Abstract
Background:
The KIAA1199 transcript is upregulated in colon adenomas and downregulated upon β-catenin knockdown.
Methods:
Transcript profiling was performed on >500 colon biopsies, methylation profiling data were compared with transcript data. Immunohistochemistry assessed KIAA1199 protein expression in 270 stage II/III tumours (>3 years follow-up). The effects of stable KIAA1199 knockdown in SW480 cells (three different constructs) were studied using transcriptional profiling, proliferation and protein analysis.
Results:
The KIAA1199 transcript was strongly upregulated in 95% of adenocarcinomas. Absent expression in normal mucosa correlated with KIAA1199 promotor methylation. Nuclear and cytoplasmic KIAA1199 protein expression was identified in colon adenocarcinomas and other types of cancers. A subpopulation of patients with tumours strongly expressing KIAA1199 in the nucleus showed a better outcome with regard to recurrence as lung or liver metastases. The KIAA1199 knockdown affected the cell cycle and the Wnt-signalling pathway. Reduced cellular proliferation and decreased KI67, phosphorylated retinoblastoma, β-catenin and ASCL2 protein expression supported these findings. Eighteen Wnt-signalling genes differentially expressed upon KIAA1199 knockdown correlated with the KIAA1199 expression profile in clinical specimens.
Conclusion:
The KIAA1199 knockdown attenuates the effects of the Wnt/β-catenin signalling and it may thus be regarded as a regulatory part of this pathway.
Similar content being viewed by others
Main
Colorectal cancers (CRC) account for ∼10% of the total worldwide cancer cases, with an overall 5 years survival of around 50% (Boyle and Levin, 2008). Early diagnosis and improved treatment of CRC requires the identification of new biomarkers as well as insights into the molecular mechanisms of human carcinogenesis.
Most CRCs are caused by aberrant Wnt-signalling, which in 70–80% of cases are rooted in mutational inactivation of the tumour-suppressor gene APC (Bienz and Clevers, 2000). This leads to stabilisation and nuclear translocation of the proto-oncogene β-catenin. β-catenin has a crucial role in cell–cell adhesion and is part of the transcription factor complex β-catenin/TCF, recruiting numerous co-regulators to achieve activation or inhibition of target gene promoters in response to Wnt-signalling (Gregorieff and Clevers, 2005). Subsequent to APC inactivation follow mutations in oncogenes-like K-Ras and tumour-suppressor genes like TP53 (Kinzler and Vogelstein, 1996). Additional genetic changes may appear in CRC, for example, the apparent loss of the response to transforming-growth factor-β (TGF-β) (Polyak, 1996). The TGF-β-signalling occurs through Smads-2/3/4, translocating to the nucleus to regulate transcription. However, Smad4 also has a TGF-β-independent function as a tumour suppressor cooperating with β-catenin/Lef to regulate target gene expression (Tian et al, 2009).
In a previous transcript profiling study on a small sample set, we identified the KIAA1199 transcript to be strongly upregulated in colon adenocarcinomas stage I-IV compared with normal mucosa (Birkenkamp-Demtroder et al, 2002). The KIAA1199 was also found to be upregulated in colon adenomas (Sabates-Bellver et al, 2007). Induction of TCF4 proteins or siRNA-mediated knockdown of β-catenin in LS174 colon cells decreased KIAA1199 transcript expression (Sabates-Bellver et al, 2007). The TCF4 protein (TCF7L2) is a transcription factor involved in the Wnt/β-catenin-signalling pathway and findings of genome-wide TCF4 ChIP-on-chip analyses indicate that the KIAA1199 locus is surrounded by four TCF4-binding regions (Sabates-Bellver et al, 2007), suggesting an involvement of KIAA1199 in the Wnt/β-catenin-signalling pathway.
The KIAA1199 gene (Hs.459088, chr.15q25.1, TMEM2 L) encodes a 150 kDa protein and was originally described as an inner ear protein (Abe et al, 2003). KIAA1199 has been associated with cancer tissues: high KIAA1199 transcript levels were found in mortal but not immortal renal cell carcinoma cells (Michishita et al, 2006) whereas low KIAA1199 expression in gastric cancers was related to significant better outcome of the patients (Matsuzaki et al, 2009).
Despite previous studies indicating an upregulation of KIAA1199 in neoplasic CRC tissues, this has not been confirmed in a large set of samples, neither with regard to transcript or protein levels, nor with regard to clinical outcome. Moreover, KIAA1199's putative cellular functions and pathway interactions have not been reported previously.
The aim of this study was to characterise the KIAA1199 transcript and protein expression in colorectal adenocarcinomas, the correlation of KIAA1199 expression with patient outcome and the identification of KIAA1199-related pathways to elucidate the role of KIAA1199 in CRC.
Materials and methods
Tissue samples
Data from four different tissue sample sets were used in this study: set 1: U133A-arrays, n=122 (Kruhoffer et al, 2005); set 2: U133plus2.0-arrays, n=389 (Andersen et al, 2009); set 3: Exon1.0ST-arrays, n=40 (Øster et al, 2011); set 4: Exon1.0ST-arrays, n=27, (Thorsen et al, 2008)). Informed written consent was obtained from all patients and the studies were approved by the Central Denmark Region Committees on Biomedical Research Ethics.
