Abstract
Background:
This randomised study compared the detection rate of cervical intraepithelial neoplasia-positive (CIN2+) based on histology in women performing repeated self-sampling of vaginal fluid (VF) for human papillomavirus (HPV) test with a control group following the ordinary screening by Pap smear cytology.
Methods:
36390 women aged 30–49 years scheduled for invitation to organised screening were randomised in two groups, one to perform self-sampling of VF for HPV test (n=17 997, HPV arm) and the other group to perform screening by PAP smear cytology (n=18 393, control arm). HPV positive women in the HPV arm repeated the self-sampling and the HPV test on average 4.4 months later and those with two consecutive positive HPV tests were referred to colposcopy. Outcome was CIN2+ based on histology during 18-month follow-up.
Results:
Participation rate was 47% in the HPV arm and 39% in the control arm. The HPV prevalence in the first self-sampling was 6.9%, and 71% of these women were HPV positive in their second test. For the per-protocol approach, cumulative prevalence of histological CIN2+ in the HPV arm was 20.2 per 1000 women screened as compared to 10.8 in the control arm. The cumulative prevalence of CIN2+ diagnosed per 1000 years screened was 160.8 in the HPV arm as compared with 25.4 in the control arm.
Conclusions:
Repeated self-sampling of VF and HPV test had more than a two-fold higher discovery rate of CIN2+ per 1000 women screened as compared with PAP smear cytology.
Similar content being viewed by others
Main
Randomised trials have shown that human papillomavirus (HPV)-based primary screening provides 60–70% better protection against cervical intraepithelial neoplasia 2-positive (CIN2+) and invasive cervical cancer than cytology (Ronco et al, 2014; Ogilvie et al, 2017). HPV-based primary screening has higher sensitivity as compared with cytology, but lower specificity due to transient HPV infections. To increase the specificity of HPV-based screening additional biomarkers have been suggested for triaging of HPV-positive women, including cytology and methylation (De Strooper et al, 2016; Luttmer et al, 2016). At present, only cytology is recommended for cotesting of HPV-positive women (Saslow et al, 2012). However, since the sensitivity of cytology is lower than for the HPV test, HPV-infected women may have CIN despite normal cytology and be at risk of developing cancer in the time between screening rounds (Mittal et al, 2017). According to the European Guidelines women who are HPV positive but cytology negative at screening should be recalled for follow-up in 1 year (Arbyn et al, 2010; EU Publications, 2015). As an alternative strategy for managing women with a positive HPV screening test, we have proposed that the HPV test should be repeated in 4–6 months, and thereby it would be possible to distinguish between persistent and transient HPV infections. This strategy requires that a second sample is obtained from the woman, but if the follow-up sample is collected by self-sampling, this does not represent a burden on the health-care system. We have previously shown that ∼40% of women that are HPV positive in their screening test have cleared their infection after 4–6 months (Gyllensten et al, 2011). The strategy of repeating the HPV test results in substantially fewer women requiring follow-up, and a higher specificity for identification of CIN2+ (Gyllensten et al, 2011).
The efficacy of organised screening also depends on the population coverage, and self-sampling of vaginal fluid (VF) at a woman’s own convenience is an attractive strategy to increase the coverage (Stenvall et al, 2006, 2007; Sanner et al, 2009). Providing self-sampling for HPV test to non-attendees has been shown to increase attendance rate and the detection rate of CIN2+ (Wikstrom et al, 2011; Sancho-Garnier et al, 2013) and is equally sensitive for identification of women with CIN2+ as HPV test based on clinician-obtained cervical samples (Snijders et al, 2013). We have shown that self-sampling of VF identifies the high-risk HPV infections found by assisted sampling on the cervix and by histology in women with biopsy-defined cervical lesions (Gustavsson et al, 2011). Self-sampling yields consistent HPV results across menstrual cycle phases and calendar time and, thus, can be used at any time convenient for the woman (Sanner et al, 2015).
The reliability of self-sampling as a screening strategy depends on a number of factors, among those the sampling device. A number of sampling devices have been used in different studies, including brushes, swabs and lavage-based tools. A meta-analysis concluded that the variation seen between studies most likely reflects the use of different sampling devices (Snijders et al, 2013). We used a soft silicon brush for sampling of VF and the indicating FTA elute card, which is a dry storage medium, for application, transport and storage of clinical material (Gustavsson et al, 2011; Sanner et al, 2015). The clinical material applied to this card is confined to a small area instead of diluted in a large liquid volume, such as when using liquid-based transport solutions. Samples applied to the FTA card are stable at room temperature and samples stored for 1 year have shown identical HPV typing results to samples collected using liquid-based media (Barth et al, 2016). Several studies have shown good agreement with respect to HPV detection between the FTA card and liquid-based media (Guan et al, 2012; Wang et al, 2015). To handle the variation in amount of clinical material that may result from self-sampling, it is recommended that a sensitive, PCR-based method, is used for HPV typing (Snijders et al, 2013).
Here, we report the results of the first randomised study comparing the efficacies of using repeated self-sampling of VF and HPV test with Pap smear cytology for the identification of women with CIN2+ based on histology.
