Abstract
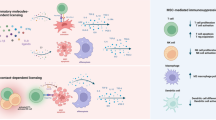
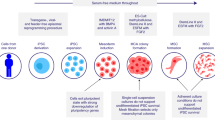
Mesenchymal stromal cells (MSCs) have immunomodulatory effects and are increasingly being used for the treatment of acute and chronic GVHD. Although they seem immuno-privileged, they induce alloresponses, but the risk of immunization is poorly characterized. After infusion, they first reach the lungs, liver and spleen, and are then difficult to trace. Several mechanisms are involved in stromal cells suppressing alloreactivity, such as induction of regulatory T cells, but whether or not this will also affect leukemic relapse or increase infections is not known. Although several encouraging pilot studies have been published, there have been few prospective randomized trials. There may be a bias in the literature, as negative results are seldom published, and there have been few comparative studies with other immunosuppressive regimens. Most animal models have failed to show any effect on GVHD. Several questions remain to be answered for optimization of stromal cell therapy. Which source is optimal–BM, fat, cord or decidua? Can stromal cells be replaced by exosomes, which culture conditions are most appropriate and at what passage and how frequently should cells be administered? More research is required to move stromal cell therapy forward to become an established treatment for acute and chronic GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mendez-Ferrer S, Michurina TV, Ferraro F, Mazloom AR, Macarthur BD, Lira SA et al. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 2010; 466: 829–834.
Li MQ, Hou XF, Shao J, Tang CL, Li DJ . The DSCs-expressed CD82 controls the invasiveness of trophoblast cells via integrinbeta1/MAPK/MAPK3/1 signaling pathway in human first-trimester pregnancy. Biol Reprod 2010; 82: 968–979.
Nancy P, Tagliani E, Tay CS, Asp P, Levy DE, Erlebacher A . Chemokine gene silencing in decidual stromal cells limits T cell access to the maternal-fetal interface. Science 2012; 336: 1317–1321.
Oreshkova T, Dimitrov R, Mourdjeva M . A cross-talk of decidual stromal cells, trophoblast, and immune cells: a prerequisite for the success of pregnancy. Am J Reproduct Immunol 2012; 68: 366–373.
Le Blanc K, Tammik L, Sundberg B, Haynesworth SE, Ringden O . Mesenchymal stem cells inhibit and stimulate mixed lymphocyte cultures and mitogenic responses independently of the major histocompatibility complex. Scand J Immunol 2003; 57: 11–20.
Karlsson H, Erkers T, Nava S, Ruhm S, Westgren M, Ringden O . Stromal cells from term fetal membrane are highly suppressive in allogeneic settings in vitro. Clin Exp Immunol 2012; 167: 543–555.
Jones S, Horwood N, Cope A, Dazzi F . The antiproliferative effect of mesenchymal stem cells is a fundamental property shared by all stromal cells. J Immunol 2007; 179: 2824–2831.
Le Blanc K, Rasmusson I, Sundberg B, Gotherstrom C, Hassan M, Uzunel M et al. Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 2004; 363: 1439–1441.
Ringden O, Uzunel M, Rasmusson I, Remberger M, Sundberg B, Lonnies H et al. Mesenchymal stem cells for treatment of therapy-resistant graft-versus-host disease. Transplantation 2006; 81: 1390–1397.
Tse WT, Pendleton JD, Beyer WM, Egalka MC, Guinan EC . Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 2003; 75: 389–397.
Le Blanc K, Tammik C, Rosendahl K, Zetterberg E, Ringden O . HLA expression and immunologic properties of differentiated and undifferentiated mesenchymal stem cells. Exp Hematol 2003; 31: 890–896.
Beggs KJ, Lyubimov A, Borneman JN, Bartholomew A, Moseley A, Dodds R et al. Immunologic consequences of multiple, high-dose administration of allogeneic mesenchymal stem cells to baboons. Cell Transplant 2006; 15: 711–721.
Eliopoulos N, Stagg J, Lejeune L, Pommey S, Galipeau J . Allogeneic marrow stromal cells are immune rejected by MHC class I- and class II-mismatched recipient mice. Blood 2005; 106: 4057–4065.
Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE . Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a nonmyeloablative setting. Blood 2006; 108: 2114–2120.
Sundin M, Ringden O, Sundberg B, Nava S, Gotherstrom C, Le Blanc K . No alloantibodies against mesenchymal stromal cells, but presence of anti-fetal calf serum antibodies, after transplantation in allogeneic hematopoietic stem cell recipients. Haematologica 2007; 92: 1208–1215.
Gholamrezanezhad A, Mirpour S, Bagheri M, Mohamadnejad M, Alimoghaddam K, Abdolahzadeh L et al. In vivo tracking of 111In-oxine labeled mesenchymal stem cells following infusion in patients with advanced cirrhosis. Nuclear Med Biol 2011; 38: 961–967.
von Bahr L, Batsis I, Moll G, Hagg M, Szakos A, Sundberg B et al. Analysis of tissues following mesenchymal stromal cell therapy in humans indicates limited long-term engraftment and no ectopic tissue formation. Stem Cells 2012; 30: 1575–1578.
Moll G, Jitschin R, von Bahr L, Rasmusson-Duprez I, Sundberg B, Lonnies L et al. Mesenchymal stromal cells engage complement and complement receptor bearing innate effector cells to modulate immune responses. PLoS One 2011; 6: e21703.
Li Y, Lin F . Mesenchymal stem cells are injured by complement after their contact with serum. Blood 2012; 120: 3436–3443.
Moll G, Rasmusson-Duprez I, von Bahr L, Connolly-Andersen AM, Elgue G, Funke L et al. Are therapeutic human mesenchymal stromal cells compatible with human blood? Stem Cells 2012; 30: 1565–1574.
Uhlin M, Sairafi D, Berglund S, Thunberg S, Gertow J, Ringden O et al. Mesenchymal stem cells inhibit thymic reconstitution after allogeneic cord blood transplantation. Stem Cells Develop 2012; 21: 1409–1417.
Uhlin M, Wikell H, Sundin M, Blennow O, Maeurer M, Ringden O et al. Risk factors for Epstein Barr virus related post-transplant lymphoproliferative disease after allogeneic hematopoietic stem cell transplantation. Haematologica, (e-pub ahead of print 20 September 2013; doi:10.3324/haematol.2013.087338).
Remberger M, Ringden O . Treatment of severe acute graft-versus-host disease with mesenchymal stromal cells: a comparison with non-MSC treated patients. Int J Hematol 2012; 96: 822–824.
Ning H, Yang F, Jiang M, Hu L, Feng K, Zhang J et al. The correlation between cotransplantation of mesenchymal stem cells and higher recurrence rate in hematologic malignancy patients: outcome of a pilot clinical study. Leukemia 2008; 22: 593–599.
Gupta D, Treon SP, Shima Y, Hideshima T, Podar K, Tai YT et al. Adherence of multiple myeloma cells to bone marrow stromal cells upregulates vascular endothelial growth factor secretion: therapeutic applications. Leukemia 2001; 15: 1950–1961.
Roccaro AM, Sacco A, Maiso P, Azab AK, Tai YT, Reagan M et al. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J Clin Invest 2013; 123: 1542–1555.
Martin PJ, Uberti J, Soiffer R, Klingemann H, Waller EK, Daly AS et al. Prochymal improves response rates in patients with steroid-refractory acute graft versus host disease (SR-GVHD) involving the liver and gut: results of a randomized placebo-controlled multicenter phase III trial in GVHD. Biol Blood Marrow Transplant 2010; 16 (suppl 2): S169–S170.
Badillo AT, Peranteau WH, Heaton TE, Quinn C, Flake AW . Murine bone marrow derived stromal progenitor cells fail to prevent or treat acute graft-versus-host disease. Br J Haematol 2008; 141: 224–234.
Jeon MS, Lim HJ, Yi TG, Im MW, Yoo HS, Choi JH et al. Xenoreactivity of human clonal mesenchymal stem cells in a major histocompatibility complex-matched allogeneic graft-versus-host disease mouse model. Cellular Immunol 2010; 261: 57–63.
