Abstract
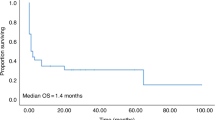
Between January 2001 and July 2006, 1013 patients received autologous hematopoietic cell transplants (AHCT) at Canada's largest transplant center. In this retrospective cohort study of AHCT patients admitted to the intensive care unit (ICU), we describe the outcomes following ICU admission and the variables measured in the first 24 h of ICU admission associated with overall ICU mortality. Results indicate a 3.3% ICU admission rate (n=34) with 13 deaths (1% overall mortality rate, 38% in ICU mortality rate). The worst outcome was in AL amyloid patients of whom 28% were admitted to the ICU, with an ICU mortality rate of 55%. The Sequential Organ Failure Assessment (SOFA) score and Acute Physiology and Chronic Health Evaluation (APACHE II) score in the first 24 h were statistically associated with mortality by univariate analysis. Other variables measured at 24 h and associated with ICU mortality included multiorgan failure, mechanical ventilation, inotropic support >4 h and Gram-negative sepsis. Our data indicate that ICU admission in the autotransplant population is rare and that it is influenced by underlying diagnosis, with AL amyloid patients having the highest risk. Our observations may assist clinical decision-making regarding the continuation of intensive care delivered 24 h after ICU admission.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Price KJ, Thall PF, Kish SK, Shannon VR, Andersson BS . Prognostic indicators for blood and marrow transplant patients admitted to an intensive care unit. Am J Respir Crit Care Med 1998; 158: 876–884.
Copeland E . Hematopioetic stem cell transplantation. N Eng J Med 2006; 354: 1813–1826.
Torrecilla C, Cortes JL, Chamorro C, Rubio JJ, Galdos P, Dominguez de Villota E . Prognostic assessment of the acute complications of bone marrow transplantation requiring intensive therapy. Intensive Care Med 1988; 14: 393–398.
Afessa B, Tefferi A, Hoogland HC, Letendre L, Peters SG . Outcomes of recipients of bone marrow transplants who require intensive care support. Mayo Clin Proc 1992; 67: 117–122.
Crawford SW, Schwartz DA, Petersen FB, Clark JG . Mechanical ventilation after marrow transplantation: risk factors and clinical outcomes. Am Rev Respir Dis 1988; 137: 682–687.
Rubenfeld CD, Crawford SW . Withdrawing life support from mechanically ventilated recipients of bone marrow transplantation: a case for evidence-based guidelines. Ann Intern Med 1996; 125: 625–633.
Naeem N, Reed MD, Creger RJ, Youngner SJ, Lazarus HM . Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006; 37: 119–133.
Gratwohl A, Passweg J, Baldomero H, Horisberger B, Urbano-Ispizua A . Economics, health care systems and utilization of hematopoietic stem cell transplants in Europe. Br J Haematol 2002; 117: 451–468.
Khassawneh BY, White P J, Anaissie EJ, Barlogie B, Hiller FC . Outcome from mechanical ventilation after autologous peripheral blood stem cell transplantation. Chest 2002; 121: 185–188.
Afessa B, Tefferi A, Dunn WF, Litzow MR . Intensive care unit support and acute physiology and chronic health evaluation III performance in hematopoietic stem cell transplant recipients. Crit Care Med 2003; 31: 1715–1721.
Soubani AO, Kseibi E, Bander JJ, Klein JL, Khanchandani G, Ahmed HP et al. Outcome and prognostic factors of hematopoietic stem cell transplantation recipients admitted to a medical ICU. Chest 2004; 126: 1604–1611.
Kew AK, Couban S, Patrick W, Thompson K, White D . Outcome of hematopoietic stem cell transplant recipients admitted to the intensive care unit. Biol Blood Marrow Transplant 2006; 12: 301–395.
Jackson SR, Tweeddale MG, Barnett MJ, Spinelli JJ, Sutherland HJ, Reece DE et al. Admission of bone marrow transplant recipients admitted to an intensive care unit: outcome, survival and prognostic factors. Bone Marrow Transplant 1998; 21: 697–704.
Vincent JL, Moreno R, Takala J, Willatts A, De Mondonca A, Bruining H et al. The SOFA score to describe organ dysfunction/failure. Intensive Care Med 1996; 22: 707–710.