Materials and methods
Study population
This randomised intervention study was performed in Uppsala County, Sweden, between 2013 and 2015. The cervical cancer-screening programme in Uppsala County during that period invited women aged 23–49 years (3-year intervals) to Pap smear cytology testing, while women aged 50–60 years were invited to HPV testing (5-year intervals). Women not attending their current screening round were recalled the following year.
Women scheduled for invitation to the organised screening programme between 2013 to 2015 were randomised into two groups: a control group representing women in the screening programme and whose clinical records were used for comparison, and an intervention group that performed repeated self-sampling and HPV test. The protocol for each group is described below. A computer-based allocation process was used for randomisation. During the first year (2013) many of the invited women were never-attenders, resulting in a low participation rate. The women and the clinicians performing the colposcopy were not blinded to the study group, but the pathologists were blinded to the study group.
To be eligible for the study, the woman had to be between 30 and 49 years at entry (date of invitation), having no previous hysterectomy, no current pregnancy and no clinical test results (Pap smear cytology, HPV test or histology) relating to cervical cancer registered within 1 year before the date of invitation. The follow-up period was 18 months from the date of invitation. Participants received oral and/or written information and consent was given. The study was approved by the Regional Ethics Committee in Uppsala (Dnr 2012/099).
HPV arm
Women in the HPV arm were sent an invitation together with a package including information on how to perform the sampling at home, a sampling brush, a FTA card and a preaddressed return envelope. Women who choose not to participate were returned to the screening based on Pap smear cytology. The FTA card was returned by regular mail to the HPV lab at Uppsala University for HPV testing. A reminder was sent to women who did not return their self-sample within 3 weeks. Women who were HPV positive in their first self-sample were informed of the test result within 2 weeks after their sample was returned for HPV testing. These women were informed that they would repeat the self-sampling in 3–6 months, but could contact a gynaecologist in case of questions or symptoms. Women who were HPV positive in two consecutive self-sampling tests were referred to colposcopy and eventual biopsies. Women who were HPV negative in their first or second HPV test were referred back to the regular screening programme.
Control arm
Women were invited to organised screening and managed according to the clinical routine in Uppsala County, where a midwife performed sampling on the cervix for Pap smear cytology. Women with CIN2+ based on cytology were referred to colposcopy within a month, while women with CIN1/ASCUS based on cytology were offered follow-up tests in about 3 months, including both Pap smear cytology and HPV test, according to the clinical routine procedure. HPV-negative women with or without CIN1/ASCUS cytology in the follow-up were returned to the screening programme, while all HPV-positive women, with or without CIN2+ cytology, were referred to colposcopy and eventual biopsies. Women with normal cytology in the follow-up were returned to the regular screening program. Women who did not attend screening were reminded and invited again in 12 months. All gynaecology and colposcopy was performed at the Clinic of Obstetrics and Gynaecology, Uppsala University Hospital and all cytology and histology was performed at the Clinic of Pathology and Cytology, Uppsala University Hospital.
Outcome
The outcome was the number of women with CIN2+ based on histology diagnosed during the 18 months from date of invitation. We included squamous cell carcinoma, adenocarcinoma and adenosquamous carcinoma.
Sample size and power calculations
The study was designed to detect a two-fold difference in the incidence of CIN2+ based on histology between the HPV arm (expected incidence: 1.6%) and the PAP smear cytology arm (expected incidence: 0.8%). These incidence estimates were based on clinical data from Uppsala County and using α-value=0.05. Given these prevalence estimates, a sample size of 5000 women in each arm was found to be necessary to achieve a statistical power higher than 99% to identify a two-fold difference in the incidence of CIN2+ based on histology. The study was not powered to detect an effect of CIN3+.
Self-sampling and sample processing
The self-sampling procedure was based on using a silicon brush and the indicating FTA elute micro card and regular mail both for distribution of the kit and return of the sample. The method for self-sampling of VF has been described previously (Gustavsson et al, 2011). The women were instructed to perform self-sampling of VF using the Rovers Viba-brush (Rover Medical Devices BV, Oss, The Netherlands) and apply the VF sample to the indicating FTA elute micro card (GE Healthcare, Cardiff, UK art. no WB129308). Together with the sampling kit women received an instruction describing how to perform the collection of VF (Figure 1) and a link to a dedicated homepage at the Uppsala University Hospital with an animation of the self-sampling procedure. Self-samples of VF were returned to the HPV laboratory at Uppsala University by regular mail using preaddressed and postage-paid envelopes. At the HPV laboratory, the FTA cards were processed using a dedicated automated laboratory system (easyPunch STARlet; Hamilton Robotics, Bonaduz, Switzerland), which collects each card, takes a photograph of the sample collection area, identifies the parts of the sampling deposition area with the highest amount of cellular material using a machine learning software and then takes four punches with a 3-mm diameter knife from the area containing most material and deposits the punches in a single well in a 96-well microtiter plate. DNA extraction from punches was performed as described earlier (Gustavsson et al, 2009b).
HPV testing system
HPV testing was performed using the real-time PCR-based assay hpVIR (Moberg et al, 2003; Gustavsson et al, 2009a). Briefly, this test detects and quantifies the following HPV types: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58 and 59 and also measures a human single copy gene (HMBS), which serves as a control for that the samples contain sufficient amounts of cellular material for the test to be informative. The limit of detection for the nuclear single copy gene HMBS and HPV was both set to 10 copies per PCR.
