Abstract
Background:
We recently showed that synthetic phosphoethanolamine reduces tumour growth and inhibits lung metastasis in vivo. Here, we investigated its anti-leukaemia effects using acute promyelocytic leukaemia (APL) as a model.
Methods:
Cytotoxic effects of Pho-s on leukaemia cells were evaluated by MTT assay. Leukaemic cells obtained from hCG-PML-RARa transgenic mice were transplanted to NOD/SCID mice. After the animals were diagnosed as leukaemic, treatment started with Pho-s using all-trans retinoid acid or daunorubicin as positive control or and saline control. Cell morphology and immunophenotyping were used to detect the undifferentiated blast cells in the spleen, liver and bone marrow. The induction of apoptosis in vitro and in malignant leukaemic clones was evaluated.
Results:
Synthetic phosphoethanolamine is cytotoxic and induces apoptosis through the mitochondrial pathway in vitro to leukaemia cell lines. In vivo Pho-s exhibits anti-proliferative effects in APL model reducing the number of CD117+ and Gr-1+ immature myeloid cells in the BM, spleen and liver. Synthetic phosphoethanolamine impairs the expansion of malignant clones CD34+/CD117+, CD34+ and Gr-1+ in the BM. In addition, Pho-s induces apoptosis of immature cells in the spleen and liver, a notable effect.
Conclusion:
Synthetic phosphoethanolamine has anti-leukaemic effects in an APL model by inhibiting malignant clone expansion, suggesting that it is an interesting compound for leukaemia treatment.
Similar content being viewed by others
Main
The primary amine phosphoethanolamine is a central precursor in the biosynthesis of membrane phospholipids. It is a precursor of phosphatidylcholine and phosphatidylethanolamine, which are both involved in the turnover of cell membranes (Ellison et al, 1987). These phospholipids take part in the lipid signalling pathways and may induce its effects either by direct stimulation of membrane receptors or through the generation of second messengers (Eyster, 2007). We recently reported that Pho-s has potent effects on a wide variety of solid tumour cell lines. Interestingly, Pho-s presents anti-proliferative and pro-apoptotic effects on melanoma cells by inducing an arrest of the cell cycle in G2/M with the loss of mitochondrial potential and increased caspase-3 activity (Ferreira et al, 2011). In addition, Pho-s showed antitumor effects in vivo on the Ehrlich ascites carcinoma and melanoma models. Most importantly, the results showed that Pho-s inhibits tumour growth and increases the lifespan of animals without causing liver or haematological toxicity (Ferreira et al, 2012).
Recently, the putative therapeutic potential of Pho-s was validated in a renal carcinoma model, on which the notable in vivo results showed that Pho-s potentially inhibits lung metastasis in nude mice. Indeed, Pho-s also inhibited endothelial cell proliferation, migration and tube formation by inducing a cell cycle arrest at the G2/M phase. It caused a decrease in cyclin D1 mRNA, VEGFR1 gene transcription and VEGFR1 receptor expression. Interestingly, using cyclosporin A, a specific inhibitor of cyclophilin and Z-VAD-fmk, a pan-caspase inhibitor, Pho-s was confirmed to induce apoptosis through the mitochondrial-dependent pathway (Ferreira et al, 2013a, 2013b).
The effect of Pho-s on leukaemic cells has not been established. Based on the fact that leukaemic cells are more sensitive than their normal counterparts to molecules that impair energy generation by mitochondria, we hypothesised that Pho-s may have anti-leukaemia effects. The sensitivity to drugs that target mitochondria (also known as mitocans) is particularly well characterised in acute promyelocytic leukaemia (APL), a subtype of subtype of acute myeloid leukaemia associated with the t(15;17)-PML/RARa gene rearrangement (Rohlena et al, 2011; dos Santos et al, 2012) Moreover, APL cells are sensitive to the differentiating effect of the all-trans retinoic acid (ATRA) and therefore constitute a good model to determine if Pho-s has differentiating action. Finally, APL cells are resistant to several pro-apoptotic stimuli and in vitro and in vivo data suggest that APL resistance to apoptosis is mediated by the fusion protein PML/RARa, which is degradated by arsenic trioxide treatment through a mechanism involving mitochondria-dependant generation of reactive oxygen species (Freitas et al, 2009). Therefore, in the present work we investigated the in vitro and in vivo cytotoxic activity of Pho-s against leukaemia, using APL as a model.
Materials and Methods
Chemical
Synthetic phosphoethanolamine was prepared according to Outhouse (1936) with purity over 99% analysed by high-performance liquid chromatography. The 1 M stock solution was diluted in water and monoethanolamine to adjust the pH to 7.2. It was stored at room temperature and diluted in phosphate buffered saline (PBS-vehicle) for the in vitro and vivo tests.
