Abstract
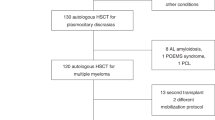
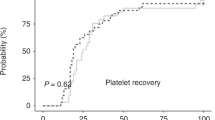
Outcomes in multiple myeloma (MM) have improved significantly with novel agent therapy and autologous stem cell transplantation (ASCT). ASCTs are typically planned as either tandem or a single transplant with additional stored PBSCs available for a second salvage transplant. To accommodate these strategies, many centers routinely collect and store adequate PBSCs for two ASCTs. We analyzed the cost associated with this practice by determining the expenses of PBSC collection, cryopreservation and storage, and the ultimate use of additional cryopreserved PBSCs in patients who had undergone at least one ASCT. There were 889 MM patients transplanted between 1993 and 2011 at our center. Most (N=726) had residual PBSCs in storage after their first ASCT (ASCT1). Only 135 patients underwent a second ASCT within a median of 14 months after ASCT1. The percentage of patients receiving a second ASCT declined over time. The resources required to collect and store unused PBSCs added up to 336 extra patient days of apheresis and 41 587 extra patient months of cryopreservation, translating into an average extra cost per patient of US$4981.12. A reconsideration of conventional PBSC collection and storage practices would save significant cost for the majority of MM patients who never undergo a second ASCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF et al. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. Intergroupe Francais du Myelome. N Engl J Med 1996; 335: 91–97.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K et al. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med 2003; 348: 1875–1883.
Attal M, Harousseau JL, Facon T, Guilhot F, Doyen C, Fuzibet JG et al. Single versus double autologous stem-cell transplantation for multiple myeloma. N Engl J Med 2003; 349: 2495–2502.
Regelink JC, van Roessel CH, van Galen KP, Ossenkoppele GJ, Huijgens PC, Zweegman S . Long-term follow-up of tandem autologous stem-cell transplantation in multiple myeloma. J Clin Oncol 2010; 28: e741–e743 author reply e744–e775.
Auner HW, Szydlo R, Rone A, Chaidos A, Giles C, Kanfer E et al. Salvage autologous stem cell transplantation for multiple myeloma relapsing or progressing after up-front autologous transplantation. Leuk Lymphoma 2013; 54: 2200–2204.
Lemieux E, Hulin C, Caillot D, Tardy S, Dorvaux V, Michel J et al. Autologous stem cell transplantation: an effective salvage therapy in multiple myeloma. Biol Blood Marrow Transplant 2013; 19: 445–449.
Michaelis LC, Saad A, Zhong X, Le-Rademacher J, Freytes CO, Marks DI et al. Salvage second hematopoietic cell transplantation in myeloma. Biol Blood Marrow Transplant 2013; 19: 760–766.
Huijgens PC, Dekker-Van Roessel HM, Jonkhoff AR, Admiraal GC, Zweegman S, Schuurhuis GJ et al. High-dose melphalan with G-CSF-stimulated whole blood rescue followed by stem cell harvesting and busulphan/cyclophosphamide with autologous stem cell transplantation in multiple myeloma. Bone Marrow Transplant 2001; 27: 925–931.
Cavo M, Tosi P, Zamagni E, Cellini C, Tacchetti P, Patriarca F et al. Prospective, randomized study of single compared with double autologous stem-cell transplantation for multiple myeloma: Bologna 96 clinical study. J Clin Oncol 2007; 25: 2434–2441.
Singhal S, Mehta J, Desikan R, Ayers D, Roberson P, Eddlemon P et al. Antitumor activity of thalidomide in refractory multiple myeloma. N Engl J Med 1999; 341: 1565–1571.
Cavo M, Tacchetti P, Patriarca F, Petrucci MT, Pantani L, Galli M et al. Bortezomib with thalidomide plus dexamethasone compared with thalidomide plus dexamethasone as induction therapy before, and consolidation therapy after, double autologous stem-cell transplantation in newly diagnosed multiple myeloma: a randomised phase 3 study. Lancet 2010; 376: 2075–2085.
Kim HJ, Yoon SS, Lee DS, Sohn SK, Eom HS, Lee JL et al. Sequential vincristine, adriamycin, dexamethasone (VAD) followed by bortezomib, thalidomide, dexamethasone (VTD) as induction, followed by high-dose therapy with autologous stem cell transplant and consolidation therapy with bortezomib for newly diagnosed multiple myeloma: results of a phase II trial. Ann Hematol 2012; 91: 249–256.
McCarthy PL, Owzar K, Hofmeister CC, Hurd DD, Hassoun H, Richardson PG et al. Lenalidomide after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1770–1781.
Attal M, Harousseau JL, Leyvraz S, Doyen C, Hulin C, Benboubker L et al. Maintenance therapy with thalidomide improves survival in patients with multiple myeloma. Blood 2006; 108: 3289–3294.
Attal M, Lauwers-Cances V, Marit G, Caillot D, Moreau P, Facon T et al. Lenalidomide maintenance after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1782–1791.