Mielcarek M, Storb R, Georges GE, Golubev L, Nikitine A, Hwang B et al. Mesenchymal stromal cells fail to prevent acute graft-versus-host disease and graft rejection after dog leukocyte antigen-haploidentical bone marrow transplantation. Biol Blood Marrow Transplant 2011; 17: 214–225.
Sadeghi B, Ringden O . Mesenchymal stem cells for graft-versus-host disease in experimental animal models. In: Gross G, Häupl T (eds). Stem Cell-Dependent Therapies: Mesenchymal stem cells in chronic inflammatory disorders. De Gruyter: Berlin, pp 125–141 2013.
Sudres M, Norol F, Trenado A, Gregoire S, Charlotte F, Levacher B et al. Bone marrow mesenchymal stem cells suppress lymphocyte proliferation in vitro but fail to prevent graft-versus-host disease in mice. J Immunol 2006; 176: 7761–7767.
Paulson K, Saeed M, Mills J, Cuvelier GD, Kumar R, Raymond C et al. Publication bias is present in blood and marrow transplantation: an analysis of abstracts at an international meeting. Blood 2011; 118: 6698–6701.
Murray IR, West CC, Hardy WR, James AW, Park TS, Nguyen A et al. Natural history of mesenchymal stem cells, from vessel walls to culture vessels. Cell Mol Life Sci, (e-pub ahead of print 25 October 2013).
Tang W, Zeve D, Suh JM, Bosnakovski D, Kyba M, Hammer RE et al. White fat progenitor cells reside in the adipose vasculature. Science 2008; 322: 583–586.
Crisan M, Yap S, Casteilla L, Chen CW, Corselli M, Park TS et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008; 3: 301–313.
Warrier S, Haridas N, Bhonde R . Inherent propensity of amnion-derived mesenchymal stem cells towards endothelial lineage: vascularization from an avascular tissue. Placenta 2012; 33: 850–858.
Cossu G, Bianco P . Mesoangioblasts—vascular progenitors for extravascular mesodermal tissues. Curr Opin Genet Develop 2003; 13: 537–542.
Galli D, Innocenzi A, Staszewsky L, Zanetta L, Sampaolesi M, Bai A et al. Mesoangioblasts, vessel-associated multipotent stem cells, repair the infarcted heart by multiple cellular mechanisms: a comparison with bone marrow progenitors, fibroblasts, and endothelial cells. Arteriosclerosis, Thrombosis Vascular Biol 2005; 25: 692–697.
Ochs K, Sahm F, Opitz CA, Lanz TV, Oezen I, Couraud PO et al. Immature mesenchymal stem cell-like pericytes as mediators of immunosuppression in human malignant glioma. J Neuroimmunol 2013; 265: 106–116.
Keating A . Mesenchymal stromal cells: new directions. Cell Stem Cell 2012; 10: 709–716.
Le Blanc K, Mougiakakos D . Multipotent mesenchymal stromal cells and the innate immune system. Nat Rev Immunol 2012; 12: 383–396.
Nauta AJ, Fibbe WE . Immunomodulatory properties of mesenchymal stromal cells. Blood 2007; 110: 3499–3506.
Tolar J, Le Blanc K, Keating A, Blazar BR . Concise review: hitting the right spot with mesenchymal stromal cells. Stem Cells 2010; 28: 1446–1455.
Uccelli A, Moretta L, Pistoia V . Mesenchymal stem cells in health and disease. Nat Rev Immunol 2008; 8: 726–736.
Bartholomew A, Sturgeon C, Siatskas M, Ferrer K, McIntosh K, Patil S et al. Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 2002; 30: 42–48.
Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002; 99: 3838–3843.
von Bahr L, Sundberg B, Lonnies L, Sander B, Karbach H, Hagglund H et al. Long-term complications, immunologic effects, and role of passage for outcome in mesenchymal stromal cell therapy. Biol Blood Marrow Transplant 2012; 18: 557–564.
Selmani Z, Naji A, Gaiffe E, Obert L, Tiberghien P, Rouas-Freiss N et al. HLA-G is a crucial immunosuppressive molecule secreted by adult human mesenchymal stem cells. Transplantation 2009; 87 (suppl 9): S62–S66.
Aggarwal S, Pittenger MF . Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005; 105: 1815–1822.
