Abstract
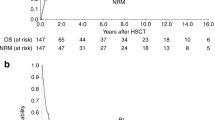
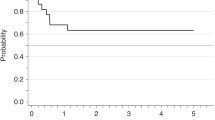
Although there are now fewer allo-SCTs performed for CML, leukemic relapse post transplant remains a persistent problem. To better define clinical and biological parameters determining postrelapse outcome, we studied 59 patients with CML relapsing after HLA-identical sibling allo-SCT between 1993 and 2008. Eighteen (30.5%) were transplanted in advanced phase and 41 (69.5%) in chronic phase. With a median follow-up from relapse of 7.9 years, 5-year post relapse survival (PRS) was 62%. Multivariate analysis found disease status at transplant, time to diagnosis of relapse from transplant and pretransplant tyrosine kinase inhibitor (TKI) use as significant factors associated with PRS. Analysis of BCR-ABL transcript expression in the hematopoietic progenitor compartment was performed in 36 patients (22 relapsed, 8 non-relapsed and 6 TKI alone controls). Patients with BCR-ABL expression in their early hematopoietic stem cell compartment (Lineage−CD34+CD38−CD90+) had worse survival irrespective of the disease status. We conclude that disease status remains the strongest clinical prognostic factor for PRS in CML following allo-SCT. The persistence of BCR-ABL expression in the progenitor cell compartment in some patients after SCT emphasizes the need to target CML-leukemia stem cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barrett AJ, van Rhee F. . Graft-versus-leukaemia. Baillieres Clin Haematol 1997; 10: 337–355.
Ringden O, Pavletic SZ, Anasetti C, Barrett AJ, Wang T, Wang D et al. The graft-versus-leukemia effect using matched unrelated donors is not superior to HLA-identical siblings for hematopoietic stem cell transplantation. Blood 2009; 113: 3110–3118.
Clift RA, Buckner CD, Thomas ED, Doney K, Fefer A, Neiman PE et al. Treatment of chronic granulocytic leukaemia in chronic phase by allogeneic marrow transplantation. Lancet 1982; 2: 621–623.
Goldman JM, Apperley JF, Jones L, Marcus R, Goolden AW, Batchelor R et al. Bone marrow transplantation for patients with chronic myeloid leukemia. N Engl J Med 1986; 314: 202–207.
Speck B, Bortin MM, Champlin R, Goldman JM, Herzig RH, McGlave PB et al. Allogeneic bone-marrow transplantation for chronic myelogenous leukaemia. Lancet 1984; 1: 665–668.
Olavarria E, Ottmann OG, Deininger M, Clark RE, Bandini G, Byrne J et al. Response to imatinib in patients who relapse after allogeneic stem cell transplantation for chronic myeloid leukemia. Leukemia 2003; 17: 1707–1712.
Kantarjian HM, O'Brien S, Cortes JE, Giralt SA, Rios MB, Shan J et al. Imatinib mesylate therapy for relapse after allogeneic stem cell transplantation for chronic myelogenous leukemia. Blood 2002; 100: 1590–1595.
Ullmann AJ, Hess G, Kolbe K, Friedrich-Freksa A, Meyer RG, Gschaidmeier H et al. Current results on the use of imatinib mesylate in patients with relapsed Philadelphia chromosome positive leukemia after allogeneic or syngeneic hematopoietic stem cell transplantation. Keio J Med 2003; 52: 182–188.
Kim YJ, Kim DW, Lee S, Kim YL, Hwang JY, Park YH et al. Cytogenetic clonal evolution alone in CML relapse post-transplantation does not adversely affect response to imatinib mesylate treatment. Bone Marrow Transplant 2004; 33: 237–242.
DeAngelo DJ, Hochberg EP, Alyea EP, Longtine J, Lee S, Galinsky I et al. Extended follow-up of patients treated with imatinib mesylate (gleevec) for chronic myelogenous leukemia relapse after allogeneic transplantation: durable cytogenetic remission and conversion to complete donor chimerism without graft-versus-host disease. Clin Cancer Res 2004; 10: 5065–5071.
Goldman JM, Melo JV. . Chronic myeloid leukemia—advances in biology and new approaches to treatment. N Engl J Med 2003; 349: 1451–1464.