Sonneveld P, Schmidt-Wolf IG, van der Holt B, El Jarari L, Bertsch U, Salwender H et al. Bortezomib induction and maintenance treatment in patients with newly diagnosed multiple myeloma: results of the randomized phase III HOVON-65/ GMMG-HD4 trial. J Clin Oncol 2012; 30: 2946–2955.
Lenalidomide in Treating Patients Who Are Undergoing Autologous Stem Cell Transplant for Multiple Myeloma. (nd) https://clinicaltrials.gov/ct2/show/results/NCT00114101?sect=X70156#outcome1 (last accessed 10 December 2014).
Burzynski JA, Toro JJ, Patel RC, Lee S, Greene RE, Ochoa-Bayona JL et al. Toxicity of a second autologous peripheral blood stem cell transplant in patients with relapsed or recurrent multiple myeloma. Leuk Lymphoma 2009; 50: 1442–1447.
Olin RL, Vogl DT, Porter DL, Luger SM, Schuster SJ, Tsai DE et al. Second auto-SCT is safe and effective salvage therapy for relapsed multiple myeloma. Bone Marrow Transplant 2009; 43: 417–422.
Jimenez-Zepeda VH, Mikhael J, Winter A, Franke N, Masih-Khan E, Trudel S et al. Second autologous stem cell transplantation as salvage therapy for multiple myeloma: impact on progression-free and overall survival. Biol Blood Marrow Transplant 2012; 18: 773–779.
Fermand JP, Ravaud P, Chevret S, Divine M, Leblond V, Belanger C et al. High-dose therapy and autologous peripheral blood stem cell transplantation in multiple myeloma: up-front or rescue treatment? Results of a multicenter sequential randomized clinical trial. Blood 1998; 92: 3131–3136.
Moreau P, San Miguel J, Ludwig H, Schouten H, Mohty M, Dimopoulos M et al. Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2013; 24: vi133–vi137.
Cavo M, Rajkumar SV, Palumbo A, Moreau P, Orlowski R, Blade J et al. International Myeloma Working Group consensus approach to the treatment of multiple myeloma patients who are candidates for autologous stem cell transplantation. Blood 2011; 117: 6063–6073.
Anderson KC, Alsina M, Bensinger W, Biermann JS, Cohen AD, Devine S et al. Multiple myeloma, version 1.2013. J Natl Compr Cancer Netw 2013; 11: 11–17.
Byrne M, Salmasinia D, Leather H, Cogle CR, Davis A, Hsu JW et al. Tandem autologous stem cell transplantation for multiple myeloma patients based on response to their first transplant—a Prospective Phase II Study. Clin Med Insights Oncol 2014; 8: 101–105.
Cavo M, Salwender H, Rosiñol L, Moreau P, Petrucci M, Blau I et al. Double vs Single Autologous Stem Cell Transplantation after Bortezomib-Based Induction Regimens for Multiple Myeloma: An Integrated Analysis of Patient-Level Data from Phase European III Studies. American Society of Hematology: New Orleans, LA, USA, 2013.
Calandra G, McCarty J, McGuirk J, Tricot G, Crocker SA, Badel K et al. AMD3100 plus G-CSF can successfully mobilize CD34+ cells from non-Hodgkin’s lymphoma, Hodgkin’s disease and multiple myeloma patients previously failing mobilization with chemotherapy and/or cytokine treatment: compassionate use data. Bone Marrow Transplant 2008; 41: 331–338.
DiPersio JF, Stadtmauer EA, Nademanee A, Micallef IN, Stiff PJ, Kaufman JL et al. Plerixafor and G-CSF versus placebo and G-CSF to mobilize hematopoietic stem cells for autologous stem cell transplantation in patients with multiple myeloma. Blood 2009; 113: 5720–5726.
DiPersio JF, Micallef IN, Stiff PJ, Bolwell BJ, Maziarz RT, Jacobsen E et al. Phase III prospective randomized double-blind placebo-controlled trial of plerixafor plus granulocyte colony-stimulating factor compared with placebo plus granulocyte colony-stimulating factor for autologous stem-cell mobilization and transplantation for patients with non-Hodgkin's lymphoma. J Clin Oncol 2009; 27: 4767–4773.
Acknowledgements
We greatly appreciate the contribution of the database managers at the Fred Hutchinson Cancer Research Center and Seattle Cancer Care Alliance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This study or any of its parts have not been presented elsewhere.
Rights and permissions
About this article
Cite this article
Phipps, C., Linenberger, M., Holmberg, L. et al. Utilization of stored autologous PBSCs to support second autologous transplantation in multiple myeloma patients in the era of novel agent therapy. Bone Marrow Transplant 50, 663–667 (2015). https://doi.org/10.1038/bmt.2015.13
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.13
This article is cited by
-
Trends in utilization of stored cryopreserved autologous peripheral hematopoietic cells intended for a second (or beyond) autologous hematopoietic cell transplantation in patients with multiple myeloma: a single center experience
Bone Marrow Transplantation (2023)
-
Significant costs and low utilization of stored peripheral blood stem cells for salvage autologous transplant in multiple myeloma patients including those meeting mSMART criteria
Bone Marrow Transplantation (2021)