Statistical methods
The data were first analysed using a per-protocol approach, that is, including only women assigned to the two arms who complied with the protocol. The cumulative prevalence of CIN2+ based on histology per 1000 women screened, as well as per 1000 years screened was calculated from the date of invitation to the end point for each woman during the 18-month follow-up period in the two study arms. The data weres also analysed using an intention-to-treat approach, by also including women in the HPV arm who were HPV positive in their self-sample screening test, but on their own initiative had a clinical test performed at a health clinic before receiving their second kit for self-sampling. All statistical calculations were performed in R (R Core Team, 2014). P-values were corrected for multiple testing using Bonferroni correction and a P-value <0.05 was considered significant, unless stated otherwise.
Results
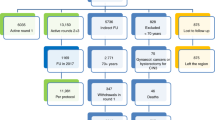
The number of women included and excluded at each stage of the study, as well as the number of CIN2+ detected is shown in Figure 2, and baseline information and statistical calculations are shown in Table 1. The control arm included 16 364 eligible women who were invited for Pap smear cytology, following the clinical routine in Uppsala County, out of which 6364 performed Pap smear cytology. The HPV arm included 17 046 eligible women that received a sampling kit, and among these 7997 performed self-sampling of VF. The mean age of participants and non-participants for the age groups 30–39, 40–49 and 30–49 years was similar in both study arms (Table 1). Also, the mean age of participants was similar between the two study arms (Table 1). The participation rate in the HPV arm was 47% (7997 out of 17 046) as compared with 39% (6364 out of 16 364) in the cytology arm (P<2.2 × 10−16, two-sided Binomial test).
Study design with number of women included and excluded at different steps in HPV arm (self-sampling and repeated HPV test, left) and the Control arm (Pap smear cytology, right). Comments: Excluded not eligible: Women with previous hysterectomy, current pregnancy and clinical test results (cytology, HPV test or histology) relating to cervical cancer registered within 1 year before the date of invitation. Abnormal cytology: ASCUS, CIN 1, CIN 2, CIN 3. Clinical follow-up: HPV test and/or cytology. No CIN2+: CIN1, normal histology or diagnosis unrelated to cervical cancer.
In the HPV arm, 6.3% of the women (554 out of 7997) were HPV positive in their first HPV test. Women with a HPV-positive screening sample showed high compliance (90%, 501 out of 554) to perform a second self-sampling and HPV test. The second HPV test, performed on average 4.4 months after the first, was positive in 71% (355 out of 501) of the women. Following the per-protocol approach, 49% (162 out of 330) of the women in the HPV arm who were HPV positive in the follow-up test received a CIN2+ diagnosis based on histology, and of these women, 48% (77 out of 162) had CIN3+ and 52% (85 out of 162) had CIN2.
Among the women who were HPV positive in their first self-sample, 53 did not perform the second self-sampling, but 37 of them instead had a follow-up test taken at a health-care centre before receiving the second self-sampling kit. Among these 37 women, 24 had a biopsy taken and 13 of the 24 women had CIN2+ histology (4 had CIN3+ and 9 had CIN2). The 13 women with CIN2+ diagnosis was included in the intention-to-treat calculation (Figure 2).
In the control arm, 3.5% (222 out of 6364) had abnormal cytology based on their Pap smear screening sample and 85% (188 out of 222) of these women participated in the clinical follow-up. Among the women with a CIN2+ or CIN1/ASCUS cytology screening sample, 101 received a histology diagnosis and 61% (69 out of 101) of these had CIN2+. Among the 69 women with CIN2+ histology, 75% (52 out of 69) had CIN3+ and 25% (17 out of 69) had CIN2.
The compliance rate in the HPV arm, that is, the participation rate in screening times the fraction of women with a first HPV-positive sample that performed a second self-sampling, was 0.47 (7997/17 046) × 0.90 (501/554)=0.42. By comparison, the participation rate in cytology screening times the return rate for follow-up testing by cytology and HPV was 0.39 (6364/16 364) × 0.85 (188/222)=0.33.
We first examined the time required to receive a diagnosis as Normal or CIN2+. The median time to diagnosis as Normal (i.e. the time required for a woman to be returned to the screening population) was 2.6 times longer in the control arm (82 days) than in the HPV arm (32 days) (Table 1), while the time to a CIN2+ diagnosis was similar between the HPV arm (253 days) and the control arm (300 days).
The cumulative prevalence of women with the diagnosis as Normal, CIN2 or CIN2+ per 1000 years screened is shown in Figures 3A–C. Figure 3A shows the very rapid rate of receiving the diagnosis as Normal in the HPV arm, relative to the control arm. The cumulative prevalence of CIN2+ per 1000 years screened was about six times higher in the HPV arm (160.8) as compared with the control arm (25.4); a difference that is statistically significant (Table 1 and Figure 3C). Much of the difference in the cumulative prevalence of CIN2+ can be attributed to the detection of CIN2 (Figure 3B).
