Abstract
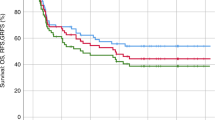
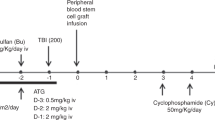
Although the feasibility of using HLA-mismatched unrelated donors as an alternate graft source for haematopoietic SCT (HSCT) has been shown, little is known about the safety of HLA-mismatched DLI for the treatment of relapse. We examined the outcome of 58 consecutive leukaemia patients who received escalating-dose DLI for treatment of relapse after alemtuzumab-conditioned myeloablative unrelated donor HSCT at our institution. High-resolution HLA typing on stored DNA samples revealed mismatches in 28/58 patients who were considered HLA-matched at the time of transplantation. Following DLI from HLA-matched (10/10) (n=30) or -mismatched (7-9/10) (n=28) unrelated donors, we found no significant difference in the incidence of acute GVHD (17.2% versus 23.1%, P=0.59), probability of remission at 3 years (62.1% versus 63.9%, P=0.89) or 5-year OS (89.8% versus 77.7%, P=0.22). We conclude that escalating-dose DLI can be safely given to HLA-mismatched recipients following T-depleted myeloablative HSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Pavlu J, Szydlo RM, Goldman JM, Apperley J . Three decades of transplantation for chronic myeloid leukaemia: what have we learned? Blood 2011; 117: 755–763.
Baldomero H, Gratwohl M, Gratwohl A, Tichelli A, Niederwieser D, Madrigal A et al. The EBMT activity survey 2009: trends over the past 5 years. Bone Marrow Transplant 2011; 46: 485–501.
Craddock CF . Full-intensity and reduced intensity allogeneic stem cell transplantation in AML. Bone marrow Transplant 2008; 31: 415–423.
Ballen KK, Korelth J, Yi-Bin Chen, Dey BR, Spitzer TR . Selection of optimal alternative graft source: mismatched unrelated donor, umbilical cord blood, or haploidentical transplantation. Blood 2012; 119: 1972–1980.
Oudshoorn M, van Walraven SM, Bakker JN, Lie JL, V D Zanden HG, Heemskerk MB et al. Hematopoietic stem cell donor selection: the Europdonor experience. Hum Immunol 2006; 67: 405–412.
Mead AJ, Thomas KJ, Morris EC, Mohamedbhai S, Denovan S, Orti G et al. HLA-mismatched unrelated donors are a viable alternative graft source for allogeneic transplantion following alemtuzumab-based reduced-intensity conditioning. Blood 2010; 115: 5147–5153.
Yagasaki H, Kojima S, Yabe H, Kato K, Kigasawa H, Sakamaki H et al. Acceptable HLA-matching in unrelated donor bone marrow transplantation for patients with acquired severe aplastic anaemia. Blood 2011; 118: 3186–3190.
Arora M, Weisdorf DJ, Spellman SR, Haagenson MD, Klein JP, Hurley CK et al. HLA-identical sibling compared with 8/8 matched and mismatched unrelated donor bone marrow transplant for chronic phase chronic myeloid leukemia. J Clin Oncol 2009; 27: 1644–1652.
Yeung DT, Parker WT, Branford S . Molecular methods in diagnosis and monitoring of haematological malignancies. Pathology 2011; 43: 566–579.
Bene MC, Kaeda JS . How and why minimal residual disease studies are necessary in leukaemia: a review from WP10 and WP12 of the European Leukaemia Net. Haematologica 2009; 94: 1135–1150.
Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W et al. Graft-versus-leukaemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 1995; 86: 2041–2050.
Dazzi F, Szydlo RM, Cross NC, Craddock C, Kaeda J, Kanfer E et al. Durability of responses following donor lymphocyte infusions for patients who relapse after allogeneic stem cell transplantation for chronic myeloid leukaemia. Blood 2000; 96: 2712–2716.
Roos-Weil D, Nguyen S, Uzunov M, Bories D, Chapiro E, Nguyen-Khac F et al. Therapy-related myelodysplastic syndrome after allogeneic BMT: successful treatment by donor lymphocyte infusions. Bone Marrow Transplant 2010; 45: 1471–1473.
Loren AW, Porter DL . Donor leukocyte infusions for the treatment of relapsed acute leukemia after allogeneic stem cell transplantation. Bone Marrow Transplant 2008; 41: 483–493.
