Abstract
Background:
High-dose chemotherapy with autologous stem cell transplantation is a cornerstone in the first-line treatment of multiple myeloma patients. However, only few factors have been identified affecting the outcome in such patients. We hypothesised that varying levels of mobilised CD34+ cells confer prognostic information in myeloma patients undergoing high-dose chemotherapy.
Methods:
We determined circulating CD34+ cells at the day of peripheral stem cell collection in 158 consecutive myeloma patients between January 2001 and August 2010. Patients were stratified into two groups (super vs normal mobilisers) with a cutoff of 100 000 peripheral CD34+ cells per ml.
Results:
We found that patients with more than 100 000 peripheral CD34+ cells per ml had a better overall survival (P=0.005) and a prolonged time to progression (P=0.0398) than patients with CD34+ cell counts below 100 000 CD34+ cells per ml. High levels of CD34+ cells were an independent marker for better overall survival and time to progression in a multivariate analysis that included disease stage, response at transplant, light-chain subtype, age, sex, and height.
Conclusion:
Our results suggest that high levels of mobilised peripheral CD34+ cells are associated with favourable outcome in myeloma patients undergoing autologous transplantation.
Similar content being viewed by others
Main
A variety of factors have been reported to affect prognosis in patients with multiple myeloma including cytogenetic abnormalities, molecular markers, cytokine profiling, and clinical parameters (Kyle and Rajkumar, 2009; Rajkumar, 2009; Barlogie et al, 2010; Dingli and Rajkumar, 2010; Munshi et al, 2011). In particular, clinical features such as high lactate dehydrogenase levels, IgA subtype, presence of extramedullary disease, renal failure, high levels of serum-free light chains or of the serum κ/λ free light-chain ratio, plasmablastic disease, or presentation as primary plasma cell leukaemia have been identified to confer unfavourable prognostic information (Munshi et al, 2011). Accordingly, risk stratification models have been established in multiple myeloma (Greipp et al, 2005; Rajkumar and Kyle, 2005; Kyle and Rajkumar, 2009; Rajkumar, 2010).
With growing insights into the genetic heterogeneity of multiple myeloma, additional prognostic factors have been proposed allowing stratification of myeloma patients into different risk categories, possibly paving the way towards a more risk-adapted therapeutic approach (Rajkumar, 2009). Such an evolving risk assessment can be based on molecular and cytogenetic abnormalities detected by conventional karyotyping, fluorescent in situ hybridisation, and/or gene expression profiling (Fermand et al, 2005; Greipp et al, 2005; Rajkumar and Kyle, 2005; Kyle and Rajkumar, 2009; Chang et al, 2010; Hose et al, 2011; Munshi et al, 2011).
High-dose chemotherapy followed by autologous stem cell transplantation is a cornerstone within the current standard treatment for symptomatic myeloma patients fit for intensive treatment (Child et al, 2003; Terpos et al, 2003; Barlogie et al, 2004; Blade et al, 2005; Fermand et al, 2005; Rajkumar and Kyle, 2005; Wang et al, 2007; Palumbo et al, 2009; Rajkumar, 2009; Lonial, 2010; Cavo et al, 2011). In fact, a number of studies have established the benefit of autologous transplantation for myeloma patients in prolonging the time to progression and, at least in some of them, also in improving overall survival (Attal et al, 1996; Child et al, 2003; Barlogie et al, 2004; Blade et al, 2005; Cavo et al, 2011).
In this retrospective study, we investigated the level of circulating CD34+ cells at the day of peripheral stem cell collection as a prognostic marker in myeloma patients. We hypothesised that excellent stem cell mobilisation is associated with an intact bone marrow homeostasis and thus confers favourable prognostic information. In fact, we found that levels of circulating CD34+ cells below 100 000 per ml at the day of stem cell collection were associated with shorter time to progression and overall survival.
Study design
Patients
A total of 158 consecutive myeloma patients underwent stem cell collection with subsequent autologous stem cell transplantation as a component of their first-line treatment between January 2001 and August 2010 at the Department of Medical Oncology, University Hospital in Bern, Switzerland. Clinical characteristics at diagnosis and mobilisation, regimens used for induction and mobilisation, and the course of the disease of the study population are summarised in Table 1 and Supplementary Figure S1. All patients had G-CSF in addition to chemotherapy for mobilisation. Chemotherapy was high-dose cyclophosphamide until December 2005 and vinorelbine since January 2006 (Bargetzi et al, 2003). No patient received a CXCR4 antagonist, and no CD34 selection was performed.
