Abstract
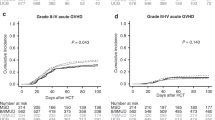
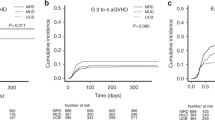
Myelodysplastic syndrome (MDS) is a heterogeneous group of hematological malignancies with considerably variable prognoses and curable only with hematopoietic cell transplantation (HCT). Few studies comparing MDS HCT outcomes between sibling and umbilical cord blood (UCB) donors exist. Using the University of Minnesota Blood and Marrow Transplant (BMT) database, we retrospectively analyzed HCT outcomes among 89 MDS patients undergoing either sibling or double UCB HCT in 2000–2013. We observed similar survival, relapse and non-relapse mortality between sibling and UCB donor sources. Relapse was increased in those with monosomal karyotype (P=0.04) and with reduced intensity conditioning (P<0.01). In summary, our data highlight similar MDS HCT outcomes regardless of donor source and support the use of UCB as an alternative donor when a sibling is unavailable.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sierra J, Perez WS, Rozman C, Carreras E, Klein JP, Rizzo JD et al. Bone marrow transplantation from HLA-identical siblings as treatment for myelodysplasia. Blood 2002; 100: 1997–2004.
Warlick ED, Cioc A, Defor T, Dolan M, Weisdorf D . Allogeneic stem cell transplantation for adults with myelodysplastic syndromes: importance of pretransplant disease burden. Biol Blood Marrow Transplant 2009; 15: 30–38.
Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood 1997; 89: 2079–2088.
Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Solé F et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood 2012; 120: 2454–2465.
Koreth J, Pidala J, Perez WS, Deeg HJ, Garcia-Manero G, Malcovati L et al. Role of reduced-intensity conditioning allogeneic hematopoietic stem-cell transplantation in older patients with de novo myelodysplastic syndromes: an international collaborative decision analysis. J Clin Oncol 2013; 31: 2662–2670.
Cutler CS, Lee SJ, Greenberg P, Deeg HJ, Pérez WS, Anasetti C et al. A decision analysis of allogeneic bone marrow transplantation for the myelodysplastic syndromes: delayed transplantation for low-risk myelodysplasia is associated with improved outcome. Blood 2004; 104: 579–585.
Ustun C, Trottier BJ, Sachs Z, DeFor TE, Shune L, Courville EL et al. Monosomal karyotype at the time of diagnosis or transplantation predicts outcomes of allogeneic hematopoietic cell transplantation in myelodysplastic syndrome. Biol Blood Marrow Transplant 2015; 21: 866–872.
Della Porta MG, Alessandrino EP, Bacigalupo A, van Lint MT, Malcovati L, Pascutto C et al. Predictive factors for the outcome of allogeneic transplantation in patients with MDS stratified according to the revised IPSS-R. Blood 2014; 123: 2333–2342.
Trottier BJ, Sachs Z, DeFor TE, Shune L, Dolan M, Weisdorf DJ et al. Novel disease burden assessment predicts allogeneic transplantation outcomes in myelodysplastic syndrome. Bone Marrow Transplant 2015; 51: 199–204.
Saber W, Cutler CS, Nakamura R, Zhang MJ, Atallah E, Rizzo JD et al. Impact of donor source on hematopoietic cell transplantation outcomes for patients with myelodysplastic syndromes (MDS). Blood 2013; 122: 1974–1982.
Lim Z, Brand R, Martino R, van Biezen A, Finke J, Bacigalupo A et al. Allogeneic hematopoietic stem-cell transplantation for patients 50 years or older with myelodysplastic syndromes or secondary acute myeloid leukemia. J Clin Oncol 2010; 28: 405–411.
Robin M, Sanz GF, Ionescu I, Rio B, Sirvent A, Renaud M et al. Unrelated cord blood transplantation in adults with myelodysplasia or secondary acute myeloblastic leukemia: a survey on behalf of Eurocord and CLWP of EBMT. Leukemia 2011; 25: 75–81.
Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A et alThe 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood 2009; 114: 937–951.