Methylation profiling on 40-microdissected colon tissue samples were analysed on Infinium HumanMethylation27 BeadChips (Illumina, San Diego, CA, USA) as described in (Øster et al, 2011). In brief, DNA extracted from fresh frozen tissue samples was bisulfite modified, whole genome amplified and hybridised to Beadchips.
Colon cell lines obtained from American Type Culture Collection (ATCC-LGC standards, Borås, Sweden) were re-authenticated via STR analysis (Masters et al, 2001) using the Cell-ID-system (G9500, Promega, Nacka, Sweden). No mycoplasma contamination was detected using nested PCR. RNA was extracted using GenElute Mammalian Total RNA Miniprep Kit (RTN350, Sigma-Aldrich, Brøndby, Denmark). RNA (RIN⩾9.9) was analysed on (a) U133plus2.0 GeneChips; comparison analysis was performed using MAS5.0 software (Affymetrix, Santa Clara, CA, USA). Probes accompanied by an Inc/Dec call and a log2 ratio ∣>0.5∣ were included, but excluded when listed as ‘absent’ or (b) ExonST1.0arrays; data were quantile-normalized with ExonRMA16-algorithm and data analysis was performed using GeneSpring GX10-software (Agilent, Hørsholm, Denmark). Genes were annotated using the Affymetrix NETAFFX annotation (NCBI Build 36.1, netaffx-build=28, Santa Clara, CA, USA).
Antibody synthesis and cloning of KIAA1199
Polyclonal rabbit anti-KIAA1199 antibodies were raised against peptide CARYSPHQDADPLKPR (amino-acids 772–776 in Q8WUJ3, Eurogentec, Seraing, Belgium). Antibody specificity of the affinity-purified antibody was confirmed by ELISA (Eurogentec), western blotting using recombinant V5-His- or GFP-C-terminal-tagged KIAA1199 proteins (full length and splice variant), cloned to a previously published procedure (Birkenkamp-Demtroder et al, 2007) as well as immunohistochemistry on formalin-fixed and paraffin embedded cells (FFPE) as described in the Supplementary data. Western Blotting was performed on 4–12% gradient gels (Olesen et al, 2005). Loading control anti-β-actin monoclonal antibody 0.05 μg ml−1 (#A-1978, clone AC-15, Sigma-Aldrich) (Marker ‘All Blue’ (Bio-Rad, Copenhagen, Denmark)).
FFPE cells
The FFPE cells 80% confluent cells were harvested by scraping, washed with 10 ml HANK's buffer (formaldehyde solution 4%, pH 7) and pelleted at 1200 r.p.m. for 5 min. The supernatant was removed and 5 ml 10% formalin were added and incubated at 4°C overnight. The supernatant was removed and the pellet was dehydrated by adding 2 × 5 ml 70% EtOH (Kemethyl A/S, Køge, Denmark), 2 × 96% EtOH, 3 × 99% EtOH and 3 × Xylen (Merck, Hellerup, Denmark), incubation 5 min each. Shandon histoplast paraffin (Hounisen, Risskov, Denmark) was added and the pellet was incubated overnight at 60°C. The pellet was transferred to a paraffin cutting tray, paraffin embedded and 8 μm thick sections were cut and immunostained.
Immunohistochemistry was performed on FFPE specimens as previously published (Birkenkamp-Demtroder et al, 2007), using 4 μm tissue sections from the colon biopsies and 8 μm sections from FFPE cell culture cells. Tissue slides were scanned with a Nanozoomer (Hamamatsu, Herrsching am Ammersee, Germany) and cores were analysed by VIS-TMA-software (Visiopharm, Hørsholm, Denmark).
Tissue microarray analysis
We analysed KIAA1199 protein expression on a UICC stage I-IV CRC TMA (COCA912-5-OL, BioCat, Heidelberg, Germany), a human various tumours and corresponding normal specimens TMA with 94 cases (T8235713-5-BC, BioCat, previously used in (Mansilla et al, 2009)) as well as a TMA with a total of 438 stage II/III samples and detailed clinical data (MOMA). Recurrence was defined as distant recurrence. Patients with other cancers than CRC, with multiple CRC, with local recurrence or recurrence in the form of carcinosis as well as with <3 years of follow-up were excluded from the study, resulting in 270 stage II samples with 3 years of follow-up. Scoring was independently done by two experienced observers (KBD, CLA). Weighted kappa statistics showed a good interobserver agreement for cytoplasmic (κ=0.8806) and nuclear KIAA1199 expression (κ=0.6077).
The shRNA-vector preparation and lentivirus production was performed according to (Stewart et al, 2003) generating five KIAA1199-specific knockdown constructs. Quantitative RT-PCR was used to validate efficient knockdown and data were normalised against GAPDH, HPRT1 and PPIA (see Supplementary data).