Statistical analysis
Patients were stratified into one group with more than 100 000 peripheral CD34+ cells per ml (super mobilisers), and a group with less than 100 000 circulating CD34+ cells per ml (normal mobilisers) at the day of apheresis. Overall survival was defined as the time from the day of stem cell harvest until death or last follow-up whichever occurred first. The time until first progression was the time from the day of apheresis until first progression or death, whichever occurred earlier, or until last follow-up if the patient remained in remission. Curves depicting overall survival and time to progression were performed using the Kaplan–Meier method. The survival analysis was performed using log-rank test. To evaluate the effects of parameters on outcome, the two groups were compared using the χ2-test or Fisher's exact test, and differences in the mean values in case of continuous variables were tested using t-test. The Cox proportional hazard regression was applied to analyse various risk factors on survival. Results were considered significant if the P-value was below 0.05. All statistical analyses and graphs were performed using graph pad prism program 5.04 (1992–2010; GraphPad Software, Inc., La Jolla, CA, USA) and Statview 5.0.1 (SAS Institute, Cary, NC, USA).
Results
A total of 158 consecutive myeloma patients undergoing autologous transplantation during their first-line treatment were stratified into two groups based on the level of circulating peripheral CD34+ cells at the day of stem cell collection. In all, 69 patients (super mobilisers) had more than 100 000 CD34+ cells per ml, whereas 89 patients had less than 100 000 CD34+ cells per ml (normal mobilisers). The individual values of all patients are depicted in a Supplementary Figure S2. The two groups showed no differences with regards to sex, age, light-chain subtype, stage at diagnosis, cytogenetics, height, weight, radiotherapy before stem cell collection, response to induction treatment, single vs tandem transplantation, time between apheresis and transplantation, or regimens used for induction or mobilisation (Table 1). The mean number of circulating CD34+ cells at the day of stem cell collection in the super mobiliser group was 179 609 CD34+ cells per ml, and 44 381 CD34+ cells per ml in the normal mobiliser group. The total number of CD34+ cells collected at apheresis was higher in the super mobiliser group (18.17 × 106 per kg vs 10.37 × 106 per kg; P=<0.0001), and patients in the super mobiliser group received more CD34+ cells at autologous transplantation (5.27 × 106 per kg vs 3.56 × 106 per kg; P=0.0055).
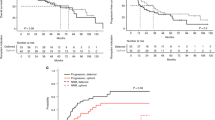
After a mean follow-up of 32.46 months, 42 patients have died, with 12 deaths occurring in the super mobiliser and 30 in the normal mobiliser group (P=0.0289). The median overall survival of all myeloma patients was 72 months (Supplementary Figure S1). Although the group of super mobilisers did not yet reach the median survival, the group of normal mobilisers had a median survival of 50 months (P=0.0050; Figure 1).
Better overall survival and longer time to progression in super mobiliser (n=69) vs normal mobiliser myeloma patients (n=89). Kaplan–Meier curves are depicted for overall survival (A) and time to progression (B) comparing the super mobiliser group (continued line) with the group of normal mobilisers (dotted line). A value of 100 000 CD34+ cells per ml circulating at the day of stem cell collection was used to stratify between the two groups. x axis in months, y axis depicts percent survival. (A) Median OS in the super mobiliser group was not reached; median OS in the normal mobiliser group was 50 months. (B) A total of 70 patients had a progression, including 27 patients in the super mobiliser group with a median TTP of 46 months compared with 43 patients in the normal mobiliser group with a median TTP of 33 months.
A total of 70 patients in our cohort had a first progression of their disease after autologous transplantation, with 27 patients progressing in the super mobiliser and 43 patients in the normal mobiliser group. The group of super mobilisers showed a longer time to progression, with a median time to progression of 46 months compared with 33 months in the normal mobiliser group (P=0.0398).
The favourable effect of high levels of circulating CD34+ cells at the day of stem cell collection was observed independent from the type of chemotherapy regimen used for induction, and it was also independent from the chemotherapy (cyclophosphamide or vinorelbine) used for mobilisation (data not shown). Better OS and TTP in the group of super mobilisers were also observed across the ISS stages, with the favourable effect reaching significance (P=0.0039 and P=0.011) for patients with ISS stage III at diagnosis (data not shown). We did not observe that the number of CD34+ cells infused at transplantation-affected OS or TTP, with P=0.754 and P=0.899, respectively, for patients below vs above the mean value of infused CD34+ cells (data not shown).
