Abstract
Background:
Golgi phosphoprotein 3 (GOLPH3) has been reported to be involved in the development of several human cancers. The present study was conducted to investigate the expression of GOLPH3 and its prognostic significance in renal cell carcinoma (RCC). Meanwhile, the function of GOLPH3 in human RCC was further investigated in cell culture models.
Methods:
Expression of GOLPH3 was examined in 43 fresh RCC tissues and paired adjacent normal renal tissues by real-time quantitative PCR and western blotting. Immunohistochemistry for GOLPH3 was performed on additional 218 RCC tissues. The clinical significance of GOLPH3 expression was analysed. Downregulation of GOLPH3 was performed using small-interfering RNA (siRNA) in Caki-1 and 786-O cells with high abundance of GOLPH3, and the effects of GOLPH3 silencing on cell proliferation, migration, invasion in vitro, and tumour growth in vivo were evaluated.
Results:
Expression of GOLPH3 was upregulated in the majority of the RCC clinical tissue specimens at both mRNA and protein levels. Clinicopathological analysis showed that GOLPH3 expression was significantly correlated with T stage (P<0.001), lymph-node status (P=0.003), distant metastasis (P<0.001), tumour-node-metastasis (TNM) stage (P<0.001), and Fuhman grade (P=0.001). Expression of GOLPH3 was inversely correlated with both overall and recurrence-free survival of RCC patients. Multivariate analysis showed that GOLPH3 expression was an independent prognostic indicator for patient’s survival. Knockdown of the GOLPH3 expression reduced cell proliferation, anchorage-independent growth, migration, invasion, and tumour growth in xenograft model mice.
Conclusions:
These results suggest that GOLPH3 expression is likely to have important roles in RCC development and progression, and that GOLPH3 is a prognostic biomarker and a promising therapeutic target for RCC.
Similar content being viewed by others
Main
Renal cell carcinoma (RCC) represents ∼3% of all adult malignancies (Rini et al, 2008), being the most common cancer of the adult kidney (Rini et al, 2009). Approximately 60 920 new cases of RCC were diagnosed in the United States in 2011, with estimated 13 120 deaths (Siegel et al, 2011). Worldwide, over 200 000 new cases of RCC are diagnosed annually and more than 100 000 deaths occur from this disease (Gupta et al, 2008). Because there are no recommended screening tests for RCC, about 25–30% of patients, at the time of diagnosis, present with metastatic disease (Lam et al, 2005). Surgery is the mainstay of therapy for early-stage, localised RCC; however, surgical resection of locally advanced disease is associated with a high recurrence rate (Jiang et al, 2008). Furthermore, for metastatic RCC (mRCC), surgery does not usually change the progressive nature of the disease (Chan and Marshall, 1998). The clinical outcome of mRCC is poor, with a median survival of only 1.5 years in a recent cohort (Patil et al, 2010). Metastatic RCC is typically resistant to conventional chemotherapy, radiotherapy, and hormonal therapy (Lam et al, 2008). Cytokine therapies have long been the standard of care in mRCC. However, these agents only provide clinical benefit to a small subset of patients and are associated with a significant toxicity (Motzer et al, 2000; Yang et al, 2003). Novel therapeutic agents that target angiogenesis, such as sunitinib and axitinib, have been demonstrated to be more effective in treating for mRCC. Objective response rates of 40–50% have been reported with these targeted agents (Rixe et al, 2007; Motzer et al, 2009; Tomita et al; 2011). Nevertheless, durable responses are rare and most patients eventually develop progressive disease (Albiges et al, 2012). Therefore, novel alternative molecular therapeutic targets need to be discovered.
Golgi phosphoprotein 3 (GOLPH3), also known as GPP34/GMx33/MIDAS, is a newly identified protein with a molecular weight of 34 kDa in the trans-Golgi matrix, which has a role in anterograde and retrograde Golgi trafficking (Wu et al, 2000; Nakashima-Kamimura et al, 2005; Snyder et al, 2006). It is localised to 5p13, a region frequently amplified in several types of cancer (Böhm et al, 2002; Yokoi et al, 2002; Gorringe et al, 2005). It has recently been recognised as a novel oncogene involved in tumorigenesis via enhancing activity of the mammalian target of rapamycin (mTOR), a serine/threonine protein kinase known to regulate cell growth, proliferation, and survival (Abraham, 2009; Scott et al, 2009). Downregulation of GOLPH3 abrogates transformation and tumour cell proliferation in GOLPH3-amplified cell lines, whereas ectopic expression of GOLPH3 drives transformation of primary cell lines and enhances mouse xenograft tumour growth in vivo (Scott et al, 2009; Scott and Chin, 2010).
Recent studies have documented that GOLPH3 is overexpressed in many types of human cancers such as breast cancer (Zeng et al, 2012), prostate cancer (Hua et al, 2012), oral tongue cancer (Li et al, 2012), oesophageal cancer (Wang et al, 2012), gastric cancer (Hu et al, 2013), and glioma (Zhou et al, 2012). More importantly, the aberrant expression of GOLPH3 in these cancers has been indicated to be correlated with clinically aggressive behaviour and patient survival. Silencing of GOLPH3 using small-interfering RNAs (siRNAs) causes reduced tumour cell migration and invasion capacity in glioma-derived cell line, U251 and U87 (Zhou et al, 2013). These findings suggest an involvement of GOLPH3 in cancer progression. However, it is still not known about its expression pattern, clinical relevance, and biological function in RCC. With this in mind, we undertook a study with two objectives: (a) to determine the correlations between GOLPH3 expression and various clinicopathologic parameters, and its prognostic value for survival of patients with RCC and (b) to determine whether silencing of GOLPH3 in RCC cell lines affects cell proliferation, migration, invasion, and in vivo tumour growth.