Immunofluorescence microscopy
Cells seeded on glass chamber slides (#178599, Nunc, Roskilde, Denmark) were fixed, stained and mounted as previously described (Birkenkamp-Demtroder et al, 2007). Primary antibodies are as follows: monospecific rabbit anti-KIAA1199 antibody (4–6 μg ml−1); anti-KI67 (1 : 400, DAKOcytomation, Copenhagen, Denmark), anti-β-catenin (1 : 400 IF, BDBiosciences, Brøndby, Denmark), anti-V5-tag (SV5-Pk1) (1 : 3000, ab27671, Abcam, Cambridge, UK), anti-RB-pS795 (1 : 250, monoclonal anti-phosphoretinoblastoma (RB) (pS795) clone RB-10, R6878, Sigma-Aldrich) and anti-ASCL2 7E2-supernatants (1 : 5, (Van der Flier et al, 2009)). The specificity of the ASCL2 antibody was proven on western blots of extracts from ASCL2 overexpressing cells, by ChIP-analyses and inducible ASCL2 RNAi in LS174 colon cancer cells (personal communication Laurens Van der Flier). Secondary antibodies are as follows: AlexaFlour 488 goat anti-mouse or goat anti-rabbit IgG highly cross-adsorbed (1 : 2000, Molecular Probes, Invitrogen, Taastrup, Denmark). Nuclei were counterstained with DAPI or Hoechst and slides were mounted with fluorescence mounting medium (DAKOcytomation).
Ingenuity pathway analysis
Data below background noise (threshold log2<4.0) were excluded from analyses (IPA version 8.5, Redwood City, CA, USA). Expression values were normalised around zero. Normalised ratios given as (−INF, −1) and (1, +INF) were submitted to ingenuity pathway analysis (IPA).
Real time cell analyses of proliferation, adhesion and migration were performed in triplicates on xCELLigence instruments (Roche, Hvidovre, Denmark).
Cell proliferation was assessed by an MTT-assay (3-(4,5-dimethylthiazol-2-yl)2,5-diphenyltetrazolium bromide, Roche) on a Synergy-HT Microplate-Reader (Biotek, Bad Friedrichshall, Germany), readout at 540 nm, absorbance at 692 nm was used as reference.
Cell adhesion was analysed as previously described (Weinacker et al, 1994). Briefly, 2000–4000 cells in a total volume of 50 μl volume (n=10) were incubated on 96-well-plates coated with fibronectin (F0895, Sigma-Aldrich), readout at 550 nm.
Statistical analysis was performed using STATA 10 (Statacorp, College Station, TX, USA). Transcript values were expressed as median log2 ±s.d. A two-tailed Student's t-test or log-rank test for survival estimates was applied and P-values <0.05 were considered as statistically significant.
Results
KIAA1199 is strongly upregulated in most tumours of the large intestine independent of their location
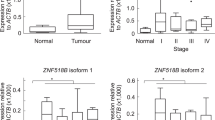
Microarray transcript profiling using U133A (set1) and U133plus2.0 GeneChips (set 2) was performed on 511 sporadic CRC tissue samples obtained from multiple institutions in four countries (Kruhoffer et al, 2005, Andersen et al, 2009). The KIAA1199 transcript was significantly upregulated (P<1E-07) in MSS and MSI colon adenocarcinomas compared with normal mucosa (Figure 1a), and was strongly expressed in >95% of colon adenocarcinomas (log2>7; Supplementary Figures 1a and b). The KIAA1199 transcript level was significantly higher in MSS than MSI tumours (P=8.9E-04). No difference was seen between samples from proximal versus distal colon or between stage II versus stage III tumours (data not shown), indicating an almost obligatory high expression of the KIAA1199 transcript in CRC. We identified a slightly, but significantly lower expression in tumours from stage II–III patients with recurrence (n=54, median log2=8.6) compared with those without recurrence (n=241, median log2=8.8, P=0.03). Immunohistochemical analysis (IHC) of KIAA1199 protein expression was performed using a monospecific, affinity-purified antibody. The antibody's specificity was proven by western blotting (Figure 1c, Supplementary Figure 2a) as well as by immunohistochemical analysis of FFPE cells (Supplementary Figure 2b).
The KIAA1199 transcript and protein expression and correlation to clinical outcome. (A) The KIAA1199 transcript profiling of 511 colon samples (normal mucosas, MSS microsatellite stable and MSI instable and adenocarcinomas). (a) The U133A GeneChips (n=122); (b) U133plus2.0 GeneChips (n=389); (details shown in Supplementary Figure 1). (B) Normal colon mucosa showed a very weak expression in the cytoplasm of single cells located in the bottom of the crypts. (C) Western Blot of SW480 cells overexpressing KIAA1199-V5-HIS protein (157 kDa). Lane 1 – Marker Bio-Rad All Blue; lane 2 – the monospecific rabbit polyclonal anti-KIAA1199 antibody was specific for KIAA1199 (specificity is shown in Supplementary Figures 2a and b). (D) Immunohistochemical analysis of 270 stage II adenocarcinomas (>3 years follow-up) using the monospecific anti-KIAA1199 antibody: group 1 showed strong nuclear but weak cytoplasmic KIAA1199 expression, group 2 showed moderate to strong cytoplasmic KIAA1199 expression, no nuclear expression. (E) In all 245 of the 270 samples were informative (118 female, 127 male). Patients without disease recurrence (n=205, median follow-up time 2198 days, range 1099–4368); patients with metastatic recurrence (n=40, median time to recurrence 830 days, range 95–2449). Kaplan–Meier survival estimates for disease-free survival showed that patients with group 1 tumours (blue line) had a significant (log-rank test P-value 0.02) better outcome than group 2 patients (red line). Green ticks: patients censored at death or end of follow-up.