Cumulative prevalence per 1000 years screened of women with the diagnosis normal (A), CIN2 (B) and CIN2+ (C) in the HPV arm (self-sampling of VF sample and HPV test) in blue and the control arm (Pap smear cytology) in red. Cumulative prevalence per 1000 women screened of women with the diagnosis Normal (D), CIN2 (E) and CIN2+ (F) in the HPV arm (self-sampling of VF sample and HPV test) in blue and the control arm (Pap smear cytology) in red. Dotted blue line shows the result for intention-to-treat (itt) and solid line per-protocol (pp). Transparent fields show 95% confidence interval based on number of diagnoses per 1000 years screened (B and C), or per 1000 women screened (E and F) during the follow-up period.
We also calculated the cumulative prevalence of women with CIN2+ histology per 1000 women screened. For the per-protocol approach, the cumulative prevalence in the HPV arm was 20.2 per 1000 women screened (162/7997 × 1000) (95% CI: 17.52, 23.75) as compared with 10.8 in the control arm (69/6364 × 1000) (95% CI: 7.77, 12.70) (Table 1 and Figure 3F). Including also women from the intention-to-treat approach, the cumulative prevalence of women with CIN2+ histology per 1000 women screened was 21.9 (175/7997 × 1000) (95% CI: 18.68, 25.09) in the HPV arm as compared with 10.8 in the control arm (69/6364 × 1000) (95% CI: 7.77, 12.70) (Table 1). The cumulative prevalence of CIN2+ was statistically significantly higher in the HPV arm than in the control arm (per-protocol and intention-to-treat combined) for both the age group 30–39 and 40–49 years (Table 1).
The two screening strategies identified about the same number of CIN3+ per 1000 women screened (HPV arm: 10.1 (81/7997 × 1000), control arm: 8.2 (52/6364 × 1000)). However, the HPV arm identified four times as many CIN2 lesions per 1000 women screened as the control arm (HPV arm: 11.7 (94/7997 × 1000), control arm: 2.7 (17/6364 × 1000)) (Table 1 and Figure 3E).
The positive predictive value (PPV) for detection of CIN2+ was 0.46 (95% CI: 0.41, 0.51, n=162 of 355) for the HPV arm and 0.37 (95% CI: 0.30, 0.44, n=69 of 188) for the cytology arm; a difference that is statistically significant (P<6.2 × 10−4, two-sided Binomial test).
Discussion
We have shown that primary screening based on self-sampling and repeated HPV test can detect more than twice as many women with CIN2+ histology than using PAP smear cytology of cervical cells. The results have important implications for the use of self-sampling, primary screening by HPV and the health-economy of organised screening programs.
Women in the HPV arm showed a higher participation rate than women in the control arm, and the compliance to perform the second HPV test was also higher than for women in the control arm to perform follow-up with HPV test and cytology. Thus, in our study women appear as likely to perform self-sampling for HPV test as attending a health clinic for cervical sampling. The high compliance rate of women with a positive HPV screening test to perform their second self-sampling (90%) indicate that the interval between the first and second self-sampling was acceptable to most women. Among the 37 women who on their own made an appointment at a health clinic after the first self-sampling test, and for which we have information, 60% reported a medical reason for the appointment rather than that they did not want to wait to perform a second self-sampling. The time between the two HPV tests in our study is similar to the follow-up time of 3 months used in organised cytology screening program in Uppsala County for women with CIN1/ASCUS cytology.
The high detection rate of CIN2+ in the HPV arm is the consequence of using the HPV test both as a screening test and in the follow-up. In the time between the HPV screening test and the repeat HPV test ∼30% of HPV-positive women cleared their infection. Among women who were HPV positive in the repeat test, and for which histology information was available, 49% (162 out of 330) had CIN2+ lesions. In a previous study of 8000 non-attendees of organised screening that were offered self-sampling at home, we used HPV test both in screening and follow-up, similar to the present study, and found an increased specificity of detecting CIN2+ histology using the repeat HPV test strategy (Gyllensten et al, 2011). The screening strategy could be further improved by extending the time between the first and second HPV test, allowing for more transient infections to clear. The European Guidelines for women with a HPV-positive, cytology-negative, screening test recommend either follow-up by repeating the HPV testing in 12 months, performing colposcopy or returning the woman to routine screening (EU Publications, 2015).
Self-sampling and HPV test detected both CIN2 and CIN2+, while cytology detected mainly CIN3+ lesions (Table 1). No difference was seen between the two study arms with respect to the detection rate of CIN3, but the HPV arm identified four times as many CIN2 per woman screened. CIN2 lesions are difficult to unambiguously identify and separate from CIN3 (Castle et al, 2007). Spontaneous regression of CIN2, as well as CIN3, have been reported, and the frequency of such events varies between studies and age groups. In younger women (below 30 years of age) regression appears to be relatively common, while there is less data for women above 40 years of age (Wang et al, 2013; Uleberg et al, 2014; Munro et al, 2016). Using a sensitive screening strategy that enables identification of CIN2 lesions, such as the one in our study, provides an opportunity for early detection, but at the same time requires a strategy for identification of lesions that may not need treatment. Biomarkers could be used to identify women with CIN2 that are most likely to progress. HPV genotyping could serve to identify women with CIN2 carrying persistent infections with HR-HPV, in contrast to those with low viral load of less carcinogenic HPV types (Schiffman et al, 2015; Lie et al, 2017). Also, p16INK4a staining has shown promise as a marker for identifying women with CIN2 that will regress spontaneously (Miralpeix et al, 2017). There is clear evidence and clinical praxis that CIN2 should be treated in this age group. According to the new Swedish national recommendations (Swedish Board For Health and Welfare (Socialstyrelsen), 2015) CIN2 as well as CIN3 lesions should be treated when identified in screening in this age group. In the light of this, we were unable to choose a different clinical outcome for the present study.