Here, we showed that GOLPH3 is frequently overexpressed in RCC tissues, and that this upregulation is significantly associated with worse prognosis of RCC patients. Depletion of GOLPH3 expression using the siRNA technique in 786-O and Caki-1 cells decreased cell proliferation, migration, and invasion. In vivo studies further showed that GOLPH3 silencing dramatically retarded xenograft tumour growth in nude mice. Together, our data highlight an important role for GOLPH3 in controlling RCC progression.
Materials and methods
Patients and surgical specimens
A series of 218 patients with histologically confirmed RCC (178 clear cell RCC, 26 papillary RCC, and 14 chromophobe RCC) were included in the study. The patients underwent nephrectomy in the Department of Urology, First Affiliated Hospital of Gannan Medical University (Ganzhou, China) between 2003 and 2011. None of the patients received chemotherapy or radiotherapy before surgery. After surgical resection, tumour specimens and the corresponding adjacent non-tumour tissues were collected and stored in liquid nitrogen until use. Parts of each sample were fixed in formalin, embedded in paraffin and stored in the Department of Pathology, First Affiliated Hospital of Gannan Medical University. In total, 141 of these 218 patients were men and 77 were women. The median age of the patients was 58 years (range, 31–76 years). All of the cases were staged according to the tumour-node-metastasis (TNM) staging system and nuclear grade was evaluated on the basis of the Fuhman criteria. The histological subtypes were classified in accordance with the 2002 AJCC/UICC classification system. Detailed information is listed in Table 1. All patients were followed with clinical and radiologic examinations. The median follow-up time was 45.4 months (range, 3–94 months). The study protocol was approved by the Ethics Committee of First Affiliated Hospital of Gannan Medical University (Ganzhou, China), and written informed consent was obtained from each patient.
Real-time quantitative PCR
Total RNA was isolated from tissue specimens or cells using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s protocol. First-strand cDNAs were synthesised using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). Quantitative real-time PCR was performed using SYBR Green PCR Master Mix (Applied Biosystems) in a 7900 Real-Time PCR System (Applied Biosystems). The PCR primers used in the study were GOLPH3: 5′-GGGCGACTCCAAGGAAAC-3′ (forward) and 5′-CAGCCACGTAATCCAGATGAT-3′ (reverse) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH): 5′-ATTCCACCCATGGCAAATTC-3′ (forward) and 5′-ATTCCACCCATGGCAAATTC-3′ (reverse). Glyceraldehyde 3-phosphate dehydrogenase was used as the reference gene. Ct values of the samples were calculated, and the relative levels of GOLPH3 mRNA were analysed by the 2−ΔΔCt method.
Western blot assay
Tissues or cells were lysed in lysis buffer containing protease inhibitor cocktail. Protein concentration was determined using a Bio-Rad protein assay system (Bio-Rad, Hercules, CA, USA). Equivalent amounts of proteins were separated by SDS–PAGE, and then transferred onto polyvinylidene difluoride membranes (Bio-Rad). After being blocked in Tris-buffered saline (TBS) containing 5% non-fat milk, the membranes were incubated with specific primary antibody against GOLPH3 (1 : 1000; ProteinTech Group, Inc., Chicago, IL, USA) at 4 °C for 12 h and then with horseradish peroxidase-conjugated anti-rabbit antibody (Zhongshan, Beijing, China) for 2 h at room temperature. Proteins were visualised using ECL (Pierce, Rockford, IL, USA) and detected using BioImaging Systems (UVP Inc., Upland, CA, USA). The relative protein levels were calculated based on GAPDH as the loading control.
Immunohistochemistry staining
Immunohistochemical staining was done on 4-μm-thick sections from tissue microarrays (TMAs). The TMA was constructed as described previously (Kononen et al, 1998). Sections were dewaxed in xylene and rehydrated in grade alcohol, followed by boiling in 10 mmol l−1 of citrate buffer (pH 6.0) for antigen retrieval. After inhibition of endogenous peroxidase activities for 30 min with methanol containing 0.3% H2O2, the sections were blocked with 2% bovine serum albumin for 30 min and incubated overnight at 4 °C with primary polyclonal rabbit anti-human GOLPH3 antibody (1 : 200; ProteinTech Group, Inc.). After washing thrice with PBS, the slides were incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG for 30 min, followed by reaction with diaminobenzidine and counterstaining with Mayer’s haematoxylin. Negative control was done by omission of the primary antibody and substituting it with non-specific rabbit IgG.