Baccarani M, Saglio G, Goldman J, Hochhaus A, Simonsson B, Appelbaum F et al. Evolving concepts in the management of chronic myeloid leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood 2006; 108: 1809–1820.
Jabbour E, Cortes J, Kantarjian HM, Giralt S, Jones D, Jones R et al. Allogeneic stem cell transplantation for patients with chronic myeloid leukemia and acute lymphocytic leukemia after Bcr-Abl kinase mutation-related imatinib failure. Blood 2006; 108: 1421–1423.
Pavlu J, Szydlo RM, Goldman JM, Apperley JF. . Three decades of transplantation for chronic myeloid leukemia: what have we learned? Blood 2011; 117: 755–763.
Silver RT. . The blast phase of chronic myeloid leukaemia. Best Pract Res Clin Haematol 2009; 22: 387–394.
van Rhee F, Lin F, Cullis JO, Spencer A, Cross NC, Chase A et al. Relapse of chronic myeloid leukemia after allogeneic bone marrow transplant: the case for giving donor leukocyte transfusions before the onset of hematologic relapse. Blood 1994; 83: 3377–3383.
Goldman JM, Majhail NS, Klein JP, Wang Z, Sobocinski KA, Arora M et al. Relapse and late mortality in 5-year survivors of myeloablative allogeneic hematopoietic cell transplantation for chronic myeloid leukemia in first chronic phase. J Clin Oncol 2010; 28: 1888–1895.
Yong AS, Goldman JM. . Relapse of chronic myeloid leukaemia 14 years after allogeneic bone marrow transplantation. Bone Marrow Transplant 1999; 23: 827–828.
Savani BN, Montero A, Kurlander R, Childs R, Hensel N, Barrett AJ. . Imatinib synergizes with donor lymphocyte infusions to achieve rapid molecular remission of CML relapsing after allogeneic stem cell transplantation. Bone Marrow Transplant 2005; 36: 1009–1015.
Goldman JM. . The significance of BCR-ABL transcripts after allogeneic stem cell transplantation for chronic myeloid leukemia. Biol Blood Marrow Transplant 2013; 19: 679–680.
Jamieson CH, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL et al. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N Engl J Med 2004; 351: 657–667.
Solomon SR, Nakamura R, Read EJ, Leitman SF, Carter C, Childs R et al. Cyclosporine is required to prevent severe acute GVHD following T-cell-depleted peripheral blood stem cell transplantation. Bone Marrow Transplant 2003; 31: 783–788.
Agarwal R, Doren S, Hicks B, Dunbar CE. . Long-term culture of chronic myelogenous leukemia marrow cells on stem cell factor-deficient stroma favors benign progenitors. Blood 1995; 85: 1306–1312.
Mensink E, van de Locht A, Schattenberg A, Linders E, Schaap N, Geurts van Kessel A et al. Quantitation of minimal residual disease in Philadelphia chromosome positive chronic myeloid leukaemia patients using real-time quantitative RT-PCR. Br J Haematol 1998; 102: 768–774.
Savani BN, Rezvani K, Mielke S, Montero A, Kurlander R, Carter CS et al. Factors associated with early molecular remission after T cell-depleted allogeneic stem cell transplantation for chronic myelogenous leukemia. Blood 2006; 107: 1688–1695.
Khoury HJ, Kukreja M, Goldman JM, Wang T, Halter J, Arora M et al. Prognostic factors for outcomes in allogeneic transplantation for CML in the imatinib era: a CIBMTR analysis. Bone Marrow Transplant 2012; 47: 810–816.
Ito S, Pophali P, Co W, Koklanaris EK, Superata J, Fahle GA et al. CMV reactivation is associated with a lower incidence of relapse after allo-SCT for CML. Bone Marrow Transplant 2013; 48: 1313–1316.
Yong AS, Szydlo RM, Goldman JM, Apperley JF, Melo JV. . Molecular profiling of CD34+ cells identifies low expression of CD7, along with high expression of proteinase 3 or elastase, as predictors of longer survival in patients with CML. Blood 2006; 107: 205–212.
Yong AS, Keyvanfar K, Eniafe R, Savani BN, Rezvani K, Sloand EM et al. Hematopoietic stem cells and progenitors of chronic myeloid leukemia express leukemia-associated antigens: implications for the graft-versus-leukemia effect and peptide vaccine-based immunotherapy. Leukemia 2008; 22: 1721–1727.