The IHC analysis revealed a strong KIAA1199 protein expression in the nucleus and/or cytoplasm of the majority of UICC stage I–IV adenocarcinomas (n=40). No relationship between staining intensity and disease stage was found (data not shown). In addition, other types of cancers from, for example, breast, kidney, lung and stomach also showed upregulated KIAA1199 protein levels (data not shown). In normal colon mucosa (n=10), KIAA1199 was either not or only very weakly expressed in the cytoplasm of single cells located in the proliferative compartment at the bottom of the crypts (Figure 1b).
To study the relation to clinical outcome of CRC, we used a TMA with 270 stage II colorectal adenocarcinomas with at least 3 years of follow-up, as stage II cancers include a small group of about 20% of patients that will progress and should be identified for chemotherapeutic treatment. Remarkably, a small group of patients with tumours strongly expressing KIAA1199 in the nucleus accompanied by weak cytoplasmic expression (Figure 1d) had a significantly (log-rank test P-value 0.02) better outcome compared with those with strong cytoplasmic, or nuclear and cytoplasmic KIAA1199 expression using Kaplan–Meier survival estimates of disease-free survival (Figure 1e).
To explore if the KIAA1199 transcript levels are regulated by DNA methylation, we investigated DNA promoter methylation in normal colon mucosas (n=6), adenomas (n=6) and adenocarcinomas (n=28), using Infinium HumanMethylation27 BeadChips (Illumina). The analysis showed that all six normal mucosa samples were methylated at target position cg20828084 (median avg beta=0.63, see details in Øster et al, 2011). This position was found to be less methylated in the majority of the stage II–III adenocarcinomas with MSI (5/6) or MSS status (23/24) (median avg β=0.47). Parallel expression profiling of these samples (set 3) showed that the KIAA1199 transcript was absent in normal and present in cancer (Supplementary Figure 1c), indicating that KIAA1199 may be regulated, at least partly, by CpG island methylation. Clinical correlation and KIAA1199 upregulation in the majority of colon adenocarcinomas prompted us to study the effects of KIAA1199 dysregulation in vitro.
KIAA1199 upregulation or knockdown in several colon cell lines alter pathway-specific transcripts and the cellular phenotype
We overexpressed V5-His-tagged-KIAA1199 in HCT116 and SW480 cell lines (protein expression and localisation are shown in Supplementary Figures 2c and d) and performed transcript profiling on U133plus2.0 arrays compared with control cells transfected with an empty vector. A 28-fold upregulation of KIAA1199 in HCT116 cells resulted in 306 deregulated genes, whereas an 80-fold upregulation in SW480 cells resulted in 1304 genes (log2 >∣0.5∣) being differentially expressed. Common to both cell lines were 33 genes. The IPA showed that molecular and cellular functions of the cell cycle (135 molecules, P=3.57E-12), cell death (224, P=4.72E-10), cell division process (P=3.65E-11), cellular growth and proliferation (236, 7.45E-07), cellular movement (66, 4.59E-06), cell morphology (133, 2.51E-06), as well as DNA replication and repair (67, 8.35E-06) were affected. Moreover, canonical pathways such as apoptosis, Ephrin-receptor- and Wnt/β-catenin-signalling were also affected (P<0.05).
Lentiviral-mediated knockdown of KIAA1199 using five different shRNA constructs yielded between 45% and >80% knockdown efficiency in SW480 cells (Supplementary Figure 3a), whereas knockdown efficiency of SW948 and LS1034 cells with 70-fold higher endogenous KIAA1199 expression was only about 50% (data not shown). To investigate the transcriptional consequences associated with KIAA1199 knockdown, transcript profiling (U133plus2.0, ExonST.1.0arrays) of SW480 cells stably transfected with an empty vector or three different knockdown constructs with favourable knockdown efficiency sh6640 (>65%), sh2396 (>75%) and sh3303 (>80%) was performed to control for OFF-target effects (Supplementary Figure 3b). A total of 3727 probesets differentially expressed in common in all three knockdowns were identified (sh-3303 log2 ratio >∣0.5∣ accompanied by log2 ratio >∣0.2∣ in either sh6640 or sh2396 or both). The IPA analysis of this gene list revealed ‘cell to cell signalling’ and ‘cellular growth and proliferation’ were significantly impacted upon KIAA1199 knockdown (P<0.01). However, most interestingly, KIAA1199 knockdown affected the expression of 67 genes involved in Wnt/β-catenin signalling, most of them downregulated upon KIAA1199 knockdown (Figures 2a and b). This impact of KIAA1199 on Wnt-signalling was further substantiated by immunofluorescence microscopy (IF) and western blotting, showing a decreased protein expression of β-catenin and the Wnt-related stem cell marker ASCL2 upon KIAA1199 knockdown with construct KIAA1199-sh3303 (Figures 2c and d). Furthermore, IF confirmed the decrease of ASCL2 upon KIAA1199 knockdown when two additional, alternative knockdown constructs, sh2396 and sh6640, were used (Supplementary Figure 4a). A total of 145 genes were found to be systematically dysregulated in SW480 cells either upon KIAA1199 overexpression or knockdown with KIAA1199-sh3303 (log2>∣0.5∣, Supplementary Table 1). Hereof, 87 genes were increased upon KIAA1199 knockdown and decreased upon overexpression, among those the tumour suppressor p53. Another set of 58 genes were decreased upon knockdown and increased after KIAA1199 overexpression, among these several genes involved in Wnt-signalling including CD44, DKK3 or CST1.