Gieseke F, Bohringer J, Bussolari R, Dominici M, Handgretinger R, Muller I . Human multipotent mesenchymal stromal cells use galectin-1 to inhibit immune effector cells. Blood 2010; 116: 3770–3779.
Erkers T, Nava S, Yosef J, Ringden O, Kaipe H . Decidual stromal cells promote regulatory T cells and suppress alloreactivity in a cell contact-dependent manner. Stem Cells Dev 2013; 22: 2596–2605.
Krampera M, Cosmi L, Angeli R, Pasini A, Liotta F, Andreini A et al. Role for interferon-gamma in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells 2006; 24: 386–398.
Ren G, Zhang L, Zhao X, Xu G, Zhang Y, Roberts AI et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell 2008; 2: 141–150.
Opitz CA, Litzenburger UM, Lutz C, Lanz TV, Tritschler I, Koppel A et al. Toll-like receptor engagement enhances the immunosuppressive properties of human bone marrow-derived mesenchymal stem cells by inducing indoleamine-2,3-dioxygenase-1 via interferon-beta and protein kinase R. Stem Cells 2009; 27: 909–919.
Meisel R, Zibert A, Laryea M, Gobel U, Daubener W, Dilloo D . Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood 2004; 103: 4619–4621.
Ryan JM, Barry F, Murphy JM, Mahon BP . Interferon-gamma does not break, but promotes the immunosuppressive capacity of adult human mesenchymal stem cells. Clin Exp Immunol 2007; 149: 353–363.
Waterman RS, Tomchuck SL, Henkle SL, Betancourt AM . A new mesenchymal stem cell (MSC) paradigm: polarization into a pro-inflammatory MSC1 or an Immunosuppressive MSC2 phenotype. PLoS One 2010; 5: e10088.
Ge W, Jiang J, Arp J, Liu W, Garcia B, Wang H . Regulatory T-cell generation and kidney allograft tolerance induced by mesenchymal stem cells associated with indoleamine 2,3-dioxygenase expression. Transplantation 2010; 90: 1312–1320.
Tatara R, Ozaki K, Kikuchi Y, Hatanaka K, Oh I, Meguro A et al. Mesenchymal stromal cells inhibit Th17 but not regulatory T-cell differentiation. Cytotherapy 2011; 13: 686–694.
Francois M, Romieu-Mourez R, Li M, Galipeau J . Human MSC suppression correlates with cytokine induction of indoleamine 2,3-dioxygenase and bystander M2 macrophage differentiation. Mol Therapy 2012; 20: 187–195.
Augello A, Tasso R, Negrini SM, Amateis A, Indiveri F, Cancedda R et al. Bone marrow mesenchymal progenitor cells inhibit lymphocyte proliferation by activation of the programmed death 1 pathway. Eur J Immunol 2005; 35: 1482–1490.
Akiyama K, Chen C, Wang D, Xu X, Qu C, Yamaza T et al. Mesenchymal-stem-cell-induced immunoregulation involves FAS-ligand-/FAS-mediated T cell apoptosis. Cell Stem Cell 2012; 10: 544–555.
Ren G, Zhao X, Zhang L, Zhang J, L'Huillier A, Ling W et al. Inflammatory cytokine-induced intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 in mesenchymal stem cells are critical for immunosuppression. J Immunol 2010; 184: 2321–2328.
Saldanha-Araujo F, Ferreira FI, Palma PV, Araujo AG, Queiroz RH, Covas DT et al. Mesenchymal stromal cells up-regulate CD39 and increase adenosine production to suppress activated T-lymphocytes. Stem cell Res 2011; 7: 66–74.
Ren G, Su J, Zhang L, Zhao X, Ling W, L'Huillie A et al. Species variation in the mechanisms of mesenchymal stem cell-mediated immunosuppression. Stem Cells 2009; 27: 1954–1962.
Min CK, Kim BG, Park G, Cho B, Oh IH . IL-10-transduced bone marrow mesenchymal stem cells can attenuate the severity of acute graft-versus-host disease after experimental allogeneic stem cell transplantation. Bone Marrow Transplant 2007; 39: 637–645.