In a multivariate analysis, the level of circulating CD34+ cells turned out to be an independent prognostic factor for OS (P=0.0011) and TTP (P=0.0228). This multivariate analysis also included light-chain subtype, sex, age, height, and disease stage at diagnosis, as well as the type of response at transplant (Table 2).
Discussion
To our knowledge, this is the first report identifying varying levels of circulating CD34+ cells at the day of stem cell collection to be a prognostic marker in myeloma patients. Although previous studies indicated that patients with various lymphoid malignancies mobilising large numbers of CD34+ cells (‘super mobilisers’) enjoy improved survival following autologous stem cell transplantation (Stockerl-Goldstein et al, 2000; Gordan et al, 2003; Bolwell et al, 2007; Hiwase et al, 2008), such data are lacking so far for myeloma patients. A small study including 39 myeloma patients found no difference in outcome (Kakihana et al, 2010). As the two groups of super vs normal mobilisers in our cohort did not differ in clinical characteristics, we can exclude one or several of such parameters, to have affected the conclusion of this analysis. With regards to the retrospective character of this study, we consider a prospective evaluation of the effect of levels of mobilised CD34+ cells on outcome to be desirable, and we are in the process of initiating such a study.
The reason for the better clinical course of myeloma patients with large numbers of circulating CD34+ cells at the day of stem cell collection remains to be elucidated. One hypothesis is that patients with a high number of circulating CD34+ cells might have ‘intact’ stem cell niches with conservation of the number of stem cells and their regulation of self-renewal and differentiation (Scadden, 2006). This intact stem cell niche status might enable such patients to mobilise large numbers of CD34+ cells during the stem cell stimulation procedure (Wilson and Trumpp, 2006). Bone marrow infiltration by malignant plasma cells at diagnosis or at stem cell collection might serve as a surrogate marker for altered stem cell homeostasis. However, we observed no difference in the mean bone marrow infiltration between the groups of super vs normal mobilisers (data not shown).
Another factor possibly affecting the conclusion of this study is the number of CD34+ cells used at autologous transplantation. Patients in the super mobiliser group in this study received higher numbers of CD34+ cells during autologous transplantation (P=0.0055). In fact, the composition of the infused cellular products, such as the number of lymphocytes and/or dendritic cells, has been reported to affect the outcome in allogenous transplantation in patients with lymphoid malignancies (Bolwell et al, 2007). However, we did not observe in our cohort of myeloma patients undergoing autologous transplantation that the number of CD34+ cells infused at transplantation affected OS or TTP, with P=0.754 and P=0.899, respectively, for patients below vs above the mean value of infused CD34+ cells. In a Supplementary Table S1, we also included the number of infused CD34+ cells in the multivariate analysis. We found that the number of circulating CD34+ cells still remained an independent factor for OS with a P-value of 0.0004. We thus conclude that the favourable effect of high numbers of circulating CD34+ cells is independent from the number of infused CD34+ cells at autologous transplantation in myeloma patients.
In conclusion, this study identified high levels of circulating CD34+ cells at the day of stem cell collection to be associated with favourable outcome in myeloma patients undergoing autologous transplantation. We propose that this biomarker might be considered to be integrated into future risk stratification in myeloma patients to select patients for a post-transplant maintenance or consolidation strategy.