KIAA1199 knockdown affects the Wnt/β-catenin signalling pathway. (A) The IPA showed that a number of genes involved in canonical Wnt/β-catenin signalling were up (yellow) or downregulated (blue) upon KIAA1199 knockdown. (B) Summary of relations between KIAA1199, β-catenin, ASCL2 and LGR5: (1) knockdown from β-catenin shuts off the Wnt/β-catenin signalling resulting in a decreased KIAA1199 transcript expression (Sabates-Bellver et al, 2007) as well as a downregulation of the ASCL2 transcript (Jubb et al, 2006). (2) Knockdown from ASCL2 results in the silencing of LGR5 (Van der Flier et al, 2009). Our data show that knockdown from KIAA1199 resulted in a decreased expression of (3) β-catenin, (4) ASCL2 and (5) LGR5 and to lesser extent some genes targeted by ASCL2 (not shown). A decreased β-catenin expression upon KIAA1199 knockdown may suggest a regulatory loop probably further affecting β-catenin signalling. (C) Immunofluorescence analysis confirmed the downregulation of β-catenin and ASCL2 upon KIAA1199 knockdown in SW480 cells compared with control cells (see also Supplementary Figure 4). Blue: DAPI nuclear stain; green: Alexa488 secondary antibody; × 630 magnification, Zeiss Axiovision, Carl Zeiss, Brock & Michelsen A/S, Birkerød, Denmark). (D) Western blot with extracts from SW480 cells stably transfected with an empty vector (lanes 2 and 4) or KIAA1199-sh3303 knockdown (lane 3, 5) was incubated with anti-β-catenin antibody in a 1 : 4000 (lane 2, 3) or 1 : 2000 dilution (lane 4, 5). β-catenin protein expression was markedly decreased upon KIAA1199 knockdown compared with control cells. β-actin was used as loading control. Lane 1=Marker Bio-Rad All Blue.
KIAA1199 knockdown impacts the cellular phenotype
Functional analyses using real time cell monitoring (RTCA) showed that knockdown using construct KIAA1199-sh3303 reduced the proliferation rate by about 50% (Figure 3a). The decrease in the proliferation rate was highly significant (P<0.0001) as confirmed by an MTT assay (Figure 3b), in agreement with reduced levels of proliferation-related transcripts. The typical granular nuclear staining of the proliferation marker KI67 (Figure 3c) as well as the strong nuclear expression of phosphoretinoblastoma (Figures 3d and e) were both decreased in KIAA1199-sh3303-depleted SW480 cells. Moreover, KIAA1199 knockdown using alternative constructs sh6640 and sh2396 confirmed these results. IF showed a markedly decreased KI67 and Rb-pS795 in KIAA1199 protein expression in KIAA1199-depleted SW480 cells although an MTT assay confirmed a significantly (P<0.05) reduced proliferation rate (Supplementary Figures 4a and b).
KIAA1199 knockdown affects proliferation of SW480 cells. SW480-KIAA1199-sh3303 colon cancer cells depleted of KIAA1199 (•) and an empty vector control (○) were seeded on E-plates on a RTCA-instrument (x-CELLigence, Roche). Experiments were performed at least in triplicates, values are shown as medians and s.d.'s for each group at selected times for a representative experiment. (A) Seeding of 4000 cells per well yielded control cells in the log phase from 36–96 h, cells were monitored in 1 min intervals. The proliferation rate of the KIAA1199-depleted cells was decreased by about 50% compared with control cells. (B) MTT-assays at selected time points confirmed a significant (P<0.05) decrease of proliferation of the KIAA1199-sh3303 depleted SW480 cells compared with control cells. (C) IF showed less nuclear expression of proliferation marker KI67 in KIAA1199-depleted SW480 cells compared with control cells using an 1 : 400 dilution of the anti-KI67 antibody (green: Alexa488-labelled secondary antibody; blue: Hoechst nuclear stain); × 630 (D) IF using a 1 : 250 dilution of the monoclonal anti-phosphoretinoblastoma antibody Rb-pS795 reacting specifically with Rb phosphorylated at serine 795 and not with non-phosphorylated Rb showed a dramatical decrease of phosphorylated Rb protein in KIAA1199-depleted SW480 cells compared with control cells. (E) Western blot with SW480 cells stably transfected with empty vector (lanes 2, 4) or KIAA1199-sh3303 knockdown (lane 3, 5), incubated with anti- RB-pS795 antibody (1 : 2000, lane 2, 3; 1 : 1000 lane 4, 5). Expression of the phosphorylated RB-pS795 protein was markedly decreased upon KIAA1199 knockdown compared with control cells. β-actin was used as loading control. Lane 1=Marker Bio-Rad All Blue.