Thery C, Ostrowski M, Segura E . Membrane vesicles as conveyors of immune responses. Nat Rev Immunol 2009; 9: 581–593.
Thery C, Ostrowski M, Segura E . Membrane vesicles as conveyors of immune responses. Nat Rev Immunol 2009; 9: 581–593.
Bruno S, Grange C, Collino F, Deregibus MC, Cantaluppi V, Biancone L et al. Microvesicles derived from mesenchymal stem cells enhance survival in a lethal model of acute kidney injury. PLoS One 2012; 7: e33115.
Lai RC, Arslan F, Lee MM, Sze NS, Choo A, Chen TS et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res 2010; 4: 214–222.
Lee C, Mitsialis SA, Aslam M, Vitali SH, Vergadi E, Konstantinou G et al. Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia-induced pulmonary hypertension. Circulation 2012; 126: 2601–2611.
Gatti S, Bruno S, Deregibus MC, Sordi A, Cantaluppi V, Tetta C et al. Microvesicles derived from human adult mesenchymal stem cells protect against ischaemia-reperfusion-induced acute and chronic kidney injury. Nephrol Dial Transplant 2011; 26: 1474–1483.
Andre F, Chaput N, Schartz NEC, Flament C, Aubert N, Bernard J et al. Exosomes as potent cell-free peptide-based vaccine. I. Dendritic cell-derived exosomes transfer functional MHC class I/peptide complexes to dendritic cells. J Immunol 2004; 172: 2126–2136.
Bruno S, Collino F, Deregibus MC, Grange C, Tetta C, Camussi G . Microvesicles derived from human bone marrow mesenchymal stem cells inhibit tumor growth. Stem Cells Develop 2013; 22: 758–771.
Kordelas L, Ludwig S, Radke P, Beelen D, Giebel B . Successful treatment of therapy-refractory acute graft-versus-host disease with mesenchymal stem cell-derived exosomes. Bone Marrow Transplant 2013; 48: s176–s177.
EL Andaloussi S, Mager I, Breakefield XO, Wood MJ . Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov 2013; 12: 347–357.
Lai RC, Yeo RW, Tan KH, Lim SK . Exosomes for drug delivery – a novel application for the mesenchymal stem cell. Biotechnol Adv 2013; 31: 543–551.
Deeg HJ . How I treat refractory acute GVHD. Blood 2007; 109: 4119–4126.
Hahn T, McCarthy PL Jr., Zhang MJ, Wang D, Arora M, Frangoul H et al. Risk factors for acute graft-versus-host disease after human leukocyte antigen-identical sibling transplants for adults with leukemia. J Clin Oncol 2008; 26: 5728–5734.
Ringden O, Nilsson B . Death by graft-versus-host disease associated with HLA mismatch, high recipient age, low marrow cell dose, and splenectomy. Transplantation 1985; 40: 39–44.
Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 2008; 371: 1579–1586.
Ringden O . Mesenchymal stem cells for treatment and prevention of graft-versus-host disease and graft failure after hematopoietic stem cell transplantation and future challenges. In: Chase LG, Vemuri MC (eds). Mesenchymal Stem Cell Therapy. Humana Press: Springer Verlag, pp 173–206 2013.
Perez-Simon JA, Lopez-Villar O, Andreu EJ, Rifon J, Muntion S, Campelo MD et al. Mesenchymal stem cells expanded in vitro with human serum for the treatment of acute and chronic graft-versus-host disease: results of a phase I/II clinical trial. Haematologica 2011; 96: 1072–1076.
von Bonin M, Stolzel F, Goedecke A, Richter K, Wuschek N, Holig K et al. Treatment of refractory acute GVHD with third-party MSC expanded in platelet lysate-containing medium. Bone Marrow Transplant 2009; 43: 245–251.
Kebriaei P, Isola L, Bahceci E, Holland K, Rowley S, McGuirk J et al. Adult human mesenchymal stem cells added to corticosteroid therapy for the treatment of acute graft-versus-host disease. Biol Blood Marrow Transplant 2009; 15: 804–811.