Cell culture
The KG-1 (human myeloid-ATCC CCL-246), K562 (human erythromyeloblastoid leukaemia-ATCC CCL-243) and Jurkat (human T-Cell leukaemia—Jurkat, Clone E6-1 ATCC TIB-152) cell lines were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA). All cell lines were routinely cultured in Modified Dulbecco’s Medium or RPMI supplemented with 2 mM L-glutamine and 5% (v/v) FCS and maintained at 37 °C in 95% humidified atmosphere, containing 5% CO2.
MTT colorimetric assay
Cells were plated in 96-well at a concentration of 1 × 104 cells per well. The cells were allowed to grow for 24 h then treated with Pho-s at concentrations ranging from 0.39 to 100 mM, in six replicates. After 24 h of treatment, cell viability was determined by MTT (3-(4,5 dimethylthiazol-2-yl)-2,5diphenyltetrazolium bromide) (Sigma, St Louis, MO, USA). Briefly, 20 μl of MTT solution were added to each well (final concentration, 0.5 mg ml−1) and the plate was incubated for an additional 4 h. Finally, 200 μl of DMSO were added to each well and the absorbance was measured in a plate reader (Thermoplate TP Reader, Gendoji-cho, Fujinomiya-shi Shizuoka-ken, Japan) at 540 nm. The 50% inhibitory concentration (IC50) values were calculated using a proper analysis (nonlinear regression-curve fit).
Experimental animals
Leukaemic cells obtained from hCG-PML-RARa transgenic mice were maintained at −80 °C. Before transplantation, the cells were thawed and suspended in RPMI 1640 with 10% FCS. After 12 h of sublethal cobalt irradiation with 250 cGy, 2 × 106 cells were intravenously injected in the ocular plexus of 10- to 12-week-old NOD/SCID mice (The Jackson Laboratory, Bar Harbour, ME, USA). These mice were bred and maintained under in pathogen-free conditions at the Animal Facility of the Fundação Hemocentro, Faculdade de Ciências Farmacêuticas de Ribeirão Preto, Universidade de São Paulo. In order to verify the successful transplantation, genotyping was performed by PCR analysis of genomic DNA using the pair of primers C1 and D as described elsewhere (van Dongen et al, 1999). To monitor the peripheral blood (PB) counts, mice were bled from the tail 3 weeks post-transplantation with hCG-PML/RARα. The transgenic mouse models harbouring the PML/RARα fusion were kindly provided by Professor Pier Paolo Pandolfi (Beth Israel Deaconess Medical Centre, Harvard Stem Cell Institute, Boston, MA, USA) and their generation has been described elsewhere (He et al, 1997).
Ethics statement and therapeutic protocol
The mice were randomly allocated to groups of five animals. After the animals were diagnosed as leukaemic, characterised with leukocytosis, anaemia and thrombocytopenia the treatment was started with the intraperitoneal administration of one of the following preparations: 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s and 1 mg kg−1 ATRA, 10 mg kg−1 daunorubicin (DA) (positive controls) and continued daily for 15 days. In addition, mice were treated with 100 μl PBS (vehicle) to induce the same stress conditions of treatment to in the control animals. All animal experimentation was conducted under a protocol in agreement with ethical principles in animal research and approved by the ethics committee for animal research of the Butantan Institute (process number 566/09).
Evaluation of anti-leukaemia effects
For monitoring PB, differential counts were performed using Leishman–Wright–Giemsa stain. The mice were considered leukaemic according to the following criteria: presence of blasts and promyelocytes in the PB; leukocytosis >30 × 103 per μl, haemoglobin <10 g dl−1) and platelets <500 × 103 per μl. Automated counts of theses cells were performed using a T-890 Coulter cell counter (Coulter Corporation, Hialeah, FL, USA), and differential counts were obtained from Leishman–Wright–Giemsa-stained smears. After the end of the treatments, leukaemic cells from bone marrow (BM), liver and spleen were obtained by flushing with RPMI 1640 (Gibco, Grand Island, NY, USA). The splenic and hepatic cell suspensions were obtained by mechanical disruption. Then, cells were washed once and resuspended in PBS at a concentration of 106 per ml. Cytospin slides were prepared in a Cytospin 3 Cell Preparation System (Shandon, Pittsburgh, PA, USA) and stained with Leishman–Wright–Giemsa. For morphological analysis, a minimum of 200 cells were counted and myeloid cells were classified as immature, intermediate or mature according to the Bethesda Proposals for Classification of non-lymphoid hematopoietic neoplasms in mice (Kogan et al, 2002).
