Abstract
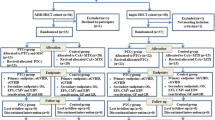
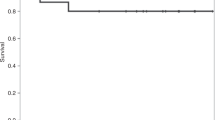
Non-remitting patients with hematologic myeloid malignancies have poor prognosis. To overcome this problem, we investigated the use of reduced-intensity preconditioning umbilical cord blood transplantation (RICBT) combined with recombinant G-CSF (rG-CSF) with high-dose Ara-C, fludarabine, melphalan, and 4 Gy of TBI in a phase I/II study in patients with non-remitting myeloid hematologic malignancies. Thirteen patients were enrolled, including 12 with non-remitting AML and one patient with blastic crisis CML (CML-BC). The patients’ median age was 45 years, with a median comorbidity index of 4. All patients received 4/6 serological HLA-antigen matched unrelated umbilical cord blood. All patients were engrafted within 30 days after RICBT (median, 20 days; range, 14–29) and achieved complete remission without prior hematopoiesis. Common grade III non-hematologic toxicities included eight cases of transient mucositis (62%) and six cases of febrile neutropenia (46%). Transplant-related mortality was 7.7%. The 1-year overall survival was 28.6% in cases without post-RICBT treatment and 83.3% in cases with post-RICBT treatment. These data suggest that in active AML and CML-BC, the combination of rG-CSF with high-dose Ara-C and fludarabine/melphalan/4 Gy TBI with a reduced-intensity preconditioning regimen is well tolerated, secures engraftment and has significant anti-leukemia activity. In addition, performing post-RICBT treatment may provide high-quality long-term survival and remission.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Druker BJ, Guilhot F, O’Brien SG, Gathmann I, Kantarjian H, Gattermann N et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. New Engl J Med 2006; 355: 2408–2417.
Slovak ML, Kopecky KJ, Cassileth PA, Harrington DH, Theil KS, Mohamed A et al. Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: a Southwest Oncology Group/Eastern Cooperative Oncology Group Study. Blood 2000; 96: 4075–4083.
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G et al. The importance of diagnostic cytogenetics on outcome in AML: analysis of 1612 patients entered into the MRC AML 10 trial. The Medical Research Council Adult and Children's Leukaemia Working Parties. Blood 1998; 92: 2322–2333.
Petersen FB, Buckner CD, Appelbaum FR, Clift RA, Sanders JE, Bensinger WI et al. Busulfan, cyclophosphamide and fractionated total body irradiation as a preparatory regimen for marrow transplantation in patients with advanced hematological malignancies: a phase I study. Bone Marrow Transplant 1989; 4: 617–623.
Davies SM, Ramsay NK, Klein JP, Weisdorf DJ, Bolwell B, Cahn JY et al. Comparison of preparative regimens in transplants for children with acute lymphoblastic leukemia. J Clin Oncol 2000; 18: 340–347.
Kanda Y, Sakamaki H, Sao H, Okamoto S, Kodera Y, Tanosaki R et al. Effect of conditioning regimen on the outcome of bone marrow transplantation from an unrelated donor. Biol Blood Marrow Transplant 2005; 11: 881–889.
Shimoni A, Hardan I, Shem-Tov N, Rand A, Herscovici C, Yerushalmi R et al. Comparison between two fludarabine-based reduced-intensity conditioning regimens before allogeneic hematopoietic stem-cell transplantation: fludarabine/melphalan is associated with higher incidence of acute graft-versus-host disease and non-relapse mortality and lower incidence of relapse than fludarabine/busulfan. Leukemia 2007; 21: 2109–2116.
Chae YS, Sohn SK, Kim JG, Cho YY, Moon JH, Shin HJ et al. New myeloablative conditioning regimen with fludarabine and busulfan for allogeneic stem cell transplantation: comparison with BuCy2. Bone Marrow Transplant 2007; 40: 541–547.
Giralt S, Thall PF, Khouri I, Wang X, Braunschweig I, Ippolitti C et al. Melphalan and purine analog-containing preparative regimens: reduced-intensity conditioning for patients with hematologic malignancies undergoing allogeneic progenitor cell transplantation. Blood 2001; 97: 631–637.
Oran B, Giralt S, Saliba R, Hosing C, Popat U, Khouri I et al. Allogeneic hematopoietic stem cell transplantation for the treatment of high-risk acute myelogenous leukemia and myelodysplastic syndrome using reduced-intensity conditioning with fludarabine and melphalan. Biol Blood Marrow Transplant 2007; 13: 454–462.
Narimatsu H, Watanabe M, Kohno A, Sugimoto K, Kuwatsuka Y, Uchida T et al. High incidence of graft failure in unrelated cord blood transplantation using a reduced-intensity preparative regimen consisting of fludarabine and melphalan. Bone Marrow Transplant 2008; 41: 753–756.