For the immunohistochemical assessment, the product of the scores of staining intensity and quantity of immunoreactive tumour cells was calculated based on the following scoring system: the intensity ranged from 0=no staining, 1=weak staining, 2=moderate staining to 3=strong staining; the quantity comprised 0=no expression, 1=positivity in <10%, 2=positivity in 10–35%, 3=positivity in 35–75%, and 4=positivity in >75%. The final immunohistochemical score (IHS; ranging from 0 to 12) was obtained by multiplication of the intensity score and the quantity score. The cutoff value for high and low expression was determined on the basis of a heterogeneity value measured through log-rank statistical analysis with respect to overall survival (OS). An IHS of ⩾6 was defined as high GOLPH3 expression and an IHS of ⩽4 indicated low GOLPH3 expression. Three board-certified pathologists (HY, SP, and XH) with >10 years of experience in uropathology evaluated the immunostaining in a blinded manner without any knowledge of the clinicopathological data. In the few instances of discrepant scoring, a consensus score was determined with a multiheaded microscope. We defined ‘consensus’ when at least two reviewers were in agreement.
Cell lines
Human RCC cell lines 786-O and Caki-1 were purchased from the American Type Culture Collection (Rockville, MD, USA). Another three human RCC cell lines, A498, ACHN, and OS-RC-2, were preserved in our institute. Immortalised normal human proximal tubule epithelial cell line HK-2 was obtained from Shanghai Cell Bank (Shanghai, China). HK-2 cells were cultured in K-SFM medium (Gibco Life Technologies, Grand Island, NY, USA), and other cells were cultured in RPMI-1640 medium (HyClone Laboratories, Logan, UT, USA) with 10% fetal bovine serum (FBS), 50 U ml−1 of penicillin, and 50 μg ml−1 of streptomycin. All cells were cultured in a sterile incubator maintained at 37 °C with 5% CO2.
Gene silencing using siRNA
The siRNA targeting GOLPH3 and control siRNA were purchased from Shanghai Genepharma Co. Ltd. (Shanghai, China). The siRNA sequence for GOLPH3 was 5′-GUUA AGAAAUGUACGGGAATT-3′. Cells were transfected with either GOLPH3 or control siRNA using Oligofectamine reagent (Invitrogen) according to the manufacturer’s instructions. Following transfection, the mRNA and protein levels were assessed 48 h later.
MTT assay
Cells were plated in 96-well culture plates at about 5 × 103 cells per well 24 h after transfection. Then, 20 μl of 5 mg ml−1 MTT solution was added to each well and incubated for 4 h at 37 °C, the media was removed from each well, and the resultant MTT formazan was solubilised in 150 μl of DMSO. The absorbance values at 490 nm were measured using a microplate reader (Bio-Rad). The experiment was repeated three times and each experiment had six replicate wells.
Colony formation assay
Cells were transfected with control or GOLPH3 siRNA for 48 h and then plated at 1 × 103 cells per well of a 6-well plate in triplicate. After 14 days, the plates were stained with crystal violet, and colonies with >50 cells were counted.
Anchorage-independent colony growth assay
Cells were transfected with control or GOLPH3 siRNA for 48 h. Cells were then plated on a 0.6% agarose base in 6-well plates (1.0 × 103 cells per well) in 1 ml of RPMI-1640 medium containing 10% FBS and 0.3% agarose. After 16 days, colonies were stained with crystal violet, and colonies containing at least 50 cells were counted.
Scratch migration assay
Cells were seeded to 12-well plates and transfected with control or GOLPH3 siRNA. At 24 h after transfection, cells were scratched using the tip of a sterile 200-μl pipette in each well. The plates were washed twice with PBS to remove the detached cells, and incubated at 37 °C in 5% CO2. Wound closure was observed and measured after 24 h. The experiment was done in triplicate.
Matrigel invasion assay
Cell invasion assay was performed using a 24-well transwell chamber with a pore size of 8 μm (Costar, New York, NY, USA). The inserts were coated with 50 μl Matrigel (dilution at 1 : 2; BD Bioscience, Franklin Lakes, NJ, USA). Cells were trypsinised after transfection with control or GOLPH3 siRNA for 48 h and transferred to the upper Matrigel chamber in 100 μl of serum-free medium containing 1 × 105 cells and incubated for 24 h. The lower chamber was filled with medium containing 10% FBS as a chemoattractant. After incubation, the non-invaded cells on the upper membrane surface were removed with a cotton tip, and the cells that passed through the filter were stained using crystal violet. The numbers of invaded cells were counted in five randomly selected high power fields under a microscope. This experiment was performed in triplicate.
Cancer xenograft model and tumorigenicity assay
Four-week-old male BALB/c nude mice (Experimental Animal Center of Shanghai, Shanghai, China) were housed in a specific pathogen-free environment at the Animal Laboratory Unit, Gannan Medical University, China. Cells (1.0 × 107) were suspended in 100 μl PBS and injected subcutaneously in the right flank region of nude mice. After 2 weeks, the injected nude mice with tumour burden were randomly divided into two groups (n=6 for each group) to receive intratumoral injection of control siRNA or GOLPH3 siRNA complex every 5 days. Each complex contained 10 μg of siRNA and 7.5 μl Oligofectamine (Invitrogen) in PBS. The tumour volumes were measured two times a week with a caliper and calculated according to the formula: length × width2/2. All mice were killed humanely after five times of treatment, and the resected tumours were weighed. All experimental manipulations were undertaken in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals, with the approval of the Scientific Investigation Board of the Gannan Medical University, Ganzhou, China.