Flow cytometry analysis
Cell suspensions of BM, spleen and liver were obtained as described above. Cells were first fixed and permeabilised with Triton X-100 0.02%, then stained with fluorescein isothiocyanate (FITC)-conjugated, allophycocyanin (APC), peridin chlorophyll protein (Per-CP) and phycoerythrin-conjugated monoclonal antibodies (mAb) and their respective isotype-matched controls (Southern Biotechnology Associates, Birmingham, AL, USA). The antibodies employed were CD117-APC/Alexa Fluor750 (immature myeloid cells); CD34-FITC (hematopoietic stem cells); CD45-Per-CP (pan leukocyte marker) and Gr-1-APC/Alexa Fluor750 (immature myeloid cells) (Biosciences Pharmingen, San Diego, CA, USA). For the exclusion of erythrocytes, cells were lysed immediately after labelling by incubation with 2 ml of FACS lysing solution (Becton Dickinson San Jose, CA, USA) as recommended by the manufacturer. The immunophenotype of the cells was determined by flow cytometry with FACScalibur (Becton Dickinson). A total of 10 000 cells per sample were acquired and the percentage of cells was recorder using FlowJo software.
Measurement of mitochondrial membrane potential
Leukaemia cells at density of 106 cells were seeded in 6-well plates and incubated overnight, and next day treated with Pho-s. After 12 h of treatment, the cells were washed once with PBS and stained with 100 nM Mytotracker Red for 15 min at 37 °C. Next, the cells were washed once with PBS and kept at 4 °C during measurements. The mitochondrial membrane potential (ΔΨ) was measured by flow cytometry with FACScalibur (Becton Dickinson). A total of 10 000 cells per sample were acquired for each sample in three independent experiments.
Apoptosis assay
Following the FACS analyses described above, gate regions for CD117+ and Gr-1+ cells, infiltrated in the spleen and liver, and were previously determined in the FACS for further analysis of apoptosis induced by the treatments. The cells were then centrifuged and the cell pellet was suspended at the concentration of 106cell per ml with binding buffer (100 μl). In vitro apoptotic effects of Pho-s were evaluated on KG-1, K562 and Jurkat cell lines, the cells were plated in six-well culture plates, grown overnight and treated with IC50 values measured. The apoptosis was measured every 2 h during a total of 8 h of treatment. Cells were then stained with Dead Cell Apoptosis Kit with Annexin V Alexa Fluor 488 and propidium iodide (PI) (BD Bioscience, San Jose, CA, USA) and incubated for 15 min at room temperature in the dark. After incubation, 400 μl of binding buffer were added and cells were analysed by flow cytometry with FACScalibur (Becton Dickinson) and data were processed using FlowJo software, determining the percentage of apoptotic cells. A minimum of 10 000 events were acquired for each sample analysed.
Detection of caspase-3 activity
To further evaluate the apoptosis mechanism, the caspase-3 activity involved in the early stage of apoptosis was evaluated. The leukaemia cells previously incubated overnight were treated with increasing concentrations of Pho-s from IC50 values for 8 h. Next, cells were lysed in 100 ml of 1 × lysis buffer. The lysates were collected and stored at −70 °C until analysed. Briefly, 50 ml of 1 × reaction buffer were added to each well used in the assay and incubated at 37 °C for 5 min. The caspase activity was measured using a caspase-3 fluorometric assay kit from Biovision. The plate was read in a CytoFluor II PerSeptive Biosystems (Farmington, MA, USA) fluorometric plate reader with excitation at 360 nm and emission at 460 nm.
Statistical analysis
All values were expressed as mean±s.d. Each value is the mean of at least three independent experiments in each group. For significance analysis Student’s t-tests and one-way analysis of variance were performed using GraphPad Prism 4.0 (San Diego, CA, USA). P-values P<0.05, 0.01 and 0.001 were considered significant.
Results
Cytotoxicity of Pho-s on leukaemia cell lines
To evaluate the cytotoxic effects of Pho-s on leukaemic cell lines, an MTT assay was performed after 24 h of treatment. Pho-s exhibited a potent dose-dependent cytotoxic effect on KG-1, K562 and Jurkat cells with IC50 values of 9; 6 and 12 mM, respectively. Of interest, among the screened cells, K562 was the most sensitive to Pho-s as demonstrated by the IC50 value (Figure 1).
Effects of Pho-s on the cell viability of KG-1, K562 and Jukart cells. Cells were seeded into 96-well plates at 104 cells per well and treated with Pho-s for 24 h. Cell viability was assessed by MTT assay and is expressed as the percentage of cells comparing the optical density (540 nm) of the treated cells with the optical density of the untreated cells. The data are representative of three independent experiments performed in triplicate.
