, Ameer GA . The role of nanocomposites in bone regeneration. J Mater Chem 2008; 18: 4233–4241.
Kim J, Bae WG, Choung HW et al. Multiscale patterned transplantable stem cell patches for bone tissue regeneration. Biomaterials 2014; 35: 9058–9067.
Ahn EH, Kim Y, Kshitiz et al. Spatial control of adult stem cell fate using nanotopographic cues. Biomaterials 2014; 35: 2401–2410.
Huang J, Grater SV, Corbellini F et al. Impact of order and disorder in RGD nanopatterns on cell adhesion. Nano Lett 2009; 9: 1111–1116.
Li X, van Blitterswijk CA, Feng Q, Cui F, Watari F . The effect of calcium phosphate microstructure on bone-related cells in vitro. Biomaterials 2008; 29: 3306–3316.
Dalby MJ, Gadegaard N, Tare R et al. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat Mater 2007; 6: 997–1003.
Yao X, Peng R, Ding J . Effects of aspect ratios of stem cells on lineage commitments with and without induction media. Biomaterials 2013; 34: 930–939.
Engler AJ, Sen S, Sweeney HL, Discher DE . Matrix elasticity directs stem cell lineage specification. Cell 2006; 126: 677–689.
Guilak F, Cohen DM, Estes BT, Gimble JM, Liedtke W, Chen CS . Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell 2009; 5: 17–26.
Jiang T, Carbone EJ, Lo KWH, Laurencin CT . Electrospinning of polymer nanofibers for tissue regeneration. Prog Polym Sci 2015; 46: 1–24.
Nair LS, Bhattacharyya S, Laurencin CT . Development of novel tissue engineering scaffolds via electrospinning. Expert Opin Biol Ther 2004 4: 659–668.
Kumar PR, Khan N, Vivekanandhan S, Satyanarayana N, Mohanty AK, Misra M . Nanofibers: effective generation by electrospinning and their applications. J Nanosci Nanotechnol 2012 12: 1–25.
Jaworek A, Sobczyk AT . Electrospraying route to nanotechnology: an overview. J Electrostat 2008; 66: 197–219.
Deitzel JM, Kleinmeyer J, Harris D, Beck Tan NC . The effect of processing variables on the morphology of electrospun nanofibers and textiles. Polymer 2001; 42: 261–272.
Ifuku S, Saimoto H . Chitin nanofibers: preparations, modifications, and applications. Nanoscale 2012; 4: 3308–3318.
Yang X, Chen X, Wang H . Acceleration of osteogenic differentiation of preosteoblastic cells by chitosan containing nanofibrous scaffolds. Biomacromolecules 2009; 10: 2772–2778.
Okamoto M, John B . Synthetic biopolymer nanocomposites for tissue engineering scaffolds. Prog Polym Sci 2013; 38: 1487–1503.
El-Ghannam A, Ning CQ, Mehta J . Cyclosilicate nanocomposite: a novel resorbable bioactive tissue engineering scaffold for BMP and bone-marrow cell delivery. J Biomed Mater Res A 2004; 71: 377–390.
Acknowledgements
This work was funded by Funding of National Natural Science Foundation of China (81470721, 31170929, 81321002), Sichuan Science and Technology Innovation Team (2014TD0001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This work is licensed under a Creative Commons Attribution NonCommercial-NoDerivs 4.0 Unported License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Gong, T., Xie, J., Liao, J. et al. Nanomaterials and bone regeneration. Bone Res 3, 15029 (2015). https://doi.org/10.1038/boneres.2015.29
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/boneres.2015.29
This article is cited by
-
The current applications of nano and biomaterials in drug delivery of dental implant
BMC Oral Health (2024)
-
Bioinspired Hollow Mesoporous Silica Nanoparticles Coating on Titanium Alloy with Hierarchical Structure for Modulating Cellular Functions
Journal of Bionic Engineering (2024)
-
Preparation of ciprofloxacin-loaded oyster shell derived hydroxyapatite composite film for biomedical applications
Journal of the Australian Ceramic Society (2023)
-
A Comprehensive Review on the Classification, Uses, Sources of Nanoparticles (NPs) and Their Toxicity on Health
Aerosol Science and Engineering (2023)
-
The improved bioactive n-HA/PA66 cage versus the PEEK cage in anterior cervical fusion: results from a 6-year follow-up and a case-matched study
BMC Musculoskeletal Disorders (2022)