Moreover, pathway analysis pointed out that KIAA1199-sh3303 knockdown reduced the actin filament or microtubule-based cellular movement comprising genes like MYO1E, KIF1B or TUBE1 as well as the maintenance of the cytoskeleton signalling involving genes like MTSS1, ARHGAP26, DIAPH2, ABLIM1 or DAAM1). RTCA-migration analysis of KIAA1199-depleted SW480 cells using CIM-plates showed a decreased migration (Supplementary Figure 5a). Moreover, IPA analysis showed that molecules modulating cell–cell contacts were altered upon KIAA1199 knockdown. RTCA analysis showed a 50% reduced cellular adhesion of KIAA1199-depleted SW480 cells (Supplementary Figure 5b). Decreased adhesion was confirmed by a colorimetric assay on fibronectin-coated plates (Supplementary Figure 5c).
In vitro impact of KIAA1199 on Wnt-signalling molecules is reflected in the clinical CRC samples
In vitro data suggested the existence of a potential correlation between the expression of KIAA1199 and Wnt-signalling molecules. Transcript data from a set of nine normal mucosas and 18 microdissected MSS-adenocarcinomas analysed on Exon1.0ST-arrays (set 4, (Thorsen et al, 2008)) showed that KIAA1199 transcript expression correlated very well with the expression of 37 genes known to be involved in Wnt/β-catenin-signalling (Pearson's coefficient 0.5–0.9) (Table 1). The KIAA1199 transcript expression correlated best with Cyclin D1 (CCND1), LGR5, AXIN2, ASCL2, CD44, PITX2 and CLDN1 ((Pearson's coefficient >0.8), Supplementary Figure 6a). Remarkably, these genes were also found to be deregulated upon KIAA1199 knockdown (Table 1). Excluding the normal samples, KIAA1199 expression still correlated well with the Wnt-signalling genes CD44, LGR5 and β-catenin, supporting that KIAA1199 has a strong relation to the Wnt/β-catenin signalling pathway.
We compared nuclear KIAA1199 protein expression with nuclear β-catenin expression in the same cores of our CRC TMA, as nuclear β-catenin corresponds to active Wnt/β-catenin signalling in vivo. Remarkably, 69% (25 out of 36) of the tumours with strong nuclear and weak cytoplasmic KIAA1199 localization showed nuclear β-catenin localization, compared with 51% (106 out of 209) of the tumours with cytoplasmic KIAA1199 expression only (Supplementary Figure 6b). The nuclear KIAA1199 protein location was significantly correlated with the presence of nuclear β-catenin (odds ratio, 2.13; P=0.014) in logistic regression analysis.
Discussion
The KIAA1199 transcript was strongly upregulated in 95% of the colon adenocarcinomas compared with normal mucosas in a total of 511 colon samples investigated. Array-based methylation analysis and comparison with transcript expression data provided evidence that KIAA1199 expression may have been shut down in normal colon mucosa by promotor methylation.
In colon cancer, we localised strong expression of the KIAA1199 protein in the cytoplasm and the nucleus of adenocarcinomas. This is different compared with gastric cancer, where the protein was exclusively localised in the cytoplasm (Matsuzaki et al, 2009). Moreover, in gastric cancer the overall 5-year survival rate was significantly better for patients with low KIAA1199 expression in the primary tumour and strong KIAA1199 expression was more frequent in patients with distant metastasis and peritoneal dissemination (Matsuzaki et al, 2009). On the contrary, in our study on colon cancer, strong KIAA1199 expression was generally not significantly correlated to lower overall survival or the development of liver or lung metastases in patients with stage II primary tumours (patients developing peritoneal dissemination were excluded from our analyses). However, strong nuclear expression favoured disease-free survival in a subgroup of the stage II patients suggesting a role for nuclear KIAA1199 in CRC.
The finding of better survival in a minor group with high KIAA1199 nuclear expression may point to the diversity in biological pathways being active during carcinogenesis and during the metastatic process. There is no doubt that Wnt-signalling is important during CRC cancer formation. However, during the metastatic process, the generally abnormal genome, as well as the complex pathway networks, may show unexpected findings. In this case, the proliferative and mobility aspects of KIAA1199 seem to be uncoupled from the metastatic process. Recent findings provide evidence for the existence of alternative pathways counteracting Wnt/β-catenin signalling. For example, Lee et al, (2010) show that the glycoprotein 90K has antitumour activity in CRC cells suppressing Wnt/β-catenin signalling by ISGylation-dependent ubiquitination of β-catenin when interacting with CD9/CD82.