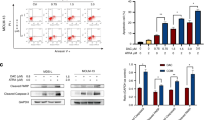
Pho-s induces in vitro apoptosis on leukaemic cell lines through the mitochondrial pathway
We evaluated whether the apoptosis is a mechanism resulting from the cytotoxic effects induced by Pho-s on leukaemic cells lines. For this purpose, we first evaluated whether Pho-s affect mitochondrial permeability transition (MPT). Our findings show that the treatment with 9 mM Pho-s to KG-1; 6 mM Pho-s to K562 and 12 mM Pho-s to Jurkat induces mitochondrial depolarisation leading to ΔΨ collapse (Figure 2A). To confirm whether the apoptotic effects of Pho-s were associated with MPT, the apoptosis was observed on leukaemic cells treated with Pho-s for different periods of time using the IC50 values calculated to each cell lines. As shown in Figure 2B after treatment with Pho-s for 8 h, a significant increase (*P<0.05) in apoptotic cells in all cell lines tested was detected with longer incubation times. Our results show that after 8 h of treatment there is an increase in apoptotic cells in response to Pho-s. This incubation time was then selected to better evaluation of caspase-3 activity. Accordingly, through the Annexin V/PI assay, we found that Pho-s causes a significant increase (*P<0.05) in caspase-3 activation in leukaemic cells after 8 h of treatment with both concentrations tested (Figure 2C). Importantly, in this study we have shown that Pho-s induces apoptosis through the mitochondrial-dependent pathway.
Pho-s induces mitochondrial membrane depolarisation and caspase-3 activity in leukemic cell lines. (A) Representative histograms of cells stained with Mitotracker red are shown. There is a substantial reduction in fluorescence emission, indicating a decrease of ΔmΨ in the leukaemic cells treated with Pho-s (black line) as compared with untreated cells (red line). (B) A substantial increase in apoptotic cells was observed after 8 h of treatment with Pho-s. (C) The increase in caspase-3 activity correlates with the increase in the frequency of apoptotic cells evaluated after the treatment. The data are representative of three independent experiments performed in triplicate. Significant differences are indicated as: *P<0.05 statistically different between Pho-s versus untreated.
Evaluation of the APL transplant model
To monitor the progression of the disease, transplanted mice were bled from the tail 15 days after transplantation. The haematological analysis reveals that the transplant was successful as evidenced by the leukocytosis (12.8±0.73%) anaemia (6.2±0.8%) and thrombocytopenia (488±45.9%) observed in the PB of the animals (Figure 3A). In addition, four animals were euthanised and leukaemia cells infiltrated in the BM, spleen and liver were analysed. Consistent with these haematological changes in PB, there was an increase in the number of blast cells in the spleen (17.5±3.5%), liver (14.0±2.8%) and BM (68.5±2.1%) (Figure 3B).
Haematological and cytological analysis of leukaemic cells. To confirm the success of the transplant, the mice were bled from the tail 15 days after transplantation. (A) The increases in the total number of leukocytes, as well as thrombocytopenia and anaemia, are evidences of the progression of the disease. (B) An increase in the number of undifferentiated cells, characteristic of blast cells, infiltrated in the bone marrow, spleen and liver was observed.
Pho-s induces a haematological response in PML-RARa leukaemic mice
We performed a cyto-morphological analysis to evaluate the antitumor effects of Pho-s in the transgenic model. An important effect observed during the leukaemic phase was an increase in the number of white blood cells of the NOD/SCID control mice, which was accompanied by an increase in the number of immature cells in the BM, PB, spleen and liver. Of note, the immature cells observed in the leukaemia model are a typical characteristic of human promyelocytes. The treatment of the mice with either Pho-s, at 40 or 80 mg kg−1, 1 mg kg−1 ATRA or 10 mg kg−1 DA (positive controls) reduced the percentage of white blood and immature cells. It is interesting to note that Pho-s and ATRA were both more efficient than DA in normalising the number of mature cells in the PB, BM spleen and liver (Table 1).
Anti-leukaemic effects of Pho-s correlate with the reduction of CD34+ CD117+ cells in the in vivo model of APL
We next evaluated the phenotype of cells infiltrated in the parenchyma. As expected, there was an increase of CD117+ and Gr-1+ cells infiltrated in the spleen and liver of the mice treated with vehicle and compared with wild-type mice (WT). Interestingly, we found that Pho-s at both 40 and 80 mg kg−1 (15±2.8%), (19.5±2.1%), respectively, induces a decrease in the number of CD117+ cells present in the spleen. In the liver, the treatment with 40 and 80 mg kg−1 of Pho-s (36±5.6%), (39±3.5%) and 1 mg kg−1 ATRA (53±1.4%) induced a reduction of CD117+ cells (**P<0.01) Figure 4A. Both CD117+ and Gr-1+ cells are frequently found in the BM, spleen, liver and lymphoid organs of patients with myelodysplastic syndromes and in tumour-bearing animals. We also observed a decrease of immature myeloid Gr-1+ cells in the spleen of mice treated with 40 and 80 mg kg−1 of Pho-s (48.5±2.0%), (47±4.2%) and 1 mg kg−1 ATRA (47.3±1.6%). In addition, the reduction of immature myeloid Gr-1+ cells in the liver of mice treated with 40 and 80 mg kg−1 of Pho-s (22.5±2.1%), (30±2.8%) and 1 mg kg−1 ATRA (19.5±3.5%) Figure 4B.