Knaus WA, Draper EA, Wagner DP, Zimmerman JE . Apache II: a severity of disease classification system. Crit Care Med 1985; 13: 818–882.
Cherif H, Martling CR, Hansen J, Kalin M, Björkholm M . Predictors of short and long-term outcome in patients with hematological disorders admitted to the intensive care unit for a life-threatening complication. Support Care Cancer 2007; 15: 1393–1398.
Stem Cell Trialists' Collaborative Group. Allogeneic peripheral blood stem-cell compared with bone marrow transplantation in the management of hematologic malignancies: an individual patient date meta-analysis of nice randomized trials. J Clin Oncol 2005; 23: 5074–5087.
Talmadge JE, Reed E, Ino K, Kessinger A, Kuszynski C, Heimann D et al. Rapid immunologic reconstitution following transplantation with mobilized peripheral blood stem cells as compared to bone marrow. Bone Marrow Transplant 1997; 19: 161–172.
Gertz MA, Lacy MQ, Dispenzieri A, Gastineau DA, Chen MG, Ansell SM et al. Stem cell transplantation for the management of primary systemic amyloidosis. Am J Med 2002; 112: 549–555.
Goodman HJ, Gillmore JD, Lachmann HJ, Wechalekar AD, Bradwell AR, Hawkins PN . Outcome of autologous hematopoietic cell transplantation for AL amyloidosis in the UK. Br J Haematol 2006; 134: 417–425.
Sanchorawala V, Skinner M, Quillen K, Finn KT, Doros G, Seldin DC . Long-term outcome of patients with AL amyloidosis treated with high-dose melphalan and stem-cell transplantation. Blood 2007; 110: 3561–3563.
Skinner M, Sanchorawala V, Seldin DC, Dember LM, Falk RH, Berk JL et al. High-dose melphalan and autologous stem-cell transplantation in patients with AL amyloidosis: a 8-year study. Ann of Int Med 2004; 140: 85–93.
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B et al. Early goal directed therapy in the treatment of septic shock. N Eng J Med 2001; 345: 1368–1377.
The Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Eng J Med 2000; 342: 1301–1308.
Azoulay R, Alberti C, Bornstain C, Leleu G, Moreau D, Recher C et al. Improved survival in cancer patients requiring mechanical ventilatory support: impact of noninvasive mechanical ventilatory support. Crit Care Med 2001; 29: 519–525.
Putsiaka DD, Price LL, Ueuzian A, Chan GW, Miller KB, Snydman DR . Blood stream infection after hematopoeitic stem cell transplantation is associated with increased mortaliy. Bone Marrow Transplant 2007; 40: 63–81.
Collin BA, Leather HL, Wingard JR, Ramphal R . Evolution, incidence, and susceptibility of bacterial bloodstream isolates from 519 bone marrow transplants. Clin Infect Dis 2001; 33: 947–953.
Acknowledgements
Dr A Keating holds the Gloria and Seymour Chair in Cell Therapy and Transplantation at University Health Network and the University of Toronto.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trinkaus, M., Lapinsky, S., Crump, M. et al. Predictors of mortality in patients undergoing autologous hematopoietic cell transplantation admitted to the intensive care unit. Bone Marrow Transplant 43, 411–415 (2009). https://doi.org/10.1038/bmt.2008.336
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2008.336
Keywords
This article is cited by
-
Prognostic Factors and Intensive Care Outcome in Post-Transplant Phase of Hematopoietic Stem Cell Transplantation
Indian Journal of Hematology and Blood Transfusion (2023)
-
Characteristics and outcomes of patients undergoing high-dose chemotherapy and autologous stem cell transplantation admitted to the intensive care unit: a single-center retrospective analysis
Annals of Hematology (2023)
-
Prognostic factors and outcome of adult allogeneic hematopoietic stem cell transplantation patients admitted to intensive care unit during transplant hospitalization
Scientific Reports (2019)
-
Prognostic factors and outcome of patients undergoing hematopoietic stem cell transplantation who are admitted to pediatric intensive care unit
BMC Pediatrics (2016)
-
Impact of critical care outreach on hematopoietic stem cell transplant recipients: a cohort study
Bone Marrow Transplantation (2011)