Breems DA, Van Putten WL, De Greef GE, Van Zelderen-Bhola SL, Gerssen-Schoorl KB, Mellink CH et al. Monosomal karyotype in acute myeloid leukemia: a better indicator of poor prognosis than a complex karyotype. J Clin Oncol 2008; 26: 4791–4797.
Brunstein CG, Setubal DC, Wagner JE . Expanding the role of umbilical cord blood transplantation. Br J Haematol 2007; 137: 20–35.
Champlin R, Khouri I, Shimoni A, Gajewski J, Kornblau S, Molldrem J et al. Harnessing graft-versus-malignancy: non-myeloablative preparative regimens for allogeneic haematopoietic transplantation, an evolving strategy for adoptive immunotherapy. Br J Haematol 2000; 111: 18–29.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant 2009; 15: 1628–1633.
Giralt S, Logan B, Rizzo D, Zhang MJ, Ballen K, Emmanouilides C et al. Reduced-intensity conditioning for unrelated donor progenitor cell transplantation: long-term follow-up of the first 285 reported to the national marrow donor program. Biol Blood Marrow Transplant 2007; 13: 844–852.
Majhail NS, Brunstein CG, Shanley R, Sandhu K, McClune B, Oran B et al. Reduced-intensity hematopoietic cell transplantation in older patients with AML/MDS: umbilical cord blood is a feasible option for patients without HLA-matched sibling donors. Bone Marrow Transplant 2012; 47: 494–498.
Warlick ED, Tomblyn M, Cao Q, Defor T, Blazar BR, Macmillan M et al. Reduced-intensity conditioning followed by related allografts in hematologic malignancies: long-term outcomes most successful in indolent and aggressive non-Hodgkin lymphomas. Biol Blood Marrow Transplant 2011; 17: 1025–1032.
Kaplan EL, Meier P . Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Lin D . Non-parametric inference for cumulative incidence functions in competing risks studies. Stat Med 1997; 16: 901–910.
Cox D . Regression models and life tables. J Royal Stat Soc B 1972; 34: 187–220.
Fine JP, Gray RJ . A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 1999; 94: 496–509.
Colett D . Modelling Survival Data in Medical Research. Chapman & Hall/CRC: London, UK, 2003.
Warlick ED, Peffault de Latour R, Shanley R, Robin M, Bejanyan N, Xhaard A et al. Allogeneic hematopoietic cell transplantation outcomes in acute myeloid leukemia: similar outcomes regardless of donor type. Biol Blood Marrow Transplant 2015; 21: 357–363.
Robin M, Ruggeri A, Labopin M, Niederwieser D, Tabrizi R, Sanz G et al. Comparison of unrelated cord blood and peripheral blood stem cell transplantation in adults with myelodysplastic syndrome after reduced-intensity conditioning regimen: a collaborative study from Eurocord (Cord blood Committee of Cellular Therapy & Immunobiology Working Party of EBMT) and Chronic Malignancies Working Party. Biol Blood Marrow Transplant 2015; 21: 489–495.
Deeg HJ, Scott BL, Fang M, Shulman HM, Gyurkocza B, Myerson D et al. Five-group cytogenetic risk classification, monosomal karyotype, and outcome after hematopoietic cell transplantation for MDS or acute leukemia evolving from MDS. Blood 2012; 120: 1398–1408.
Scott B, Pasquini M, Logan B, Wu J, Devine S, Porter DL et al. Results of a phase III randomized, multi-center study of allogeneic stem cell transplantation after high versus reduced intensity conditioning in patients with myelodysplastic syndrome (MDS) or acute myeloid leukemia (AML): Blood and Marrow Transplant Clinical Trials Network (BMT CTN) 0901 [abstract]. Blood 2015; 126: LBA-8.
Acknowledgements
This work was supported in part by grants from the National Cancer Institute P01 CA65493 (to CB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Pourhassan, H., DeFor, T., Trottier, B. et al. MDS disease characteristics, not donor source, predict hematopoietic stem cell transplant outcomes. Bone Marrow Transplant 52, 532–538 (2017). https://doi.org/10.1038/bmt.2016.303
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2016.303