Immunophenotyping by flow cytometry of CD117+ and Gr-1+ cells infiltrated in the spleen ( A ) and liver ( B ) of NOD/SCID leukaemic mice. The bar diagram shows the percentage of CD117+ and Gr-1+cells in the parenchyma from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls) and WT mice. The data are expressed as mean ±s.d. from five mice. *P<0.05; **P<0.01 and ***P<0.001 versus untreated mice, One-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
We then investigated whether Pho-s has effects against the malignant clones in the principal hematopoietic compartment, the BM. Cells were selected as the percentage of CD45+ cells to exclude mature erythrocytes, and cell subsets were generated and evaluated for the expression of CD117, CD34 markers and also for the asynchronous CD117/34 co-expression. We did not observe changes in CD117+ and CD45+/CD117+ expression as a result of any of the treatments when compared with vehicle and WT. However, the treatment with 40 mg kg−1 Pho-s (23.1±2.4%) or 10 mg kg−1 DA (20.7±2.5%) increased the percentage of CD45+ cells (Figure 5A). The treatment with 1 mg kg−1 ATRA or Pho-s 40 mg kg−1 (2.9±0.7%), (5.1±0.9%), respectively, induced a reduction in the number of CD34+ hematopoietic progenitor cells as well as of CD34+/45+cells (43.5±2.2% to 1 mg kg−1 ATRA) and (38.2±1.4% to Pho-s 40 mg kg−1) (Figure 5B). The oncogene CD117 encodes a receptor protein present on CD34+ cells and constitutes a specific phenotype observed in acute myelogenous leukaemia. Therefore, the reduction of these two asynchronous antigens, CD34+/CD117+ in response to treatment with 40 or 80 mg kg−1 of Pho-s (40.4±2.8), (69±1.4%), or 1 mg kg−1 ATRA (14.6±2.5), respectively constitute a notable antitumor effect (Figure 6A).
Immunophenotyping by flow cytometry of CD45+, CD117+ and CD34+ cells infiltrated in the BM of NOD/SCID leukaemic mice. The bar diagram shows CD45 and CD117 (A) and CD34 (B) expression in the BM from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls). The data are expressed as mean ±s.d. from five mice. *P<0.05; **P<0.01 and ***P<0.001 versus untreated mice, one-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
Immunophenotyping by flow cytometry of CD117+ and CD34+ cells infiltrated in BM of NOD/SCID leukaemic mice. The bar diagram shows CD117 and CD34 expression in the BM from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls). The data are expressed as mean ±s.d. from five mice. **P<0.01 and ***P<0.001 versus untreated mice, one-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
Pho-s induces antitumor effects by induction of apoptosis of CD117+ and Gr-1+ cells infiltrates in the spleen and liver
To further examine the antitumor effects of Pho-s, we evaluated whether apoptosis is involved in the process. For this purpose, the proportion of dead cells in the liver and spleen was assessed by Annexin V/PI double staining. Populations of CD117+ and Gr-1+ cells selected at the dot plot gates were evaluated. The apoptotic values were estimated as a percentage of the total number of CD117+ and Gr-1+ cells for each treatment. On the other hand, the fact that the number of cells labelled according to their respective antigens can vary in response to each different treatment, the percentage of apoptotic cells is shown relative to the number of CD117+ and Gr-1+ previously selected. In accordance with the reduction of CD117+ cells in the spleen, 40 mg kg−1 of Pho-s induced apoptosis of these cells (Figure 7A and B). In addition, a significant number of CD117+ cells died by apoptosis in the liver in response to treatment with Pho-s at the concentrations of the 40 and 840 mg kg−1 (Figure 8A and B). Of note, Pho-s was more potent than ATRA e DA in the induction of apoptosis of CD117+ cells infiltrated in the spleen and liver. Apoptosis induced by Pho-s was also evaluated in Gr-1+ cells from the same parenchyma. The treatments with 40 or 80 mg kg−1 of Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA induce apoptosis of immature myeloid Gr-1+ cells present in the spleen (Figure 9A and B) and liver (Figure 10A and B).