Beillard E, Pallisgaard N, van der Velden VH, Bi W, Dee R, van der Schoot E et al. Evaluation of candidate control genes for diagnosis and residual disease detection in leukemic patients using 'real-time' quantitative reverse-transcriptase polymerase chain reaction (RQ-PCR)—a Europe against cancer program. Leukemia 2003; 17: 2474–2486.
Gabert J, Beillard E, van der Velden VH, Bi W, Grimwade D, Pallisgaard N et al. Standardization and quality control studies of 'real-time' quantitative reverse transcriptase polymerase chain reaction of fusion gene transcripts for residual disease detection in leukemia—a Europe Against Cancer program. Leukemia 2003; 17: 2318–2357.
Kaeda J, O'Shea D, Szydlo RM, Olavarria E, Dazzi F, Marin D et al. Serial measurement of BCR-ABL transcripts in the peripheral blood after allogeneic stem cell transplantation for chronic myeloid leukemia: an attempt to define patients who may not require further therapy. Blood 2006; 107: 4171–4176.
Sobrinho-Simoes M, Wilczek V, Score J, Cross NC, Apperley JF, Melo JV. . In search of the original leukemic clone in chronic myeloid leukemia patients in complete molecular remission after stem cell transplantation or imatinib. Blood 2010; 116: 1329–1335.
Barrett AJ. . Understanding and harnessing the graft-versus-leukaemia effect. Br J Haematol 2008; 142: 877–888.
Boehm A, Walcherberger B, Sperr WR, Wohrer S, Dieckmann K, Rosenmayr A et al. Improved outcome in patients with chronic myelogenous leukemia after allogeneic hematopoietic stem cell transplantation over the past 25 years: a single-center experience. Biol Blood Marrow Transplant 2011; 17: 133–140.
Eisterer W, Jiang X, Christ O, Glimm H, Lee KH, Pang E et al. Different subsets of primary chronic myeloid leukemia stem cells engraft immunodeficient mice and produce a model of the human disease. Leukemia 2005; 19: 435–441.
Corbin AS, Agarwal A, Loriaux M, Cortes J, Deininger MW, Druker BJ. . Human chronic myeloid leukemia stem cells are insensitive to imatinib despite inhibition of BCR-ABL activity. J Clin Invest 2011; 121: 396–409.
Acknowledgements
This paper is dedicated to the memory of John Michael Goldman who inspired us with his insightful observations on the definition and nature of cure of CML. We thank N Hensel, S Miner, M Franco Colon and F Chinian (Hematology Branch, NHLBI, NIH, USA) for their technical support. We also thank all patients who participated in this study. This research was supported by the Intramural Research Program of the National Institutes of Health at the National Heart, Lung, and Blood Institute.
Author Contributions
Study concept and design: NAJ, SI, AJB and ASY; in vitro experiment and data collection: KK and ASY; analysis and interpretation of data: NAJ, SI, RK, MB, PM, AJB and ASY; drafting of the manuscript: NAJ, SI, RK, MB, CH, PM, AJB and ASY; statistical analysis: NAJ, SI and XT; subject enrollment and clinical data collection: MB, KL, BNS, VM, KR, RQL, AS, EK, JS, AJB and ASY; obtaining BM samples from controls: VM; and obtainment of funding and study supervision: AJB.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Bone Marrow Transplantation website
Rights and permissions
About this article
Cite this article
Jain, N., Ito, S., Tian, X. et al. Clinical and biological predictors of outcome following relapse of CML post-allo-SCT. Bone Marrow Transplant 50, 189–196 (2015). https://doi.org/10.1038/bmt.2014.249
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.249
This article is cited by
-
Allogeneic stem cell transplantation for chronic myeloid leukemia in the TKI era: population-based data from the Swedish CML registry
Bone Marrow Transplantation (2019)
-
Relapse patterns and treatment strategies in patients receiving allogeneic hematopoietic stem cell transplantation for myeloid malignancies
Annals of Hematology (2019)
-
VEGFR2-targeted fusion antibody improved NK cell-mediated immunosurveillance against K562 cells
Immunologic Research (2016)