The estimated PPV for CIN2+ based on cytology in our study (PPV=0.37) was similar to the PPV=0.30 for the Swedish population (ages 30–59 years) as reported in 2017 (Ponti et al, 2017, Table 4.13.5). The estimated PPV based on self-sampling and HPV (PPV=0.45) was substantially higher.
The application of self-sampling for HPV test has health-economic advantages. Health-economic modelling has shown that a screening strategy based on self-sampling and repeated HPV test, as used in the present study, is dominating over the use of cytology alone, cytology triaged by HPV or primary screening by HPV test based on samples collected at a midwife’s clinic (Ostensson et al, 2013). Since the strategy used in the present study results in the identification of a higher number of CIN2+ lesions, it also increases the number of colposcopies and histological examinations that have to be performed. The cost of these analyses has been included in the modelling above and the screening strategy based on self-sampling and repeated HPV test was still found to be dominating from a health-economic perspective. This is partly the consequence of that self-sampling of VF that will provide large savings to the primary health-care system. Thus, the increased cost of colposcopies and histology is favourably balanced by reduced overall costs for the screening. In exploring self-sampling strategies, the choice of method for reaching the women is important to achieve an optimal cost-efficiency. For instance, Haguenoer et al (2014) concluded that offering an in-home, return-mail kit for vaginal self-sampling is more efficient and cost-effective than a recall letter in increasing participation in cervical cancer screening (Haguenoer et al, 2014). The magnitude of the health benefit that can be achieved by self-sampling will, however, depend on the screening history and compliance of respondents (Burger et al, 2017).
There are certain limitations to our study. The sampling device and FTA card have not been compared with the more common cytobrush and liquid-based transport media in large controlled studies, but a number of studies have shown that VF can be reliably obtained using a number of sampling devices and collection media (Arbyn et al, 2014; Jentschke et al, 2016; Karjalainen et al, 2016). The VIBA-brush used has been shown to provide similar results to other sampling devices (Bais et al, 2007; Lenselink et al, 2009; Gok et al, 2012; Geraets et al, 2013; Arbyn et al, 2014). We used a laboratory developed HPV test (hpVIR) that has similar characteristics as the Hybrid Capture (HC2) test (Gustavsson et al, 2009a) and has been shown to have 100% proficiency using the WHO HPV panel (Eklund et al, 2014). Thus, the sampling device, collection medium and HPV test we used are not likely to have underestimated the number of HPV-positive women.
We compared self-sampling for HPV test to Pap smear cytology rather than Liquid Based Cytology (LBC), since that was the routine clinical test used in the organised screening in Uppsala County during the project period. International studies have shown a higher sensitivity of LBC, but no difference in specificity (Cox, 2004). Some Swedish studies have found a higher detection rate of CIN2+ (Strander et al, 2007), while other found no difference between the two methods (Froberg et al, 2013). In a survey from 2015, 8 out of 21 European countries were still only using Pap smear cytology and 15 out of 21 countries Pap smear cytology in combination with LBC (Elfström et al, 2015). Given the small difference in detection rate between LBC and Pap smear cytology in relation to that seen between cytology and self-sampling and HPV in our study, the choice of cytology method would not have any major impact on our conclusions. Also, our detection rate of CIN2+ per 1000 women in screening (1.08%) is very similar to the value reported for Sweden as a whole of 0.7% (Ponti et al, 2017, Table 4.13.3). This value later is based on women between 30 and 59 years, and therefore is expected to be somewhat lower than for the age range 30–49 years. Women scheduled for the organised screening were randomised and then returned to the organised screening (control arm) or sent information for self-sampling. This could have resulted in an age difference, but we did not detect a difference in the age distribution between participants and non-participants of each study arm or between the participants of the two study arms. Women participating in the HPV arm did not have to pay, while there was a small fee to perform Pap smear cytology. This could have introduced a bias in favour of self-sampling. Since nothing was mentioned in the information to the woman receiving the self-sampling kit regarding the fee we do not think this asymmetry had a significant impact on the participation rate. Women in the HPV arm who had not returned their sample within 3 weeks were reminded by letter, while the women in the organised screening had to wait until the following year (12 months) to be reminded. This bias might have contributed to the higher participation rate seen in the HPV arm, but the magnitude of this effect is difficult to estimate.
In summary, the strategy of self-sampling of VF and repeating the HPV test resulted in more than a two-fold higher detection rate of CIN2+ lesions as compared with Pap smear cytology. Self-sampling followed by HPV test increased the number of CIN2 lesions detected, providing an opportunity for early detection, and also underscoring the need for additional biomarkers than can identify lesions that may not need treatment.
Change history
20 March 2018
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Arbyn M, Anttila A, Jordan J, Ronco G, Schenck U, Segnan N, Wiener H, Herbert A, Von Karsa L (2010) European Guidelines for Quality Assurance in Cervical Cancer Screening. Second edition – summary document. Ann Oncol 21: 448–458.