Statistical analysis
The statistical analyses were performed using the Statistical Package for the Social Sciences, version 16.0 (SPSS Inc., Chicago, IL, USA). A paired-samples t-test was used to compare GOLPH3 mRNA and protein expression in the RCC tissues with that of their paired adjacent non-tumour tissue samples. The relationship between GOLPH3 protein expression and the clinicopathological features was analysed using χ2 tests. For survival analysis, three end points (cancer relapse, cancer progression, and death) were used to calculate recurrence-free survival (RFS) for non-metastatic patients, progression-free survival (PFS) for metastatic patients, and OS for all patients, respectively. Recurrence-free survival was defined as the time interval between the date of surgery and the date of identification of disease recurrence; PFS was defined as the time interval between the date of surgery and the date of identification of progressive disease or death, and OS was defined as the time interval between the date of surgery and the date of death. The log-rank test and Kaplan–Meier method were used for survival analyses. Univariate and multivariate analyses were done according to Cox proportional hazard model. Unpaired two-tailed Student’s t-tests were used to analyse comparisons between the two groups. In all tests, a P-value of <0.05 was regarded as statistically significant.
Results
Expression of GOLPH3 mRNA and protein in RCC tissues
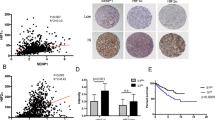
We first determined the GOLPH3 expression in 43 matched pairs of fresh RCC samples and adjacent non-tumour tissue samples. The results revealed a statistically significant elevation of GOLPH3 mRNA in tumours, as compared with the matched adjacent non-tumour tissues (P<0.001, Figure 1A). Consistent with the mRNA data, GOLPH3 protein in tumour tissues was also significantly higher than that in adjacent non-tumour tissues (P<0.001, Figure 1B). The protein level of GOLPH3 in five representative pairs of samples is shown in Figure 1C.
The expression of GOLPH3 mRNA and protein in the human RCC surgical specimens, as evaluated by real-time quantitative PCR and western blotting. (A) The relative mRNA expression of GOLPH3 was higher in 43 RCC tumour tissues than in matched adjacent non-tumour tissues (P<0.001). (B) The GOLPH3 protein expression was higher in the tumour tissues than in matched adjacent non-tumour tissues (P<0.001). (C) Expression of GOLPH3 protein in five representative pairs of RCC tissues is presented. N=non-tumour tissues; T=RCC tissues.
Immunohistochemical analysis of GOLPH3 expression in RCC samples and its relationship to clinicopathological parameters
We further analysed GOLPH3 protein level in 218 RCC tissues and 84 adjacent non-tumour tissues using an immunohistochemical approach. Representative examples of GOLPH3 staining in RCC patients are provided in Figure 2A–E. High GOLPH3 expression was observed in 116 patients (53.2%). The adjacent non-tumour tissues showed either no or very weak protein expression (Figure 1F). The association between the expression of GOLPH3 and various clinicopathological parameters is listed in Table 1. The GOLPH3 staining level significantly correlated with T stage, lymph-node status, distant metastasis, TNM stage, and Fuhman grade (all P<0.05).
Immunohistochemical staining of GOLPH3 in RCC specimens and normal renal tissues. GOLPH3 immunohistochemical staining was performed on different types of RCCs and normal renal tissues, and representative images are shown. (A) Weak staining of GOLPH3 in clear cell RCC. (B) Moderate staining of GOLPH3 in clear cell RCC. (C) Strong staining of GOLPH3 in clear cell RCC. (D) GOLPH3 staining in papillary RCC. (E) GOLPH3 staining in chromophobe RCC. (F) Extremely weak or negative GOLPH3 staining in renal tubules of normal renal tissues (original magnification: × 100 and × 400).
GOLPH3 expression and patient survival
The OS rates for patients with high GOLPH3 expression were significantly lower than that for patients with low GOLPH3 expression (Figure 3A, P<0.001). T stage, lymph-node status, distant metastasis, histopathologic subtype, Fuhman grade, and GOLPH3 expression were significant prognostic factors for OS in univariate analysis (Table 2A). By multivariable analysis, T stage (P<0.001), lymph-node status (P<0.001), distant metastasis (P=0.004), Fuhman grade (P=0.03), and GOLPH3 expression (P<0.001) were independent prognostic factors for OS (Table 2A). In non-metastasised disease (M0), the RFS rate for patients with high GOLPH3 expression was significantly lower (Figure 3B, P<0.001). T stage, lymph-node status, histopathologic subtype, Fuhman grade, and GOLPH3 expression were significant prognostic factors for RFS in univariate analysis (Table 2B). By multivariate analysis, T stage (P<0.001), lymph-node status (P<0.001), Fuhman grade (P=0.023), and GOLPH3 expression (P<0.001) were independent prognostic factors for RFS (Table 2B). In metastatic disease (M1), GOLPH3 expression was not associated with PFS (Figure 3C, P=0.403), possibly due to the limited number of patients (n=22).
We further investigated the predictive value of GOLPH3 expression within different subgroups stratified according to the T stage, TNM stage, and lymph-node status. Individuals with high GOLPH3 expression had shorter OS than those with low expression in both the T1+T2 subgroup (n=163, P=0.005; Figure 4A) and the T3+T4 subgroup (n=55, P<0.001; Figure 4B), indicating that GOLPH3 could be a valuable prognostic marker for RCC in all disease stages. Similarly, the OS was significantly shorter in patients with high GOLPH3 expression in TNM stage I+II subgroup (n=155, P=0.014; Figure 4C) and the TNM stage III+IV subgroup (n=63, P<0.001; Figure 4D), or in lymph-node status N0 (n=181, P<0.001; Figure 4E) and lymph-node status N1–2 subgroup (n=37, P=0.001; Figure 4F).