Nuclear KIAA1199 correlated significantly with the expression of nuclear β-catenin in colon adenocarcinomas. However, KIAA1199 lacks a nuclear import signal as well as predicted DNA-binding properties. Due to a transmembrane region, KIAA1199 may bind to other KIAA proteins containing domains localised to highly specialized submembranous sites, indicating a participation in intracellular signalling events (Nakayama et al, 2002). KIAA1199 interacts, for example, with KIAA0463, a plexin 2 precursor, which is a co-receptor for semaphorin involved in remodelling of the cytoskeleton, invasive growth and cell migration (Zachary et al, 2009). The KIAA1199 protein precursor is phosphorylated by the anaplastic lymphoma kinase (Daub et al, 2008), suggesting a potential role for KIAA1199 in phosphorylation networks and intracellular signalling processes.
Previously, KIAA1199 expression in colon adenomas was correlated to the expression of 21 known Wnt-signalling genes (Sabates-Bellver et al, 2007). In our study, KIAA1199 transcript expression correlated with the expression of 37 genes known to be involved in Wnt-signalling, strongly supporting an involvement of KIAA1199 in the Wnt/β-catenin pathway.
We showed that KIAA1199 overexpression in SW480 cells affected expression of genes involved in the Wnt/β-catenin pathway. About 25% of the genes differentially expressed upon KIAA1199 overexpression were also significantly differentially expressed in adenocarcinomas compared with normal mucosas, indicating a potential regulatory role for KIAA1199 in vivo. However, KIAA1199 overexpression did not have a major impact on the cells as it would have been expected from an 80-fold upregulation. One explanation may be that in SW480 cells an increase in KIAA11199 probably only results in minor changes due to a constitutively activated Wnt/β-catenin signalling.
The number of genes involved in cell-cycle regulation differentially expressed upon KIAA1199 knockdown, decreased proliferation and the decrease of KI67 and RbpS795 suggested that KIAA1199 knockdown may affect cell-cycle progression. This may potentially be achieved by affecting the EBP1–RB-E2F transcription repression as unphosphorylated RB will inhibit the activity of the E2F family of transcription factors (Sidle et al, 1996) and will thus lead to a decelerated G1-S transition (Lee and Yang, 2003).
Alternatively, ASCL2, identified to be downregulated upon KIAA1199 knockdown, may promote progression through the G2/M cell-cycle checkpoint and may be a putative regulator of proliferation overexpressed in intestinal neoplasia (Jubb et al, 2006). ASCL2 is located in stem cells and its knockdown results in the loss of stem cells in the mouse intestine of an in vivo model (Van der Flier et al, 2009). Both, ASCL2 and KIAA1199 are under β-catenin control (Sabates-Bellver et al, 2007). Our data suggest that KIAA1199 may affect the expression of ASCL2 and LGR5 and may thus be an important player in functional Wnt/β-catenin signalling. In addition, KIAA1199 knockdown resulted in a feedback-loop of negative regulation of β-catenin in SW480 cells. The attenuation of the Wnt/β-catenin signalling may also contribute to a decrease in proliferation. Interestingly, despite the constitutive activation of Wnt/β-catenin signalling in SW480 cells, the knockdown of the KIAA1199 transcript was sufficient to cause an impact on the Wnt/β-catenin pathway and, as a consequence, the observed cellular phenotype. This suggests a central role for KIAA1199 in Wnt/β-catenin signalling.
The TGF-β and Wnt-signalling pathways can independently or cooperatively regulate LEF1/TCF target genes (Letamendia et al, 2001). Recent studies reported that SMAD4/DPC4 has a TGF-β-independent function as tumour suppressor in cooperation with β-catenin/Lef to regulate target gene expression (Tian et al, 2009). In silico promotor analyses of the KIAA1199 promoter region identified binding sites, for example, TP53, LEF1/TCF or SMAD4 (Supplementary Table 2). Reconstitution of functional SMAD4 protein in SW480 cells (Stuhler et al, 2006) resulted in a 10-fold upregulation of the KIAA1199 transcript, expression was confirmed by IF and western blot (not shown), suggesting an involvement of the SMAD4/TGF-β signalling pathway as an alternative regulatory mechanism for KIAA1199 expression in vivo in the presence of functional SMAD4. Alternative regulatory mechanisms for KIAA1199 may exist, for example, the COX2-signalling cascade. Treatment of HT29 cells with a selective cyclooxygenase-2 (COX2) inhibitor resulted in a decrease of the KIAA1199 transcript level and an anti-proliferative effect (Galamb et al, 2010).
In conclusion, we provide evidence that KIAA1199 transcript and protein are highly expressed in the majority of CRCs. KIAA1199 probably participates in Wnt-signalling, affecting cell proliferation, motility and adhesion. Moreover, KIAA1199 has a clinical correlation to outcome in stage II CRC patients. These findings warrant further studies to understand KIAA1199's direct molecular interactions as well as to investigate whether KIAA1199 may be a potential biomarker or therapeutic target.