Change history
29 March 2012
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF, Casassus P, Maisonneuve H, Facon T, Ifrah N, Payen C, Bataille R (1996) A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. Intergroupe Francais du Myelome. N Engl J Med 335: 91–97
Bargetzi MJ, Passweg J, Baertschi E, Schoenenberger A, Gwerder C, Tichelli A, Burger J, Mingrone W, Herrmann R, Gratwohl A, Wernli M (2003) Mobilization of peripheral blood progenitor cells with vinorelbine and granulocyte colony-stimulating factor in multiple myeloma patients is reliable and cost effective. Bone Marrow Transplant 31: 99–103
Barlogie B, Bolejack V, Schell M, Crowley J (2010) Prognostic factor analyses of myeloma survival with intergroup trial S9321 (INT 0141): examining whether different variables govern different time segments of survival. Ann Hematol 90: 423–428
Barlogie B, Shaughnessy J, Tricot G, Jacobson J, Zangari M, Anaissie E, Walker R, Crowley J (2004) Treatment of multiple myeloma. Blood 103: 20–32
Blade J, Rosiñol L, Sureda A, Ribera JM, Diaz-Mediavilla J, Garcia-Laraña J, Victoria Mateos M, Palomera L, Fernandez-Calvo J, Marti JM, Giraldo P, Carbonell F, Callis M, Trujillo J, Gardella S, Jesus Moro M, Barez A, Soler A, Font L, Fontanillas M, San Miguel J (2005) High-dose therapy intensification compared with continued standard chemotherapy in multiple myeloma patients responding to the initial chemotherapy: long-term results from a prospective randomized trial from the Spanish cooperative group PETHEMA. Blood 106: 3755–3759
Bolwell BJ, Pohlman B, Rybicki L, Sobecks R, Dean R, Curtis J, Andresen S, Koo A, Mineff V, Kalaycio M, Sweetenham JW (2007) Patients mobilizing large numbers of CD34+ cells (‘super mobilizers’) have improved survival in autologous stem cell transplantation for lymphoid malignancies. Bone Marrow Transplant 40: 437–441
Cavo M, Rajkumar V, Palumbo A, Moreau P, Orlowski R, Bladé J, Sezer O, Ludwig H, Dimopoulos MA, Attal M, Sonneveld P, Boccadoro M, Anderson KC, Richardson PG, Bensinger W, Johnsen H, Kroeger N, Gahrton G, Leif Bergsagel P, Vesole DH, Einsele H, Jagannath S, Niesvizky R, Durie BGM, Lonial S (2011) International Myeloma Working Group (IMWG) consensus approach to the treatment of multiple myeloma patients who are candidates for autologous stem-cell transplantation. Blood 117: 6063–6073
Chang H, Trieu Y, Qi X, Jiang NN, Xu W, Reece D (2010) Impact of cytogenetics in patients with relapsed or refractory multiple myeloma treated with bortezomib: adverse effect of 1q21 gains. Leuk Res 35: 95–98
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K, Brown J, Drayson MT, Selby PJ (2003) High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med 348: 1875–1883
Dingli D, Rajkumar SV (2010) How best to use new therapies in multiple myeloma. Blood Rev 24: 91–100
Fermand JP, Katsahian S, Divine M, Leblond V, Dreyfus F, Macro M, Arnulf B, Royer B, Mariette X, Pertuiset E, Belanger C, Janvier M, Chevret S, Brouet JC, Ravaud P (2005) High-dose therapy and autologous blood stem-cell transplantation compared with conventional treatment in myeloma patients aged 55 to 65 years: long-term results of a randomized control trial from the Group Myelome-Autogreffe. J Clin Oncol 23: 9227–9233
Gordan LN, Sugrue MW, Lynch JW, Williams KD, Khan SA, Wingard JR, Moreb JS (2003) Poor mobilzation of peripheral blood stem cells is a risk factor for worse outcome in lymphoma patients undergoing autologous stem cell transplantation. Leuk Lymphoma 44: 815–820
Greipp PR, San Miguel J, Durie BG, Crowley JJ, Barlogie B, Bladé J, Boccadoro M, Child JA, Avet-Loiseau H, Kyle RA, Lahuerta JJ, Ludwig H, Morgan G, Powles R, Shimizu K, Shustik C, Sonneveld P, Tosi P, Turesson I, Westin J (2005) International staging system for multiple myeloma. J Clin Oncol 23: 3412–3420
Hiwase DK, Hiwase S, Bailey M, Bollard G, Schwarer AP (2008) Higher infused lymphocyte dose predicts higher lymphocyte recovery, which in turn, predicts superior overall survival following autologous hematopoietic stem cell transplanation for multiple myeloma. Biol Blood Marrow Transplant 14: 116–124