Arbyn M, Verdoodt F, Snijders PJ, Verhoef VM, Suonio E, Dillner L, Minozzi S, Bellisario C, Banzi R, Zhao FH, Hillemanns P, Anttila A (2014) Accuracy of human papillomavirus testing on self-collected versus clinician-collected samples: a meta-analysis. Lancet Oncol 15: 172–183.
Bais AG, Van Kemenade FJ, Berkhof J, Verheijen RH, Snijders PJ, Voorhorst F, Babovic M, Van Ballegooijen M, Helmerhorst TJ, Meijer CJ (2007) Human papillomavirus testing on self-sampled cervicovaginal brushes: an effective alternative to protect nonresponders in cervical screening programs. Int J Cancer 120: 1505–1510.
Barth H, Morel A, Mougin C, Averous G, Legrain M, Fender M, Risch S, Fafi-Kremer S, Velten M, Oudet P, Baldauf JJ, Stoll-Keller F (2016) Long-term storage and safe retrieval of human papillomavirus DNA using FTA elute cards. J Virol Method 229: 60–65.
Burger EA, Sy S, Nygard M, Kim JJ (2017) The cost-effectiveness of cervical self-sampling to improve routine cervical cancer screening: the importance of respondent screening history and compliance. Cancer Epidemiol Biomarkers Prev 26: 95–103.
Castle PE, Stoler MH, Solomon D, Schiffman M (2007) The relationship of community biopsy-diagnosed cervical intraepithelial neoplasia grade 2 to the quality control pathology-reviewed diagnoses: an ALTS report. Am J Clin Pathol 127: 805–815.
Cox JT (2004) Liquid-based cytology: evaluation of effectiveness, cost-effectiveness, and application to present practice. J Natl Compr Canc Netw 2: 597–611.
De Strooper LMA, Verhoef VMJ, Berkhof J, Hesselink AT, De Bruin HME, Van Kemenade FJ, Bosgraaf RP, Bekkers RLM, Massuger L, Melchers WJG, Steenbergen RDM, Snijders PJ, Meijer C, Heideman DAM (2016) Validation of the FAM19A4/mir124-2 DNA methylation test for both lavage- and brush-based self-samples to detect cervical (pre)cancer in HPV-positive women. Gynecol Oncol 141: 341–347.
Eklund C, Forslund O, Wallin KL, Dillner J (2014) Global improvement in genotyping of human papillomavirus DNA: the 2011 HPV LabNet International Proficiency Study. J Clin Microbiol 52: 449–459.
Elfström KM, Arnheim-Dahlström L, Von Karsa L, Dillner L (2015) Cervical cancer screening in Europe: quality assurance and organisation of programmes. Eur J Cancer 51: 950–968.
EU Publications (2015) European Guidelines for Quality Assurance in Cervical Cancer Screening EU Publications. Available at: https://publications.europa.eu/new-bookshop/?.../european-guid.10.2875/859507.
Froberg M, Norman I, Johansson B, Hjerpe A, Weiderpass E, Andersson S (2013) Liquid-based cytology with HPV triage of low-grade cytological abnormalities versus conventional cytology in cervical cancer screening. Curr Pharm Des 19: 1406–1411.
Geraets DT, Van Baars R, Alonso I, Ordi J, Torne A, Melchers WJ, Meijer CJ, Quint WG (2013) Clinical evaluation of high-risk HPV detection on self-samples using the indicating FTA-elute solid-carrier cartridge. J Clin Virol 57: 125–129.
Gok M, Van Kemenade FJ, Heideman DA, Berkhof J, Rozendaal L, Spruyt JW, Belien JA, Babovic M, Snijders PJ, Meijer CJ (2012) Experience with high-risk human papillomavirus testing on vaginal brush-based self-samples of non-attendees of the cervical screening program. Int J Cancer 130: 1128–1135.
Guan Y, Castle PE, Wang S, Li B, Feng C, Ci P, Li X, Gravitt P, Qiao YL (2012) A cross-sectional study on the acceptability of self-collection for HPV testing among women in rural China. Sex Transm Infect 88: 490–494.
Gustavsson I, Juko-Pecirep I, Backlund I, Wilander E, Gyllensten U (2009a) Comparison between the hybrid capture 2 and the hpVIR real-time PCR for detection of human papillomavirus in women with ASCUS or low grade dysplasia. J Clin Virol 45: 85–89.
Gustavsson I, Lindell M, Wilander E, Strand A, Gyllensten U (2009b) Use of FTA card for dry collection, transportation and storage of cervical cell specimen to detect high-risk HPV. J Clin Virol 46: 112–116.
Gustavsson I, Sanner K, Lindell M, Strand A, Olovsson M, Wikstrom I, Wilander E, Gyllensten U (2011) Type-specific detection of high-risk human papillomavirus (HPV) in self-sampled cervicovaginal cells applied to FTA elute cartridge. J Clin Virol 51: 255–258.
Gyllensten U, Sanner K, Gustavsson I, Lindell M, Wikstrom I, Wilander E (2011) Short-time repeat high-risk HPV testing by self-sampling for screening of cervical cancer. Br J Cancer 105: 694–697.