Kaplan–Meier analysis of the overall survival of RCC patients categorised according to the T stage, TNM stage, lymph-node status, and GOLPH3 expression. Statistical significance of the difference between curves of GOLPH3 high-expressing and low-expressing patients was compared in T stage T1 to T2 (A) and T stage T3 to T4 (B) patient subgroups, TNM stage I to II (C) and TNM stage III to IV (D) patient subgroups, lymph-node status N0 (E) and lymph-node status N1–2 (F) patient subgroups. P-values were calculated by the log-rank test.
GOLPH3 expression in RCC cell lines
We also detect the expression of GOLPH3 mRNA and protein in several human RCC cell lines as well as in an immortalised normal human proximal tubule epithelial cell line. As presented in Figure 5A, Caki-1, 786-O, A498, OS-RC-2, and ACHN showed higher level of GOLPH3 mRNA relative to the HK-2 normal proximal tubule epithelial cell line. Consistent with the mRNA data, GOLPH3 protein was also elevated in those RCC cell lines compared with the HK-2 cell line (Figure 5B).
Expression of GOLPH3 protein and mRNA and GOLPH3 knockdown by GOLPH3 siRNA in RCC cell lines. (A and B) GOLPH3 protein and mRNA expression detected in several human RCC cell lines (Caki-1, 786-O, A498, OS-RC-2, and ACHN) as well as in a normal proximal tubule epithelial cell line HK-2 by western blotting and real-time quantitative PCR, respectively. (C) Real-time quantitative PCR analysis of GOLPH3 mRNA expression in Caki-1 and 786-O cells transfected with the specific siRNA targeting GOLPH3 for 48 h. *P<0.01, relative to control. (D) Western blot analysis of GOLPH3 protein expression in Caki-1 and 786-O cells transfected with the GOLPH3 siRNA for 48 h. Efficient depletion of GOLPH3 expression was verified.
Effects of GOLPH3 depletion on cell growth in vitro
To further examine the functional role of GOLPH3 in RCC cells, we specifically knocked down its expression using siRNA technique in Caki-1 and 786-O cells expressing high levels of endogenous GOLPH3. We observed that GOLPH3 mRNA and protein levels were significantly reduced in cells transfected with specific siRNA for GOLPH3 compared with those transfected with control siRNA (Figure 5C and D). We next studied the impact of GOLPH3 silencing on cell proliferation in vitro. The results of the MTT assay showed that downregulation of GOLPH3 significantly reduced the proliferation rate in both the cell lines tested compared with the control siRNA-transfected cells (P<0.01; Figure 6A). Colony formation assay further showed that downregulation of GOLPH3 in two tested cell lines with transfection of GOLPH3 siRNA resulted in a clear reduction in the colony formation capacity compared with the control siRNA-transfected cells (P<0.01; Figure 6B).
Effects of GOLPH3 depletion on cell growth in vitro. (A) Inhibition of Caki-1 and 786-O cell proliferation by GOLPH3 siRNA tested by MTT assay. (B) Inhibition of Caki-1 and 786-O cells colony formation capacity by GOLPH3 siRNA. Values are mean±s.d. of three independent experiments. *P<0.01, relative to control.
Effect of GOLPH3 deletion on migration and invasion in vitro
In the scratch migration assay, downregulation of GOLPH3 significantly suppressed the migration capability of both Caki-1 and 786-O cells (Figure 7A). The migrating distance of Caki-1 cells was 294±20 μm in the control siRNA group and 138±16 μm in the GOLPH3 siRNA group (P<0.01). In the 786-O cells, the migrating distance was 296±22 μm in the control siRNA group and 115±12 μm in the GOLPH3 siRNA group (P<0.01). In addition, Matrigel invasion assay showed that downregulation of GOLPH3 significantly suppressed the invasiveness of both cancer cells (Figure 7B). The average cell counts crossing the matrigel-coated membrane in one high power field were 71±5 for the control siRNA group and 23±3 for the GOLPH3 siRNA group of Caki-1 cells (P<0.01); 62±4 for the control siRNA group and 18±4 for the GOLPH3 siRNA group of 786-O cells (P<0.01).
GOLPH3 depletion attenuated the migration and invasion capability of Caki-1 and 786-O cells in vitro. (A) Scratch migration assay showed that GOLPH3 decreased tumour cell migration. (B) Matrigel invasion assay showed that GOLPH3 siRNA-transfected cells resulted in low penetration through the Matrigel-coated membrane, compared with control cells. Values are mean±s.d. of three independent experiments. *P<0.01, relative to control.
Depletion of GOLPH3 inhibits the tumorigenicity in vitro and in vivo
Soft agar colony formation assay revealed that GOLPH3 depletion resulted in a significant decrease in the anchorage-independent growth ability of Caki-1 and 786-O cells (P<0.01; Figure 8A). To investigate whether GOLPH3 siRNA had the effect of inhibition on tumour growth in vivo, a nude mice xenograft model of RCC cell line Caki-1 was established, and intratumoral injection with GOLPH3 siRNA or control siRNA complex in the nude mice with tumour burden was performed. As shown in Figure 8B, the tumour volume of GOLPH3 siRNA group from day 22 to the end was significantly smaller than control siRNA groups (P<0.01). Furthermore, GOLPH3 siRNA resulted in a significant decrease in the tumour volume and weight as measured at the end of the experiment at day 42 when compared with control siRNA (P<0.01; Figure 8C and D). In addition, the relative protein expression of GOLPH3 in GOLPH3 siRNA group was decreased (Figure 8E).
GOLPH3 depletion inhibits the tumorigenicity of RCC cells in vitro and in vivo. (A) Inhibition of Caki-1 and 786-O cells anchorage-independent colony formation capacity by GOLPH3 siRNA. Data are presented as mean±s.d. *P<0.01, relative to control. (B) Knockdown of GOLPH3 by siRNA inhibits the growth of Caki-1-derived xenografts in nude mice. Top, macrographic image showed that the tumour size of GOLPH3 group was markedly smaller on the 42nd day after tumour cells inoculation than that of control group; bottom, growth curves of the tumour xenografts. Data are presented as mean±s.e. of tumour volumes at the indicated time points. *P<0.01, relative to control. (C and D) The final tumour volume and weight of GOLPH3 group were decreased than that of control group. (E) GOLPH3 siRNA inhibited the protein expression of GOLPH3 in vivo as determined by western blotting.
Discussion
In the present study, we clearly showed the clinicopathologic significance of the GOLPH3 expression on a relatively large cohort of RCC specimens. We examined first the GOLPH3 mRNA and protein expression in paired RCC samples. We found that GOLPH3 expression was upregulated at both the transcriptional and translational levels in most RCC tumour tissues. Consistent with these observations, immunohistochemical analysis also confirmed that tumour tissues exhibited abundant GOLPH3 expression compared with adjacent non-tumour tissues. These findings suggest the possibility that upregulated expression of GOLPH3 may provide a selective advantage in RCC tumorigenic processes.
Our immunohistochemical study also showed that high GOLPH3 expression was correlated significantly with the invasive and aggressive characteristics of RCCs, including T stage, lymph-node metastasis, distant metastasis, TNM stage, and Fuhman grade. These results indicated that the increased expression of GOLPH3 may have a role in RCC development and progression. Consistent with our findings, some previous immunohistochemical studies of GOLPH3 expression on other cancers have also revealed correlations between GOLPH3 expression and clinicopathologic characteristics. In breast cancer (Zeng et al, 2012) and oesophageal cancer (Wang et al, 2012), GOLPH3 expression was positively correlated with clinical stage, TNM classification, and histological differentiation. In gastric cancers (Hu et al, 2013), GOLPH3 expression was associated with tumour size, histological grade, depth of invasion, lymph-node metastasis, distant metastasis, and TNM stage.
High GOLPH3 expression has previously been suggested as a predictor of poor prognosis in different cancers (Hua et al, 2012; Li et al, 2012; Wang et al, 2012; Zhou et al, 2012; Zeng et al, 2012; Hu et al, 2013). In this study, the clinical significance of high GOLPH3 expression was further substantiated by its correlation with shorter OS and RFS time in RCC patients. Univariate survival analyses showed that high GOLPH3 expression was associated with the risk of death from RCC in the whole patient cohort. After excluding cases with initial metastases, high GOLPH3 expression increased the risk of RCC recurrence (n=196). More importantly, multivariate analysis also showed that GOLPH3 expression was an independent prognostic factor for both OS and RFS. Furthermore, when patients were stratified into subgroups according to T stage, TNM stage, and lymph-node status, high GOLPH3 expression also indicated a shorter OS time. Thus, GOLPH3 expression seems to have the potential to predict RCC patient outcome. The examination of GOLPH3 expression by immunohistochemistry may, therefore, be used as an additional tool in identifying those patients at risk of tumour recurrence and/or progression, and it may be a helpful criterion to optimise individual therapy management.
In attempting to determine the possibility of GOLPH3 as a therapeutic target, we employed the siRNA technique for knockdown of its expression and analysed its phenotype. We found that a transient knockdown of GOLPH3 strikingly inhibited growth and colony formation of RCC cells in vitro. Moreover, previous study (Zeng et al, 2012) has shown that the deletion of GOLPH3 expression had an antitumorigenic effect in vivo in breast cancer, and we similarly showed that the deletion of GOLPH3 expression in RCC cell lines inhibited tumour growth in vivo using siRNA technique. Our results were consistent with Zeng et al in terms of antitumour activity that the deletion of GOLPH3 expression caused in RCC. Consequently, GOLPH3 seemed to be a critical factor for the proliferation of cancer cells, and therefore, it should be a good therapeutic target for halting proliferation of RCC.
In addition, the findings presented in this study demonstrate that knockdown of endogenous GOLPH3 in RCC cell lines greatly reduces cell migration and invasion capacity in vitro, suggesting that GOLPH3 has an important role in RCC invasiveness. These results indicate that overexpression of GOLPH3 may contribute to the malignancy (invasion and metastasis) of RCC. The molecular mechanism by which GOLPH3 increases motile and invasive potential in cancer cells is not fully understood. In an in vitro study, GOLPH3 could regulate glioma cell migration and invasion through modulation of RhoA expression (Zhou et al, 2013). Although the present study could not prove the mechanism by which GOLPH3 promotes tumour invasion, our finding of a distinct association between GOLPH3 and tumour invasiveness may have therapeutic as well as prognostic implications.
There are several limitations to our study. First, our study was a single hospital-based and retrospective study. Future studies based on a multicentre or community-based prospective study with more extensive collection of potential cofounders are also required. Second, the molecular mechanisms of GOLPH3-induced metastasis in RCC need to be further elucidated. Third, the in vivo metastasis assay should be performed to further testify the roles of GOLPH3 in metastasis of human RCC.
In summary, the present study first showed that GOLPH3 expression was upregulated in the majority of the RCC clinical tissue specimens. High expression of GOLPH3 positively correlates with the aggressive phenotype of RCCs, and predicts poor survival outcome of patients. We have also presented experimental evidence that depletion of GOLPH3 in RCC cell lines using siRNA inhibited cell proliferation, anchorage-independent growth, migration, invasion, and tumour growth in xenograft model mice. On the basis of these findings, we conclude that GOLPH3 is functionally important in the development and progression of RCC and may serve as a new target for RCC therapy. In vivo experiments and further studies on the role of GOLPH3 are very interesting and are an area of active research at our institute.
Change history
29 April 2014
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Abraham RT (2009) GOLPH3 links the Golgi network to mTOR signaling and human cancer. Pigment Cell Melanoma Res 22: 378–379.
Albiges L, Oudard S, Negrier S, Caty A, Gravis G, Joly F, Duclos B, Geoffrois L, Rolland F, Guillot A, Laguerre B, Legouffe E, Kohser F, Dietrich PY, Theodore CA, Escudier B (2012) Complete remission with tyrosine kinase inhibitors in renal cell carcinoma. J Clin Oncol 30: 482–487.
Böhm M, Wieland I, Schmidt C, Rubben H, Allhoff EP (2002) Loss of heterozygosity on chromosome 5p13-12 predicts adverse prognosis in advanced bladder cancer independent of tumor stage and grade. J Urol 168: 2655–2658.
Chan DY, Marshall FF (1998) Surgery in advanced and metastatic renal cell carcinoma. Curr Opin Urol 8: 369–373.
Gorringe KL, Boussioutas A, Bowtell DD (2005) Novel regions of chromosomal amplification at 6p21, 5p13, and 12q14 in gastric cancer identified by array comparative genomic hybridization. Genes Chromosomes Cancer 42: 247–259.
Gupta K, Miller JD, Li JZ, Russell MW, Charbonneau C (2008) Epidemiologic and socioeconomic burden of metastatic renal cell carcinoma (mRCC): a literature review. Cancer Treat Rev 34: 193–205.
Hu BS, Hu H, Zhu CY, Gu YL, Li JP (2013) Overexpression of GOLPH3 is associated with poor clinical outcome in gastric cancer. Tumour Biol 34: 515–520.
Hua X, Yu L, Pan W, Huang X, Liao Z, Xian Q, Fang L, Shen H (2012) Increased expression of Golgi phosphoprotein-3 is associated with tumor aggressiveness and poor prognosis of prostate cancer. Diagn Pathol 7: 127.
Jiang Z, Chu PG, Woda BA, Liu Q, Balaji KC, Rock KL, Wu CL (2008) Combination of quantitative IMP3 and tumor stage: a new system to predict metastasis for patients with localized renal cell carcinomas. Clin Cancer Res 14: 5579–5584.
Kononen J, Bubendorf L, Kallioniemi A, Bärlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G, Kallioniemi OP (1998) Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 4: 844–847.
Lam JS, Klatte T, Kim HL, Patard JJ, Breda A, Zisman A, Pantuck AJ, Figlin RA (2008) Prognostic factors and selection for clinical studies of patients with kidney cancer. Crit Rev Oncol Hematol 65: 235–262.
Lam JS, Leppert JT, Belldegrun AS, Figlin RA (2005) Novel approaches in the therapy of metastatic renal cell carcinoma. World J Urol 23: 202–212.
Li H, Guo L, Chen SW, Zhao XH, Zhuang SM, Wang LP, Song LB, Song M (2012) GOLPH3 overexpression correlates with tumor progression and poor prognosis in patients with clinically N0 oral tongue cancer. J Transl Med 10: 168.
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Oudard S, Negrier S, Szczylik C, Pili R, Bjarnason GA, Garcia-del-Muro X, Sosman JA, Solska E, Wilding G, Thompson JA, Kim ST, Chen I, Huang X, Figlin RA (2009) Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol 27: 3584–3590.
Motzer RJ, Mazumdar M, Bacik J, Russo P, Berg WJ, Metz EM (2000) Effect of cytokine therapy on survival for patients with advanced renal cell carcinoma. J Clin Oncol 18: 1928–1935.
Nakashima-Kamimura N, Asoh S, Ishibashi Y, Mukai Y, Shidara Y, Oda H, Munakata K, Goto Y, Ohta S (2005) MIDAS/GPP34, a nuclear gene product, regulates total mitochondrial mass in response to mitochondrial dysfunction. J Cell Sci 118: 5357–5367.