Hose D, Rème T, Hielscher T, Moreaux J, Messner T, Seckinger A, Benner A, Shaugnessy Jr JD, Barlogie B, Zhou Y, Hillengass J, Bertsch U, Neben K, Möhler T, Rossi JF, Jauch A, Klein B, Goldtschmidt H (2011) Proliferation is a central independent prognostic factor and target for personalized and risk-adapted treatment in multiple myeloma. Haematologica 96: 87–95
Kakihana K, Ohasi K, Akiyama H, Sakamaki H (2010) Correlation between survival and number of mobilized CD34+ cells in patients with multiple myeloma or waldenström macroglobulinemia. Pathol Oncol Res 16: 583–587
Kyle RA, Rajkumar SV (2009) Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia 23: 3–9
Lonial S (2010) Presentation and risk stratification-improving prognosis for patients with multiple myeloma. Cancer Treat Rev 36 (Suppl 2): S12–S17
Munshi NC, Anderson KC, Bergsagel PL, Shaughnessy J, Palumbo A, Durie B, Fonseca R, Stewart K, Harousseau JL, Dimopoulos M, Jagannath S, Hajek R, Sezer O, Kyle R, Sonneveld P, Cavo M, Rajkumar SV, San Miguel J, Crowley J, Avet-Loiseau H (2011) Consensus recommendation for risk stratification in multiple myeloma: report of the International Myeloma Workshop Consensus Panel 2. Blood 117: 4696–4700
Palumbo A, Sezer O, Kyle R, Miguel JR, Orlowski RZ, Moreau P, Niesvizky R, Morgan G, Comenzo R, Sonneveld P, Kumar S, Hajek R, Giralt S, Bringhen S, Anderson KC, Richardson PG, Cavo M, Davies F, Bladé J, Einsele H, Dimopoulos MA, Spencer A, Dispenzieri A, Reiman T, Shimizu K, Lee JH, Attal M, Boccadoro M, Mateos M, Chen W, Ludwig H, Joshua D, Chim J, Hungria V, Turesson I, Durie BGM, Lonial S (2009) International Myeloma Working Group guidelines for the management of multiple myeloma patients ineligible for standard high-dose chemotherapy with autologous stem cell transplantation. Leukemia 23: 1716–1730
Rajkumar SV (2009) Multiple myeloma. Curr Probl Cancer 33: 7–64
Rajkumar SV (2010) Multiple myeloma: 2011 update on diagnosis, risk-stratification, and management. Am J Hematol 86: 57–65
Rajkumar SV, Kyle RA (2005) Multiple myeloma: diagnosis and treatment. Mayo Clin Proc 80: 1371–1382
Scadden DT (2006) The stem-cell niche as an entity of action. Nature 441: 1075–1079
Stockerl-Goldstein KE, Reddy SA, Horning SF, Blume KG, Chao NJ, Hu WW, Johnston LJ, Long GD, Strober S, Wong RM, Feiner RH, Kohler S, Negrin RS (2000) Favorable treatment outcome in non-Hodgkin's lymphoma patients with ‘poor’ mobilization of peripheral blood progenitor cells. Biol Blood Marrow Transplant 14: 116–124
Terpos E, Apperley JF, Samson D, Giles C, Crawley C, Kanfer E, Olavarria E, Goldman JM, Rahemtulla A (2003) Autologous stem cell transplantation in multiple myeloma: improved survival in nonsecretory multiple myeloma but lack of influence of age, status at transplant, previous treatment and conditioning regimen. A single-centre experience in 127 patients. Bone Marrow Transplant 31: 163–170
Wang S, Nademanee A, Qian D, Dagis A, Park HS, Fridey J, Smith E, Snyder D, Somlo G, Stein A, Rosenthal J, Falk P, Kogut N, Palmer J, Gaal K, Kim Y, Bhatia R, Yuan S, Kay C, Weiss L, Forman S (2007) Peripheral blood hematopoietic stem cell mobilization and collection efficacy is not an independent prognostic factor for autologous stem cell transplantation. Transfusion 47: 2207–2216
Wilson A, Trumpp A (2006) Bone marrow haematopoietic stem cell niches. Nat Rev Immunol 6: 93–106
Acknowledgements
We thank Marion Bleckmann, Barbara Muster and Myriam Degos for valuable assistance during the collection of patient data. This work was supported by grants from the Swiss National Science Foundation SF 310000-109388 and Oncosuisse OCS-01833-02-2006 to TP. The funding source had no role in publication of these data.
Author contributions
JR performed research, analysed data and wrote the paper; DR analysed data; SM performed research; BM analysed data; and TP designed research, analysed data and wrote the paper. All authors have read and approved the report in its final version.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Raschle, J., Ratschiller, D., Mans, S. et al. High levels of circulating CD34+ cells at autologous stem cell collection are associated with favourable prognosis in multiple myeloma. Br J Cancer 105, 970–974 (2011). https://doi.org/10.1038/bjc.2011.329
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2011.329