Evaluation of cell death by apoptosis using annexin V-FITC/PI staining. (A) Dot plot profile of cells from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls). The apoptotic cells selected at the gate to CD117+ infiltrates in the spleen were stained with V-FITC/PI and analysed by flow cytometry. (B) Left panel: The percentage of apoptotic CD117+ cells is shown in the bar diagram as mean ±s.d. from five mice. Right panel: The percentage of apoptotic cells is shown in relation to the total number of CD117+ infiltrates in the spleen. The values were ***P<0.001 versus untreated mice, one-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
Evaluation of cell death by apoptosis using annexin V-FITC/PI staining. (A) Dot plot profile of cells from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls). Apoptotic cells selected at the gate for CD117+ infiltrates in the liver were stained with V-FITC/PI and analysed by flow cytometry. (B) Left panel: The percentage of apoptotic CD117+ cells is shown in the bar diagram as mean ±s.d. from five mice. Right panel: The percentage of apoptotic cells is shown in relation to the total number of CD117+ infiltrates in the liver. The values were **P<0.001 versus untreated mice, one-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
Evaluation of cell death by apoptosis using annexin V-FITC/PI staining. (A) Dot plot profile of cells from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls). Apoptotic cells selected at the gate for Gr-1+ infiltrates in the spleen were stained with V-FITC/PI and analysed by flow cytometry. (B) Left panel: The percentage of apoptotic Gr-1+ cells is shown in the bar diagram as mean ±s.d. from five mice. Right panel: The percentage of apoptotic cells is shown in relation to the total number of Gr-1+ infiltrates in the spleen. The values were ***P<0.001 versus untreated mice, one-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
Evaluation of cell death by apoptosis using annexin V-FITC/PI staining. (A) Dot plot profile of cells from untreated mice and from mice treated with 40 mg kg−1 Pho-s, 80 mg kg−1 Pho-s, 1 mg kg−1 ATRA or 10 mg kg−1 DA (the last two, positive controls). The percentage of apoptotic cells selected into the gate to Gr-1+ infiltrates in the liver were stained with V-FITC/PI and analysed by flow cytometry. (B) Left panel: The percentage of apoptotic Gr-1+ cells is shown in the bar diagram as mean±s.d. from five mice. Right panel: The percentage of apoptotic cells is shown in relation to the total number of Gr-1+ infiltrates in the liver. The values were ***P<0.001 versus untreated mice, one-way analysis of variance (ANOVA) with Tukey–Kramer post-hoc test.
Discussion
In this study, we demonstrate for the first time that Pho-s has antitumor activity against leukaemia cells. Thus, the Pho-s shows antitumor activity not only against solid tumours but also against haematological malignancies. It is interesting to note that Pho-s produced cytotoxic effects on these cells independently of specific characteristics, such as resistance to apoptosis and chemotherapy. As we demonstrated that Pho-s is cytotoxic to leukaemic cell lines, we investigated the underlying mechanisms. Here, we show that Pho-s triggers cell death by apoptosis through the mitochondrial-dependent pathway. Interestingly, our results indicate that the apoptotic effects of Pho-s most likely involve the disruption of ΔΨ combined with an increase of MPT and the release of pro-apoptotic members from the mitochondria, which induces an increase in caspase-3 activity leading to phosphatidylserine externalisation. It is in agreement with our recent report that showed that Pho-s induces apoptosis via the intrinsic pathway in murine renal carcinoma (Renca cells) and in human breast cancer (MCF-7 cells) (Ferreira et al, 2013a, 2013b). Taken together, our results show that Pho-s has in vitro pro-apoptotic activity against leukaemic cell lines.
Our in vivo study was carried out on a well-established transgenic APL model which develops leukaemia 14 days after transplantation (de Figueiredo-Pontes et al., 2011). Our results are in accordance with this well-established transgenic model, which in the leukaemic phase is characterised by a marked increase in white blood cells and thrombocytopenia. After the animals were diagnosed in the acute phase, the anti-leukaemia effects of Pho-s were evaluated. Exploring the antitumor effects, we first demonstrated that Pho-s induces a decrease in blast cells and leukocytes in the PB. This finding, while preliminary, suggests that Pho-s exhibits anti-proliferative effects in APL blocking the dissemination of malignant clones. In accordance with these results, 40 and 80 mg kg−1 of Pho-s reduced the leukaemic progenitors (immature cells). To the best of our knowledge and based on the literature, this is strong evidence that Pho-s has anti-leukaemia effects. These results consistently indicate that Pho-s blocks the capacity of malignant clones to acquire self-renewal capacity and consequently act as malignant stem cells (Stuart et al, 2009). Indeed, Pho-s inhibits malignant clone expansions, which could subsequently originate undifferentiated blast cells, contributing to the maintenance of the disease (Minami et al, 2012).
To further evaluate how Pho-s inhibits the progression of leukemogenesis, we performed an immunophenotypic analysis. One unanticipated finding was the decrease, in the spleen, CD117+ cells. CD117 is a c-kit proto-oncogene that encodes a transmembrane tyrosine kinase receptor involved in the proliferation of leukaemic cells (Rossi et al, 2013). Interestingly, this reduction was observed only upon treatment with 40 and 80 mg kg−1 of Pho-s. It suggests in turn that Pho-s has a good biodistribution profile, allowing it to act as an antitumor compound against tumour cell infiltrates in different organs. Notably, Pho-s, 1 mg kg−1 ATRA and 10 mg kg−1 DA also reduce the population of CD117+ in the liver, which can result in a normal progression and differentiation of the myeloid lineage. Importantly, Gr-1+ immature myeloid cells, generally present in patients and animals with tumours (Li et al, 2004) were suppressed by treatment with Pho-s, ATRA and DA. Thus far, these results suggest that Pho-s impaired the malignant clone expansion and proliferation. We then investigated whether apoptosis is a mechanism involved in the elimination of the malignant clones. Our results clearly show that the anti-leukaemia effects of Pho-s can be attributed to the induction of apoptosis especially of cells CD117 an antigen present is the early stages of myeloid differentiation and Gr-1+ immature myeloid cells. In this regard, we recently demonstrated that Pho-s induces apoptosis in carcinoma renal murine in a mitochondrial-dependent manner (Ferreira et al, 2013a, 2013b).