Haguenoer K, Sengchanh S, Gaudy-Graffin C, Boyard J, Fontenay R, Marret H, Goudeau A, Pigneaux De Laroche N, Rusch E, Giraudeau B (2014) Vaginal self-sampling is a cost-effective way to increase participation in a cervical cancer screening programme: a randomised trial. Br J Cancer 111: 2187–2196.
Jentschk M, Chen K, Arbyn M, Hertel B, Noskowicz M, Soergel P, Hillemanns P (2016) Direct comparison of two vaginal self-sampling devices for the detection of human papillomavirus infections. J Clin Virol 82: 46–50.
Karjalainen L, Anttila A, Nieminen P, Luostarinen T, Virtanen A (2016) Self-sampling in cervical cancer screening: comparison of a brush-based and a lavage-based cervicovaginal self-sampling device. BMC Cancer 16: 221.
Lenselink CH, De Bie RP, Van Hamont D, Bakkers JM, Quint WG, Massuger LF, Bekkers RL, Melchers WJ (2009) Detection and genotyping of human papillomavirus in self-obtained cervicovaginal samples by using the FTA cartridge: new possibilities for cervical cancer screening. J Clin Microbiol 47: 2564–2570.
Lie AK, Trope A, Skare GB, Bjorge T, Jonassen CM, Brusegard K, Lonnberg S (2018) HPV genotype profile in a Norwegian cohort with ASC-US and LSIL cytology with three year cumulative risk of high grade cervical neoplasia. Gynecol Oncol 148: 111–117.
Luttmer R, De Strooper LM, Dijkstra MG, Berkhof J, Snijders PJ, Steenbergen RD, Van Kemenade FJ, Rozendaal L, Helmerhorst TJ, Verheijen RH, Ter Harmsel WA, Van Baal WM, Graziosi PG, Quint WG, Spruijt JW, Van Dijken DK, Heideman DA, Meijer CJ (2016) FAM19A4 methylation analysis in self-samples compared with cervical scrapes for detecting cervical (pre)cancer in HPV-positive women. Br J Cancer 115: 579–587.
Miralpeix E, Genoves J, Maria Sole-Sedeno J, Mancebo G, Lloveras B, Bellosillo B, Alameda F, Carreras R (2017) Usefulness of p16INK4a staining for managing histological high-grade squamous intraepithelial cervical lesions. Mod Pathol 30: 304–310.
Mittal SBP, Muwonge R, Banerjee D, Ghosh I, Sengupta Mm, Das P, Dey P, Mandal R, Panda C, Biswas J, Sankaranarayanan R (2017) Risk of high-grade precancerous lesions and invasive cancers in high-risk HPV-positive women with normal cervix or CIN 1 at baseline – a population-based cohort study. Int J Cancer 140: 1850–1859.
Moberg M, Gustavsson I, Gyllensten Ub (2003) Real-time PCR-based system for simultaneous quantification of human papillomavirus types associated with high risk of cervical cancer. J Clin Microbiol 41: 3221–3228.
Munro A, Powell RG, Bowen S, Spilsbury K, O'leary P, Semmens JB, Codde J, Williams V, Steel N, Leung Y (2016) Spontaneous regression of CIN2 in women aged 18-24 years: a retrospective study of a state-wide population in Western Australia. Acta Obstet Gynecol Scand 95: 291–298.
Ogilvie GS, Krajden M, Van Niekerk D, Smith LW, Cook D, Ceballos K, Lee M, Gentile L, Gondara L, Elwood-Martin R, Peacock S, Stuart G, Franco EL, Coldman AJ (2017) HPV for cervical cancer screening (HPV FOCAL): complete round 1 results of a randomised trial comparing HPV-based primary screening to liquid-based cytology for cervical cancer. Int J Cancer 140: 440–448.
Ostensson E, Hellstrom AC, Hellman K, Gustavsson I, Gyllensten U, Wilander E, Zethraeus N, Andersson S (2013) Projected cost-effectiveness of repeat high-risk human papillomavirus testing using self-collected vaginal samples in the Swedish cervical cancer screening program. Acta Obstet Gynecol Scand 92: 830–840.
Ponti A, Anttila A, Ronco G, Senore C (2017) Cancer screening in the European Union. Report on the Implementation of the Council Recommendation on Cancer Screening International Agency for Research on Cancer. Available at: http://edz.bib.uni-mannheim.de/daten/edz-a/gdgv/17/2017_cancerscreening_2ndreportimplementation_en.pdf.
R Core Team (2014) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, Available at: URL http://www.R-project.org/.
Ronco G, Dillner J, Elfstrom KM, Tunesi S, Snijders PJ, Arbyn M, Kitchener H, Segnan N, Gilham C, Giorgi-Rossi P, Berkhof J, Peto J, Meijer CJ International, HPVSWG (2014) Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. Lancet 383: 524–532.
Sancho-Garnier H, Tamalet C, Halfon P, Leandri FX, Le Retraite L, Djoufelkit K, Heid P, Davies P, Piana L (2013) HPV self-sampling or the Pap-smear: a randomised study among cervical screening nonattenders from lower socioeconomic groups in France. Int J Cancer 133: 2681–2687.