Toor A, Rodriguez T, Bauml M, Mathews H, Shanti S, Senitzer D et al. Feasibility of conditioning with thymoglobulin and reduced intensity TBI to reduce acute GVHD in recipients of allogeneic SCT. Bone Marrow Transplant 2008; 42: 723–731.
Kim HJ, Min WS, Cho BS . Successful prevention of acute graft-versus-host disease using low-dose antithymocyte globulin after mismatched, unrelated, hematopoietic stem cell transplantation for acute myelogenous leukemia. Biol Blood Marrow Transplant 2009; 15: 704–717.
Or R, Hadar E, Bitan M, Resnick IB, Aker M, Ackerstein A et al. Safety and efficacy of donor lymphocyte infusions following mismatched stem cell transplantation. Biol Blood Marrow Transplant 2006; 12: 1295–1301.
Prepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus conference in acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 1974; 18: 295–303.
Hanahan D, Weinberd R . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–676.
Simula MP, Marktel S, Fozza C, Kaeda J, Szydlo RM, Nadal E et al. Response to donor lymphocyte infusions for chronic myeloid leukemia is dose-dependent: the importance of escalating the cell dose to maximize therapeutic efficacy. Leukemia 2007; 21: 943–948.
Rizzieri DA, Dev P, Long GD, Gasparetto C, Sullivan KM, Horwitz M et al. Response and toxicity of donor lymphocyte infusions following T-cell depleted non-myeloablative allogeneic hematopoietic SCT from 3-6/6 HLA matched donors. Bone Marrow Transplant 2009; 43: 327–333.
Huang XJ, Liu DH, Liu KY, Xu LP, Chen YH, Wang Y et al. Modified donor lymphocyte infusion after HLA-mismatched/haploidentical T cell-replete hematopoietic stem cell transplantation for prophylaxis of relapse of leukemia in patients with advanced leukemia. J Clin Immunol 2008; 28: 276–283.
Basak GW, de Wreede LC, van Biezen A, Wiktor-Jedrzejczak W, Halaburda K, Schmid C et al. Donor lymphocyte infusions for the treatment of chronic myeloid leukemia relapse following peripheral blood or bone marrow stem cell transplantation. Bone Marrow Transplant 2013; 48: 837–842.
Peggs KS, Thomson K, Hart DP, Geary J, Morris EC, Yong K et al. Dose-escalated donor lymphocyte infusions following reduced intensity transplantation: toxicity, chimerism, and disease responses. Blood 2004; 103: 1548–1556.
Olavarria E, Siddique S, Griffiths MJ, Avery S, Byrne JL, Piper KP et al. Posttransplantation imatinib as a strategy to postpone the requirement for immunotherapy in patients undergoing reduced-intensity allografts for chronic myeloid leukemia. Blood 2007; 110: 4614–4617.
Schroeder T, Czibere A, Platzbecker U, Bug G, Uharek L, Luft T et al. Azacitidine and donor lymphocyte infusions as first salvage therapy for relapse of AML or MDS allogeneic stem cell transplantation. Leukemia 2013; 27: 1229–1235.
Acknowledgements
This work was supported by Leuka (registered charity number 286231) and the National Institute for Health Research (NIHR) Biomedical Research Centre.
Author’s contribution
AI performed data collection, analysed the data and wrote the paper; RB performed research and analysed the data; RS provided reagents and analysed the data; GD analysed the data and contributed to writing the paper; LF and EB provided sample material and contributed to the paper; DM and SM analysed the data and contributed to writing the paper, D, MM, EK, JP, AR, IR, JG, JA and FD provided patient care, and contributed to writing of the paper; RS analysed the data and wrote the paper; KR designed and supervised the research, analysed the data and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr John Goldman is Editor in Chief of Bone Marrow Transplantation. The remaining authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Innes, A., Beattie, R., Sergeant, R. et al. Escalating-dose HLA-mismatched DLI is safe for the treatment of leukaemia relapse following alemtuzumab-based myeloablative allo-SCT. Bone Marrow Transplant 48, 1324–1328 (2013). https://doi.org/10.1038/bmt.2013.69
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2013.69
Keywords
This article is cited by
-
Donor lymphocyte infusions for B-cell malignancies relapse after T-cell replete allogeneic hematopoietic cell transplantation
Bone Marrow Transplantation (2019)
-
New strategies of DLI in the management of relapse of hematological malignancies after allogeneic hematopoietic SCT
Bone Marrow Transplantation (2016)
-
Allogeneic transplantation for CML in the TKI era: striking the right balance
Nature Reviews Clinical Oncology (2016)