Miyakoshi S, Yuji K, Kami M, Kusumi E, Kishi Y, Kobayashi K et al. Successful engraftment after reduced-intensity umbilical cord blood transplantation for adult patients with advanced hematological diseases. Clin Cancer Res 2004; 10: 3586–3592.
Yuji K, Miyakoshi S, Kato D, Miura Y, Myojo T, Murashige N et al. Reduced-intensity unrelated cord blood transplantation for patients with advanced malignant lymphoma. Biol Blood Marrow Transplant 2005; 11: 314–318.
Tomonari A, Takahashi S, Ooi J, Nakaoka T, Takasugi K, Uchiyama M et al. Cord blood transplantation for acute myelogenous leukemia using a conditioning regimen consisting of granulocyte colony-stimulating factor-combined high-dose cytarabine, fludarabine, and total body irradiation. Eur J Haematol 2006; 77: 46–50.
Takahashi S, Ooi J, Tomonari A, Konuma T, Tsukada N, Oiwa-Monna M et al. Comparative single-institute analysis of cord blood transplantation from unrelated donors with bone marrow or peripheral blood stem-cell transplants from related donors in adult patients with hematologic malignancies after myeloablative conditioning regimen. Blood 2007; 109: 1322–1330.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005; 106: 2912–2919.
Kodera Y, Morishima Y, Kato S, Akiyama Y, Sao H, Matsuyama T et al. Analysis of 500 bone marrow transplants from unrelated donors (UR-BMT) facilitated by the Japan Marrow Donor Program: confirmation of UR-BMT as a standard therapy for patients with leukemia and aplastic anemia. Bone Marrow Transplant 1999; 24: 995–1003.
Nishihira H, Kato K, Isoyama K, Takahashi TA, Kai S, Kato S et al. The Japanese cord blood bank network experience with cord blood transplantation from unrelated donors for haematological malignancies: an evaluation of graft-versus-host disease prophylaxis. Br J Haematol 2003; 120: 516–522.
Kanda Y, Mineishi S, Saito T, Saito A, Yamada S, Ohnishi M et al. Long-term low-dose acyclovir against varicella-zoster virus reactivation after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2001; 28: 689–692.
Hughes WT, Armstrong D, Bodey GP, Bow EJ, Brown AE, Calandra T et al. 2002 guidelines for the use of antimicrobial agents in neutropenic patients with cancer. Clin Infect Dis 2002; 34: 730–751.
Freifeld AG, Bow EJ, Sepkowitz KA, Boeckh MJ, Ito JI, Mullen CA et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis 2011; 52: 427–431.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15: 825–828.
Sullivan KM, Agura E, Anasetti C, Appelbaum F, Badger C, Bearman S et al. Chronic graft-versus-host disease and other late complications of bone marrow transplantation. Semin Hematol 1991; 28: 250–259.
Thiede C, Florek M, Bornhäuser M, Ritter M, Mohr B, Brendel C et al. Rapid quantification of mixed chimerism using multiplex amplification of short tandem repeat markers and fluorescence detection. Bone Marrow Transplant 1999; 23: 1055–1060.
Kanda Y . Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transplant 2013; 48: 452–458.
Kishi Y, Kami M, Miyakoshi S, Kanda Y, Murashige N, Teshima T et al. Early immune reaction after reduced-intensity cord-blood transplantation for adult patients. Transplantation 2005; 80: 34–40.
Atsuta Y, Suzuki R, Yoshimi A, Gondo H, Tanaka J, Hiraoka A et al. Unification of hematopoietic stem cell transplantation registries in Japan and establishment of the TRUMP System. Int J Hematol 2007; 86: 269–274.
Rizzieri DA, Long GD, Vredenburgh JJ, Gasparetto C, Morris A, Stenzel TT et al. Successful allogeneic engraftment of mismatched unrelated cord blood following a nonmyeloablative preparative regimen. Blood 2001; 98: 3486–3488.
Chao NJ, Koh LP, Long GD, Gasparetto C, Horwitz M, Morris A et al. Adult recipients of umbilical cord blood transplants after nonmyeloablative preparative regimens. Biol Blood Marrow Transplant 2004; 10: 569–575.
Mancías-Guerra C, Ruiz-Delgado GJ, Manzano C, Díaz-Hernandez MA, Tarín-Arzaga LC, González-Llano O et al. Umbilical cord blood transplantation using non-myeloablative conditioning: the Mexican experience. Hematology 2006; 11: 355–359.
Misawa M, Kai S, Okada M, Nakajima T, Nomura K, Wakae T et al. Reduced-intensity conditioning followed by unrelated umbilical cord blood transplantation for advanced hematologic malignancies: rapid engraftment in bone marrow. Int J Hematol 2006; 83: 74–79.