Change history
29 March 2012
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Abe S, Usami S, Nakamura Y (2003) Mutations in the gene encoding KIAA1199 protein, an inner-ear protein expressed in Deiters’ cells and the fibrocytes, as the cause of nonsyndromic hearing loss. J Hum Genet 48 (11): 564–570
Andersen CL, Christensen LL, Thorsen K, Schepeler T, Sorensen FB, Verspaget HW, Simon R, Kruhoffer M, Aaltonen LA, Laurberg S, Orntoft TF (2009) Dysregulation of the transcription factors SOX4, CBFB and SMARCC1 correlates with outcome of colorectal cancer. Br J Cancer 100 (3): 511–523
Bienz M, Clevers H (2000) Linking colorectal cancer to Wnt signaling. Cell 103 (2): 311–320
Birkenkamp-Demtroder K, Christensen LL, Olesen SH, Frederiksen CM, Laiho P, Aaltonen LA, Laurberg S, Sorensen FB, Hagemann R, Orntoft TF (2002) Gene expression in colorectal cancer. Cancer Res 62 (15): 4352–4363
Birkenkamp-Demtroder K, Mansilla F, Sørensen FB, Kruhøffer M, Christensen L, Aaltonen LA, Verspaget H, Ørntoft TF (2007) Phosphoprotein Keratin 23 accumulates in MSS but not MSI colon cancers in vivo and impacts viability and proliferation in vitro. Mol Oncol 1 (2): 181–195
Boyle P, Levin B (2008) World Cancer Report. International Agency for Research on Cancer-ISBN-9789283204237. World Health Organization WHO Press: Geneva, Switzerland
Daub H, Olsen JV, Bairlein M, Gnad F, Oppermann FS, Korner R, Greff Z, Keri G, Stemmann O, Mann M (2008) Kinase-selective enrichment enables quantitative phosphoproteomics of the kinome across the cell cycle. Mol Cell 31 (3): 438–448
Galamb O, Spisak S, Sipos F, Toth K, Solymosi N, Wichmann B, Krenacs T, Valcz G, Tulassay Z, Molnar B (2010) Reversal of gene expression changes in the colorectal normal-adenoma pathway by NS398 selective COX2 inhibitor. Br J Cancer 102 (4): 765–773
Gregorieff A, Clevers H (2005) Wnt signaling in the intestinal epithelium: from endoderm to cancer. Genes Dev 19 (8): 877–890
Jubb AM, Chalasani S, Frantz GD, Smits R, Grabsch HI, Kavi V, Maughan NJ, Hillan KJ, Quirke P, Koeppen H (2006) Achaete-scute like 2 (ascl2) is a target of Wnt signalling and is upregulated in intestinal neoplasia. Oncogene 25 (24): 3445–3457
Kinzler KW, Vogelstein B (1996) Lessons from hereditary colorectal cancer. Cell 87 (2): 159–170
Kruhoffer M, Jensen JL, Laiho P, Dyrskjot L, Salovaara R, Arango D, Birkenkamp-Demtroder K, Sorensen FB, Christensen LL, Buhl L, Mecklin JP, Jarvinen H, Thykjaer T, Wikman FP, Bech-Knudsen F, Juhola M, Nupponen NN, Laurberg S, Andersen CL, Aaltonen LA, Orntoft TF (2005) Gene expression signatures for colorectal cancer microsatellite status and HNPCC. Br J Cancer 92 (12): 2240–2248
Lee JH, Bae JA, Lee JH, Seo YW, Kho DH, Sun EG, Lee SE, Cho SH, Joo YE, Ahn KY, Chung IJ, Kim KK (2010) Glycoprotein 90K, downregulated in advanced colorectal cancer tissues, interacts with CD9/CD82 and suppresses the Wnt/beta-catenin signal via ISGylation of beta-catenin. Gut 59 (7): 907–917
Lee MH, Yang HY (2003) Regulators of G1 cyclin-dependent kinases and cancers. Cancer Metastasis Rev 22 (4): 435–449
Letamendia A, Labbe E, Attisano L (2001) Transcriptional regulation by Smads: crosstalk between the TGF-beta and Wnt pathways. J Bone Joint Surg Am 83-A (Suppl 1) (Part 1): S31–S39
Mansilla F, da Costa KA, Wang S, Kruhoffer M, Lewin TM, Orntoft TF, Coleman RA, Birkenkamp-Demtroder K (2009) Lysophosphatidylcholine acyltransferase 1 (LPCAT1) overexpression in human colorectal cancer. J Mol Med 87 (1): 85–97
Masters JR, Thomson JA, Daly-Burns B, Reid YA, Dirks WG, Packer P, Toji LH, Ohno T, Tanabe H, Arlett CF, Kelland LR, Harrison M, Virmani A, Ward TH, Ayres KL, Debenham PG (2001) Short tandem repeat profiling provides an international reference standard for human cell lines. Proc Natl Acad Sci USA 98 (14): 8012–8017
Matsuzaki S, Tanaka F, Mimori K, Tahara K, Inoue H, Mori M (2009) Clinicopathologic Significance of KIAA1199 Overexpression in Human Gastric Cancer. Ann Surg Oncol 16 (7): 2042–2051
Michishita E, Garces G, Barrett JC, Horikawa I (2006) