Abstract
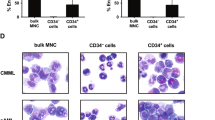
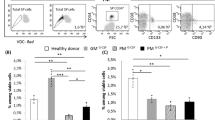
Autologous SCT (auto-SCT) introduces a reduced tolerance to chemotherapy even in patients with adequate engraftment, suggesting long-term effects of the transplantation procedure on the BM capacity. To study the hematopoietic cell compartment after auto-SCT, CD34+ BM cells (n=16) from patients at 6–9 months after auto-SCT were studied with regard to the progenitor subsets, colony frequency and cell cycle status. The BM compartments were studied in vivo using PET tracer 3-fluoro-3-deoxy-L-thymidine (18F-FLT PET). BM CD34+ cells after auto-SCT were compared with normal CD34+ cells and showed a phenotypic shift from common myeloid progenitor (CMP mean percentage 3.7 vs 19.4%, P=0.001) to granulocyte–macrophage progenitor (GMP mean percentage 51.8 vs 27.6%, P=0.01). In addition, a reduced clonogenic potential and higher cycling activity especially of the GMP fraction (41%±4 in G2/S phase vs 19%±2, P=0.03) were observed in BM after auto-SCT compared with normal. The enhanced cycling activity was confirmed in vivo by showing a significantly higher uptake of the 18F-FLT PET tracer by the BM compartment. This study shows that auto-SCT results in defects of the hematopoietic compartment at least 6 months after auto-SCT, characterized by changes in the composition of progenitor subsets and enhanced in vitro and in vivo cycling activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lokhorst HM, Schmidt-Wolf I, Sonneveld P, van der Holt B, Martin H, Barge R et al. Thalidomide in induction treatment increases the very good partial response rate before and after high-dose therapy in previously untreated multiple myeloma. Haematologica 2008; 93: 124–127.
Vellenga E, van Putten WL, van ‘t Veer MB, Zijlstra JM, Fibbe WE, van Oers MH et al. Rituximab improves the treatment results of DHAP-VIM-DHAP and ASCT in relapsed/progressive aggressive CD20+ NHL: a prospective randomized HOVON trial. Blood 2008; 111: 537–543.
Noach EJ, Ausema A, van Os R, Akkerman I, Koopal S, Weersing E et al. Chemotherapy prior to autologous bone marrow transplantation impairs long-term engraftment in mice. Exp Hematol 2003; 31: 528–534.
Domenech J, Linassier C, Gihana E, Dayan A, Truglio D, Bout M et al. Prolonged impairment of hematopoiesis after high-dose therapy followed by autologous bone marrow transplantation. Blood 1995; 85: 3320–3327.
Widmann T, Kneer H, König J, Herrmann M, Pfreundschuh M . Sustained telomere erosion due to increased stem cell turnover during triple autologous hematopoietic stem cell transplantation. Exp Hematol 2008; 36: 104–110.
Thornley I, Sutherland DR, Nayar R, Sung L, Freedman MH, Messner HA . Replicative stress after allogeneic bone marrow transplantation: changes in cycling of CD34+CD90+ and CD34+CD90- hematopoietic progenitors. Blood 2001; 97: 1876–1878.
Manz MG, Miyamoto T, Akashi K, Weissman IL . Prospective isolation of human clonogenic common myeloid progenitors. Proc Natl Acad Sci USA 2002; 99: 11872–11877.
Edvardsson L, Dykes J, Olofsson T . Isolation and characterization of human myeloid progenitor populations–TpoR as discriminator between common myeloid and megakaryocyte/erythroid progenitors. Exp Hematol 2006; 34: 599–609.
Olthof SG, Fatrai S, Drayer AL, Tyl MR, Vellenga E, Schuringa JJ . Downregulation of STAT5 in CD34+ cells promotes megakaryocytic development while activation of STAT5 drives erythropoiesis. Stem Cells 2008; 26: 1732–1742.
Shields AF, Grierson JR, Dohmen BM, Machulla HJ, Stayanoff JC, Lawhorn-Crews JM et al. Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 1998; 4: 1334–1336.
Agool A, Schot BW, Jager PL, Vellenga E . 18F-FLT PET in hematologic disorders: a novel technique to analyze the bone marrow compartment. J Nucl Med 2006; 47: 1592–1598.
Schepers H, van Gosliga D, Wierenga AT, Eggen BJ, Schuringa JJ, Vellenga E . STAT5 is required for long-term maintenance of normal and leukemic human stem/progenitor cells. Blood 2007; 110: 2880–2888.
Bhatia R, Van Heijzen K, Palmer A, Komiya A, Slovak ML, Chang KL . Longitudinal assessment of hematopoietic abnormalities after autologous hematopoietic cell transplantation for lymphoma. J Clin Oncol 2005; 23: 6699–6711.
Rossi DJ, Bryder D, Zahn JM, Ahlenius H, Sonu R, Wagers AJ et al. Cell intrinsic alterations underlie hematopoietic stem cell aging. Proc Natl Acad Sci USA 2005; 102: 9194–9199.
Rossi DJ, Bryder D, Seita J, Nussenzweig A, Hoeijmakers J, Weissman IL . Deficiencies in DNA damage repair limit the function of haematopoietic stem cells with age. Nature 2007; 447: 725–729.
Ju Z, Jiang H, Jaworski M, Rathinam C, Gompf A, Klein C et al. Telomere dysfunction induces environmental alterations limiting hematopoietic stem cell function and engraftment. Nat Med 2007; 13: 742–747.
Kimura T, Asada R, Wang J, Kimura T, Morioka M, Matsui K et al. Identification of long-term repopulating potential of human cord blood-derived CD34-flt3- severe combined immunodeficiency-repopulating cells by intra-bone marrow injection. Stem Cells 2007; 25: 1348–1355.
Li Q, Hisha H, Yasumizu R, Fan TX, Yang GX, Li Q et al. Analyses of very early hemopoietic regeneration after bone marrow transplantation: comparison of intravenous and intrabone marrow routes. Stem Cells 2007; 25: 1186–1194.
Frassoni F, Gualandi F, Podestà M, Raiola AM, Ibatici A, Piaggio G et al. Direct intrabone transplant of unrelated cord-blood cells in acute leukaemia: a phase I/II study. Lancet Oncol 2008; 9: 831–839.
Castello S, Podestà M, Menditto VG, Ibatici A, Pitto A, Figari O et al. Intra-bone marrow injection of bone marrow and cord blood cells: an alternative way of transplantation associated with a higher seeding efficiency. Exp Hematol 2004; 32: 782–787.
Acknowledgements
This study was supported by a grant of the Tekke Huizenga Fonds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Bone Marrow Transplantation website
Supplementary information
Rights and permissions
About this article
Cite this article
Woolthuis, C., Agool, A., Olthof, S. et al. Auto-SCT induces a phenotypic shift from CMP to GMP progenitors, reduces clonogenic potential and enhances in vitro and in vivo cycling activity defined by 18F-FLT PET scanning. Bone Marrow Transplant 46, 110–115 (2011). https://doi.org/10.1038/bmt.2010.75
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.75
Keywords
This article is cited by
-
Nuclear medicine imaging of multiple myeloma, particularly in the relapsed setting
European Journal of Nuclear Medicine and Molecular Imaging (2017)
-
Radionuclide imaging of bone marrow disorders
European Journal of Nuclear Medicine and Molecular Imaging (2011)