Komatsu T, Narimatsu H, Yoshimi A, Kurita N, Kusakabe M, Hori A et al. Successful engraftment of mismatched unrelated cord blood transplantation following reduced intensity preparative regimen using fludarabine and busulfan. Ann Hematol 2007; 86: 49–54.
Miyakoshi S, Kami M, Tanimoto T, Yamaguchi T, Narimatsu H, Kusumi E et al. Tacrolimus as prophylaxis for acute graft-versus-host disease in reduced intensity cord blood transplantation for adult patients with advanced hematologic diseases. Transplantation 2007; 84: 316–322.
Brunstein CG, Barker JN, Weisdorf DJ, DeFor TE, Miller JS, Blazar BR et al. Umbilical cord blood transplantation after nonmyeloablative conditioning: impact on transplantation outcomes in 110 adults with hematologic disease. Blood 2007; 110: 3064–3070.
Uchida N, Wake A, Takagi S, Yamamoto H, Kato D, Matsuhashi Y et al. Umbilical cord blood transplantation after reduced-intensity conditioning for elderly patients with hematologic diseases. Biol Blood Marrow Transplant 2008; 14: 583–590.
Delaney M, Cutler CS, Haspel RL, Yeap BY, McAfee SL, Dey BR et al. High-resolution HLA matching in double-umbilical-cord-blood reduced-intensity transplantation in adults. Transfusion 2009; 49: 995–1002.
Cutler C, Ballen K . Reduced-intensity conditioning and umbilical cord blood transplantation in adults. Bone Marrow Transplant 2009; 44: 667–671.
Ballen KK, Gluckman E, Broxmeyer HE . Umbilical cord blood transplantation: the first 25 years and beyond. Blood 2013; 122: 491–498.
Mattsson J, Ringdén O, Storb R . Graft failure after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2008; 14: 165–170.
Takagi S, Masuoka K, Uchida N, Ishiwata K, Araoka H, Tsuji M et al. High incidence of haemophagocytic syndrome following umbilical cord blood transplantation for adults. Br J Haematol 2009; 147: 543–553.
Carpenter PA, Snyder DS, Flowers ME, Sanders JE, Gooley TA, Martin PJ et al. Prophylactic administration of imatinib after hematopoietic cell transplantation for high-risk Philadelphia chromosome-positive leukemia. Blood 2007; 109: 2791–2793.
Nishiwaki S, Miyamura K, Kato C, Terakura S, Ohashi K, Sakamaki H et al. Impact of post-transplant imatinib administration on Philadelphia chromosome-positive acute lymphoblastic leukaemia. Anticancer Res 2010; 30: 2415–2418.
Ribera JM, Oriol A, González M, Vidriales B, Brunet S, Esteve J et al. Concurrent intensive chemotherapy and imatinib before and after stem cell transplantation in newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Final results of the CSTIBES02 trial. Haematologica 2010; 95: 87–95.
Milhem M, Mahmud N, Lavelle D, Araki H, DeSimone J, Saunthararajah Y et al. Modification of hematopoietic stem cell fate by 5aza 2'deoxycytidine and trichostatin A. Blood 2004; 103: 4102–4110.
Platzbecker U, Wermke M, Radke J, Oelschlaegel U, Seltmann F, Kiani A et al. Azacitidine for treatment of imminent relapse in MDS or AML patients after allogeneic HSCT: results of the RELAZA trial. Leukemia 2012; 26: 381–389.
Moon C, Kim SH, Park KS, Choi BK, Lee HS, Park JB et al. Use of epigenetic modification to induce FOXP3 expression in naïve T cells. Transplant Proc 2009; 41: 1848–1854.
Choi J, Ritchey J, Prior JL, Holt M, Shannon WD, Deych E et al. In vivo administration of hypomethylating agents mitigate graft-versus-host disease without sacrificing graft-versus-leukemia. Blood 2010; 116: 129–139.
Goodyear O, Agathanggelou A, Novitzky-Basso I, Siddique S, McSkeane T, Ryan G et al. Induction of a CD8+ T-cell response to the MAGE cancer testis antigen by combined treatment with azacitidine and sodium valproate in patients with acute myeloid leukemia and myelodysplasia. Blood 2010; 116: 1908–1918.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gotoh, M., Yoshizawa, S., Katagiri, S. et al. A novel reduced-intensity umbilical cord blood transplantation using a recombinant G-CSF combined with high-dose Ara-C for active myeloid malignancies. Bone Marrow Transplant 49, 955–960 (2014). https://doi.org/10.1038/bmt.2014.66
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.66
This article is cited by
-
Fatal Cyberlindnera fabianii fungemia in a patient with mixed phenotype acute leukemia after umbilical cord blood transplantation
International Journal of Hematology (2016)