The expression of the CD34+ and CD117+ antigens on the t(15; 17) constitute the phenotype used as a marker for the diagnosis and prognosis of APL. It is also interesting to note that Pho-s causes the reduction in the BM of asynchronous antigen expression CD34+/CD117+ and it is characteristically observed in acute myelogenous leukaemia (Santana et al, 2006). In addition, Pho-s inhibits the increase of malignant clones in the BM compartment by causing a reduction of CD34+ and Gr-1+ immature myeloid cells (Li et al, 2012). The major importance of these results is that Pho-s inhibits the growth, differentiation and clonal expansion of malignant cells. In this study, the therapeutic effects of Pho-s are equivalent to those of ATRA, a potent granulocytes differentiator, which induces high rates of complete remission, even in cases resistant to chemotherapy (Gausdal et al, 2013).
In summary, we found that Pho-s is cytotoxic in vitro to leukaemia cell lines. Most importantly, we demonstrate several anti-leukaemic affects in vivo: Pho-s exhibits anti-proliferative effects in APL by blocking the dissemination of malignant clones; it reduces the number of CD117+ cells and Gr-1+ immature myeloid cells in the BM, spleen and liver. Pho-s impairs the expansion of malignant clones CD34+/CD117+ as well as CD34+ and Gr-1+ in the BM. In addition, Pho-s induces apoptosis of immature cells in the spleen and liver, which is a remarkable effect. Overall, Pho-s is a compound that shows anti-leukaemic effects in an APL model by inhibiting malignant clone expansion, suggesting that it is an interesting compound for an anti-leukaemia treatment.
Change history
26 November 2013
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
de Figueiredo-Pontes LL, Assis PA, Santana-Lemos BA, Jácomo RH, Lima AS, Garcia AB, Thomé CH, Araújo AG, Panepucci RA, Zago MA, Nagler A, Falcão RP, Rego EM (2011) Halofuginone has anti-proliferative effects in acute promyelocytic leukaemia by modulating the transforming growth factor beta signaling pathway. PLoS One 10: e26713.
dos Santos GA, Abreu e Lima RS, Pestana CR, Lima AS, Scheucher PS, Thomé CH, Gimenes-Teixeira HL, Santana-Lemos BA, Lucena-Araujo AR, Rodrigues FP, Nasr R, Uyemura SA, Falcão RP, de Thé H, Pandolfi PP, Curti C, Rego EM (2012) (+)α-Tocopheryl succinate inhibits the mitochondrial respiratory chain complex I and is as effective as arsenic trioxide or ATRA against acute promyelocytic leukaemia in vivo. Leukaemia 26: 451–460.
Ellison DW, Beal MF, Martin JB (1987) Phosphoethanolamine and ethanolamine are decreased in Alzheimer’s disease and Huntington’s disease. Brain Res 417: 389–392.
Eyster KM (2007) The membrane and lipids as integral participants in signal transduction: lipid signal transduction for the non-lipid biochemist. Adv Physiol Educ 31: 5–16.
Ferreira AK, Meneguelo R, Neto SC, Chierice OG, Maria DA (2011) Synthetic phosphoethanolamine induces apoptosis through caspase-3 pathway by decreasing expression of Bax/Bad protein and changes cell cycle in melanoma. J Cancer Sci Ther 3: 053–059.
Ferreira AK, Meneguelo R, Pereira A, Mendonca-Filho OR, Chierice GO, Maria DA (2012) Anticancer effects of synthetic phosphoethanolamine on Ehrlich ascites tumor: an experimental study. Anticancer Res 32: 95–104.
Ferreira AK, Freitas VM, Levy D, Ruiz JL, Bydlowski SP, Rici RE, Filho OM, Chierice GO, Maria DA (2013a) Anti-angiogenic and anti-metastatic activity of synthetic phosphoethanolamine. PLoS One 8: e57937.
Ferreira AK, Meneguelo R, Pereira A, Filho OM, Chierice GO, Maria DA (2013b) Synthetic phosphoethanolamine induces cell cycle arrest and apoptosis in human breast cancer MCF-7 cells through the mitochondrial pathway. Biomed Pharmacother S0753-3322 (13): 00015–2.
Freitas RA, Silva dos Santos GA, Gimenes Teixeira HL, Scheucher PS, Lucena-Araujo AR, Lima AS, Abreu e Lima RS, Garcia AB, Jordão AA Jr, Falcão RP, Vannucchi H, Rego EM (2009) Apoptosis induction by (+)alpha-tocopheryl succinate in the absence or presence of all-trans retinoic acid and arsenic trioxide in NB4, NB4-R2 and primary APL cells. Leuk Res 33: 958–963.