Sanner K, Wikstrom I, Gustavsson I, Wilander E, Lindberg JH, Gyllensten U, Olovsson M (2015) Daily self-sampling for high-risk human papillomavirus (HR-HPV) testing. J Clin Virol 73: 1–7.
Sanner K, Wikstrom I, Strand A, Lindell M, Wilander E (2009) Self-sampling of the vaginal fluid at home combined with high-risk HPV testing. Br J Cancer 101: 871–874.
Saslow D, Solomon D, Lawson HW, Killackey M, Kulasingam SL, Cain JM, Garcia FA, Moriarty AT, Waxman AG, Wilbur DC, Wentzensen N, Downs LS Jr, Spitzer M, Moscicki AB, Franco EL, Stoler MH, Schiffman M, Castle PE, Myers ER, Chelmow D, Herzig A, Kim JJ, Kinney W, Herschel WL, Waldman J (2012) American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. J Low Genit Tract Dis 16: 175–204.
Schiffman M, Vaughan LM, Raine-Bennett TR, Castle PE, Katki HA, Gage JC, Fetterman B, Befano B, Wentzensen N (2015) A study of HPV typing for the management of HPV-positive ASC-US cervical cytologic results. Gynecol Oncol 138: 573–578.
Snijders PJ, Verhoef VM, Arbyn M, Ogilvie G, Minozzi S, Banzi R, Van Kemenade FJ, Heideman DA, Meijer CJ (2013) High-risk HPV testing on self-sampled versus clinician-collected specimens: a review on the clinical accuracy and impact on population attendance in cervical cancer screening. Int J Cancer 132: 2223–2236.
Stenvall H, Wikstrom I, Wilander E (2006) Human papilloma virus testing of vaginal smear obtained with a novel self-sampling device. Acta Dermatol Venereol 86: 465–467.
Stenvall H, Wikstrom I, Wilander E (2007) High prevalence of oncogenic human papilloma virus in women not attending organized cytological screening. Acta Dermatol Venereol 87: 243–245.
Strander B, Andersson-Ellstrom A, Milsom I, Radberg T, Ryd W (2007) Liquid-based cytology versus conventional Papanicolaou smear in an organized screening program: a prospective randomised study. Cancer 111: 285–291.
Swedish Board For Health And Welfare (Socialstyrelsen) (2015) Screening för livmoderhalscancer. Rekommendations och bedömningsunderlag. ISBN 978-91-7555-322-1. Report 2015-6-29 Available at: www.socialstyrelsen.se/publikationer2015/2015-6-39.
Uleberg KE, Ovestad IT, Munk AC, Brede C, Van Diermen B, Gudlaugsson E, Janssen EA, Hjelle A, Baak JP (2014) Prediction of spontaneous regression of cervical intraepithelial neoplasia lesions grades 2 and 3 by proteomic analysis. Int J Proteomics 2014: 129064.
Wang SM, Colombara D, Shi JF, Zhao FH, Li J, Chen F, Chen W, Li SM, Zhang X, Pan QJ, Belinson JL, Smith JS, Qiao YL (2013) Six-year regression and progression of cervical lesions of different human papillomavirus viral loads in varied histological diagnoses. Int J Gynecol Cancer 23: 716–723.
Wang SM, Hu SY, Chen W, Chen F, Zhao FH, He W, Ma XM, Zhang YQ, Wang J, Sivasubramaniam P, Qiao YL (2015) Feasibility and accuracy evaluation of three human papillomavirus assays for FTA card-based sampling: a pilot study in cervical cancer screening. BMC Cancer 15: 848.
Wikstrom I, Lindell M, Sanner K, Wilander E (2011) Self-sampling and HPV testing or ordinary Pap-smear in women not regularly attending screening: a randomised study. Br J Cancer 105: 337–339.
Acknowledgements
This work was supported by the Swedish Cancer Foundation, the Swedish Medical Research Council and the Foundation for Strategic Research. We thank all of the participants and the staff who made this study possible.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The funders of the study had no role in the design, data collection, data analysis, data interpretation or writing of the report. All authors had full access to the data in the study and the corresponding author (UG) had final responsibility for the decision to submit for publication. The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Gustavsson, I., Aarnio, R., Berggrund, M. et al. Randomised study shows that repeated self-sampling and HPV test has more than two-fold higher detection rate of women with CIN2+ histology than Pap smear cytology. Br J Cancer 118, 896–904 (2018). https://doi.org/10.1038/bjc.2017.485
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2017.485
Keywords
This article is cited by
-
Incidence of oncogenic HPV and HPV-related dysplasia five years after a negative HPV test by self-sampling in elderly women
Infectious Agents and Cancer (2022)
-
Acceptance of Self-Sampling Among Long-Term Cervical Screening Non-Attenders with HPV-Positive Results: Promising Opportunity for Specific Cancer Education
Journal of Cancer Education (2021)
-
Implementation of HPV Self-sampling in Cervical Screening Programs Increases Participation Rates: A Literature Review
Indian Journal of Gynecologic Oncology (2021)
-
Cost-effectiveness analysis of repeated self-sampling for HPV testing in primary cervical screening: a randomized study
BMC Cancer (2020)
-
Temporal changes in the vaginal microbiota in self-samples and its association with persistent HPV16 infection and CIN2+
Virology Journal (2020)