Patil S, Ishill N, Deluca J, Motzer RJ (2010) Stage migration and increasing proportion of favorable-prognosis metastatic renal cell carcinoma patients: implications for clinical trial design and interpretation. Cancer 116: 347–354.
Rini BI, Campbell SC, Escudier B (2009) Renal cell carcinoma. Lancet 373: 1119–1132.
Rini BI, Rathmell WK, Godley P (2008) Renal cell carcinoma. Curr Opin Oncol 20: 300–306.
Rixe O, Bukowski RM, Michaelson MD, Wilding G, Hudes GR, Bolte O, Motzer RJ, Bycott P, Liau KF, Freddo J, Trask PC, Kim S, Rini BI (2007) Axitinib treatment in patients with cytokine-refractory metastatic renal-cell cancer: a phase II study. Lancet Oncol 8: 975–984.
Scott KL, Chin L (2010) Signaling from the Golgi: mechanisms and models for Golgi phosphoprotein 3-mediated oncogenesis. Clin Cancer Res 16: 2229–2234.
Scott KL, Kabbarah O, Liang MC, Ivanova E, Anagnostou V, Wu J, Dhakal S, Wu M, Chen S, Feinberg T, Huang J, Saci A, Widlund HR, Fisher DE, Xiao Y, Rimm DL, Protopopov A, Wong KK, Chin L (2009) GOLPH3 modulates mTOR signalling and rapamycin sensitivity in cancer. Nature 459: 1085–1090.
Siegel R, Ward E, Brawley O, Jemal A (2011) Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 61: 212–236.
Snyder CM, Mardones GA, Ladinsky MS, Howell KE (2006) GMx33 associates with the trans-Golgi matrix in a dynamic manner and sorts within tubules exiting the Golgi. Mol Biol Cell 17: 511–524.
Tomita Y, Uemura H, Fujimoto H, Kanayama HO, Shinohara N, Nakazawa H, Imai K, Umeyama Y, Ozono S, Naito S, Akaza H (2011) Key predictive factors of axitinib (AG-013736)-induced proteinuria and efficacy: a phase II study in Japanese patients with cytokine-refractory metastatic renal cell carcinoma. Eur J Cancer 47: 2592–2602.
Wang JH, Chen XT, Wen ZS, Zheng M, Deng JM, Wang MZ, Lin HX, Chen K, Li J, Yun JP, Luo RZ, Song LB (2012) High expression of GOLPH3 in esophageal squamous cell carcinoma correlates with poor prognosis. PLoS One 7: e45622.
Wu CC, Taylor RS, Lane DR, Ladinsky MS, Weisz JA, Howell KE (2000) GMx33: a novel family of trans-Golgi proteins identified by proteomics. Traffic 1: 963–975.
Yang JC, Sherry RM, Steinberg SM, Topalian SL, Schwartzentruber DJ, Hwu P, Seipp CA, Rogers-Freezer L, Morton KE, White DE, Liewehr DJ, Merino MJ, Rosenberg SA (2003) Randomized study of high-dose and low-dose interleukin-2 in patients with metastatic renal cancer. J Clin Oncol 21: 3127–3132.
Yokoi S, Yasui K, Saito-Ohara F, Koshikawa K, Iizasa T, Fujisawa T, Terasaki T, Horii A, Takahashi T, Hirohashi S, Inazawa J (2002) A novel target gene, SKP2, within the 5p13 amplicon that is frequently detected in small cell lung cancers. Am J Pathol 161: 207–216.
Zeng Z, Lin H, Zhao X, Liu G, Wang X, Xu R, Chen K, Li J, Song L (2012) Overexpression of GOLPH3 promotes proliferation and tumorigenicity in breast cancer via suppression of the FOXO1 transcription factor. Clin Cancer Res 18: 4059–4069.
Zhou J, Xu T, Qin R, Yan Y, Chen C, Chen Y, Yu H, Xia C, Lu Y, Ding X, Wang Y, Cai X, Chen J (2012) Overexpression of Golgi phosphoprotein-3 (GOLPH3) in glioblastoma multiforme is associated with worse prognosis. J Neurooncol 110: 195–203.
Zhou X, Zhan W, Bian W, Hua L, Shi Q, Xie S, Yang D, Li Y, Zhang X, Liu G, Yu R (2013) GOLPH3 regulates the migration and invasion of glioma cells though RhoA. Biochem Biophys Res Commun 433: 338–344.
Acknowledgements
We are very grateful for the sincere help and excellent technical support by the Laboratory of Cell Biology in Gannan Medical University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Xue, Y., Wu, G., Liao, Y. et al. GOLPH3 is a novel marker of poor prognosis and a potential therapeutic target in human renal cell carcinoma. Br J Cancer 110, 2250–2260 (2014). https://doi.org/10.1038/bjc.2014.124
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2014.124
Keywords
This article is cited by
-
GOLPH3 promotes endotoxemia-induced liver and kidney injury through Golgi stress-mediated apoptosis and inflammatory response
Cell Death & Disease (2023)
-
GOLPH3 high expression predicts poor prognosis in patients with resected non-small cell lung cancer: an immunohistochemical analysis
Tumor Biology (2014)
-
Overexpression of GOLPH3 protein is associated with worse prognosis in patients with epithelial ovarian cancer
Tumor Biology (2014)