Gausdal G, Wergeland A, Skavland J, Nguyen E, Pendino F, Rouhee N, McCormack E, Herfindal L, Kleppe R, Havemann U, Schwede F, Bruserud O, Gjertsen BT, Lanotte M, Ségal-Bendirdjian E, Døskeland SO (2013) Cyclic AMP can promote APL progression and protect myeloid leukemia cells against anthracycline-induced apoptosis. Cell Death Dis 28: e516.
He LZ, Tribioli C, Rivi R, Peruzzi D, Pelicci PG, Soares V, Cattoretti G, Pandolfi PP (1997) Acute leukemia with promyelocytic features in PML/RARalpha transgenic mice. Proc Natl Acad Sci 94: 5302–5307.
Kogan SC, Ward JM, Anver MR, Berman JJ, Brayton C, Cardiff RD, Carter JS, de Coronado S, Downing JR, Fredrickson TN, Haines DC, Harris AW, Harris NL, Hiai H, Jaffe ES, MacLennan IC, Pandolfi PP, Pattengale PK, Perkins AS, Simpson RM, Tuttle MS, Wong JF, Morse HC 3rd Hematopathology subcommittee of the Mouse Models of Human Cancers Consortium (2002) Bethesda proposals for classification of nonlymphoid hematopoietic neoplasms in mice. Blood 100: 238–245.
Li Q, Pan PY, Gu P, Xu D, Chen SH (2004) Role of immature myeloid Gr-1+ cells in the development of antitumor immunity. Cancer Res 64: 1130–1139.
Li YF, Deng Z, Din BH, Zhu JB (2012) Effect of homoharringtonine on bone marrow CD34+CD117+cells in patients with chronic myelogenous leukemia. Leuk Lymphoma 53: 934–939.
Minami Y, Abe A, Minami M, Kitamura K, Hiraga J, Mizuno S, Ymamoto K, Sawa M, Inagaki Y, Miyamura K, Naoe T (2012) Retention of CD34+ CML stem/progenitor cells during imatinib treatment and rapid decline after treatment with second-generation BCR-ABL inhibitors. Leukemia 9: 2142–2143.
Outhouse EL (1936) Amino-ethyl phosphoric ester from tumours. Biochem J 30: 197–201.
Rohlena J, Dong LF, Ralph SJ, Neuzil J (2011) Anticancer drugs targeting the mitochondrial electron transport chain. Antioxid Redox Signal 15: 2951–2974.
Rossi G, Nomdedéu Guinot JF, Fontana A, Minervini MM, García-Dabrio MC, Cascavilla N (2013) CD117-CD15 in acute myeloid leukemia: no role as LAIP in the study of minimal residual disease. Eur J Haematol 90: 171–174.
Santana BA, Pintão MC, Abreu e Lima RS, Scheucher PS, Santos GA, Garcia AB, Falcão RP, Rego EM (2006) Asynchronous expression of myeloid antigens in leukaemic cells in a PML/RARalpha transgenic mouse model. Braz J Med Biol Res 39: 615–620.
Stuart SA, Minami Y, Wang JY (2009) The CML stem cell: evolution of the progenitor. Cell Cycle 9: 1338–1343.
van Dongen JJ, Macintyre EA, Gabert JA, Delabesse E, Rossi V, Saglio G, Gottardi E, Rambaldi A, Dotti G, Griesinger F, Parreira A, Gameiro P, Diáz MG, Malec M, Langerak AW, San Miguel JF, Biondi A (1999) Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia. Leukemia 13: 1901–1928.
Acknowledgements
This work was supported by a Sao Paulo Research Foundation—(FAPESP) Grant (2007/50571-3; Doctorate and Master Fellowship 2008/56089-1; 2010/50220-9).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Ferreira, A., Santana-Lemos, B., Rego, E. et al. Synthetic phosphoethanolamine has in vitro and in vivo anti-leukemia effects. Br J Cancer 109, 2819–2828 (2013). https://doi.org/10.1038/bjc.2013.510
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2013.510
Keywords
This article is cited by
-
Novel inhibitor of hematopoietic cell kinase as a potential therapeutic agent for acute myeloid leukemia
Cancer Immunology, Immunotherapy (2022)
-
(–)-Epigallocatechin-3-gallate induces apoptosis and differentiation in leukaemia by targeting reactive oxygen species and PIN1
Scientific Reports (2021)
-
Modulation of pro-apoptotic effects and mitochondrial potential on B16F10 cells by DODAC/PHO-S liposomes
BMC Research Notes (2018)
-
Antiproliferative and proapoptotic effects of DODAC/synthetic phosphoethanolamine on hepatocellular carcinoma cells
BMC Pharmacology and Toxicology (2018)