Lucchini G, Introna M, Dander E, Rovelli A, Balduzzi A, Bonanomi S et al. Platelet-lysate-expanded mesenchymal stromal cells as a salvage therapy for severe resistant graft-versus-host disease in a pediatric population. Biol Blood Marrow Transplant 2010; 16: 1293–1301.
Fang B, Song Y, Liao L, Zhang Y, Zhao RC . Favorable response to human adipose tissue-derived mesenchymal stem cells in steroid-refractory acute graft-versus-host disease. Transplant Proc 2007; 39: 3358–3362.
Wu KH, Chan CK, Tsai C, Chang YH, Sieber M, Chiu TH et al. Effective treatment of severe steroid-resistant acute graft-versus-host disease with umbilical cord-derived mesenchymal stem cells. Transplantation 2011; 91: 1412–1416.
Ringden O, Erkers T, Nava S, Uzunel M, Iwarsson E, Conrad R et al. Fetal membrane cells for treatment of steroid-refractory acute graft-versus-host disease. Stem Cells 2013; 31: 592–601.
Di Ianni M, Falzetti F, Carotti A, Terenzi A, Castellino F, Bonifacio E et al. Tregs prevent GVHD and promote immune reconstitution in HLA-haploidentical transplantation. Blood 2011; 117: 3921–3928.
Highfill SL, Rodriguez PC, Zhou Q, Goetz CA, Koehn BH, Veenstra R et al. Bone marrow myeloid-derived suppressor cells (MDSCs) inhibit graft-versus-host disease (GVHD) via an arginase-1-dependent mechanism that is up-regulated by interleukin-13. Blood 2010; 116: 5738–5747.
Zheng J, Liu Y, Liu M, Xiang Z, Lam KT, Lewis DB et al. Human CD8+ regulatory T cells inhibit GVHD and preserve general immunity in humanized mice. Sci Transl Med 2013; 5: 168ra9.
Weng JY, Du X, Geng SX, Peng YW, Wang Z, Lu ZS et al. Mesenchymal stem cell as salvage treatment for refractory chronic GVHD. Bone Marrow Transplant 2010; 45: 1732–1740.
Lazarus HM, Koc ON, Devine SM, Curtin P, Maziarz RT, Holland HK et al. Cotransplantation of HLA-identical sibling culture-expanded mesenchymal stem cells and hematopoietic stem cells in hematologic malignancy patients. Biol Blood Marrow Transplant 2005; 11: 389–398.
Bernardo ME, Ball LM, Cometa AM, Roelofs H, Zecca M, Avanzini MA et al. Co-infusion of ex vivo-expanded, parental MSCs prevents life-threatening acute GVHD, but does not reduce the risk of graft failure in pediatric patients undergoing allogeneic umbilical cord blood transplantation. Bone Marrow Transplant 2011; 46: 200–207.
Ball LM, Bernardo ME, Roelofs H, Lankester A, Cometa A, Egeler RM et al. Cotransplantation of ex vivo expanded mesenchymal stem cells accelerates lymphocyte recovery and may reduce the risk of graft failure in haploidentical hematopoietic stem-cell transplantation. Blood 2007; 110: 2764–2767.
Liu K, Chen Y, Zeng Y, Xu L, Liu D, Chen H et al. Coinfusion of mesenchymal stromal cells facilitates platelet recovery without increasing leukemia recurrence in haploidentical hematopoietic stem cell transplantation: a randomized, controlled clinical study. Stem Cells Dev 2011; 20: 1679–1685.
Kuzmina LA, Petinati NA, Parovichnikova EN, Lubimova LS, Gribanova EO, Gaponova TV et al. Multipotent mesenchymal stromal cells for the prophylaxis of acute graft-versus-host disease-a phase II study. Stem Cells Int 2012; 2012: 968213.
Ball LM, Bernardo ME, Roelofs H, van Tol MJ, Contoli B, Zwaginga JJ et al. Multiple infusions of mesenchymal stromal cells induce sustained remission in children with steroid-refractory, grade III-IV acute graft-versus-host disease. Br J Haematol 2013; 163: 501–509.
Ringden O, Keating A . Mesenchymal stromal cells as treatment for chronic GVHD. Bone Marrow Transplant 2011; 46: 163–164.
Ringden O, Leblanc K . Pooled MSCs for treatment of severe hemorrhage. Bone Marrow Transplant 2011; 46: 1158–1160.
Ringden O, Uzunel M, Sundberg B, Lonnies L, Nava S, Gustafsson J et al. Tissue repair using allogeneic mesenchymal stem cells for hemorrhagic cystitis, pneumomediastinum and perforated colon. Leukemia 2007; 21: 2271–2276.
Jung JW, Kwon M, Choi JC, Shin JW, Park IW, Choi BW et al. Familial occurrence of pulmonary embolism after intravenous, adipose tissue-derived stem cell therapy. Yonsei Med J 2013; 54: 1293–1296.
Chauhan D, Uchiyama H, Akbarali Y, Urashima M, Yamamoto K, Libermann TA et al. Multiple myeloma cell adhesion-induced interleukin-6 expression in bone marrow stromal cells involves activation of NF-kappa B. Blood 1996; 87: 1104–1112.
Miura M, Miura Y, Padilla-Nash HM, Molinolo AA, Fu B, Patel V et al. Accumulated chromosomal instability in murine bone marrow mesenchymal stem cells leads to malignant transformation. Stem Cells 2006; 24: 1095–1103.
Tarte K, Gaillard J, Lataillade JJ, Fouillard L, Becker M, Mossafa H et al. Clinical-grade production of human mesenchymal stromal cells: occurrence of aneuploidy without transformation. Blood 2010; 115: 1549–1553.
Tolar J, Nauta AJ, Osborn MJ, Panoskaltsis Mortari A, McElmurry RT, Bell S et al. Sarcoma derived from cultured mesenchymal stem cells. Stem Cells 2007; 25: 371–9.
Paliogianni F, Boumpas DT . Prostaglandin E2 inhibits the nuclear transcription of the human interleukin 2, but not the Il-4, gene in human T cells by targeting transcription factors AP-1 and NF-AT. Cell Immunol 1996; 171: 95–101.
Sato K, Ozaki K, Oh I, Meguro A, Hatanaka K, Nagai T et al. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood 2007; 109: 228–234.
Zhang HC, Liu XB, Huang S, Bi XY, Wang HX, Xie LX et al. Microvesicles derived from human umbilical cord mesenchymal stem cells stimulated by hypoxia promote angiogenesis both in vitro and in vivo. Stem Cells Dev 2012; 21: 3289–3297.
Bruno S, Grange C, Collino F, Deregibus MC, Cantaluppi V, Biancone L et al. Microvesicles derived from mesenchymal stem cells enhance survival in a lethal model of acute kidney injury. PLoS One 2012; 7: e33115.
Ciceri F, Bonini C, Stanghellini MT, Bondanza A, Traversari C, Salomoni M et al. Infusion of suicide-gene-engineered donor lymphocytes after family haploidentical haemopoietic stem-cell transplantation for leukaemia (the TK007 trial): a non-randomised phase I-II study. Lancet Oncol 2009; 10: 489–500.
Acknowledgements
Helen Kaipe: The Swedish Research Council (K2012-99X-22013-01-3), the Cancer Society in Stockholm (121092), the Swedish Society of Medicine, the Childrens’ Cancer Foundation (PR2013-0020), Clas Groschinsky Foundation and Karolinska Institutet. Olle Ringdén: The Swedish Cancer Society (CAN2011/419), the Swedish Research Council (K2011-64X-05971-31-6), the Childrens’ Cancer Foundation (PROJ12/021), the Cancer Society in Stockholm (111293) and Karolinska Institutet.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kaipe, H., Erkers, T., Sadeghi, B. et al. Stromal cells–are they really useful for GVHD?. Bone Marrow Transplant 49, 737–743 (2014). https://doi.org/10.1038/bmt.2013.237
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2013.237
Keywords
This article is cited by
-
Mesenchymal stromal cell infusion to treat steroid-refractory acute GvHD III/IV after hematopoietic stem cell transplantation
Bone Marrow Transplantation (2017)
-
Bone marrow-derived mesenchymal stem cells (JR-031) for steroid-refractory grade III or IV acute graft-versus-host disease: a phase II/III study
International Journal of Hematology (2016)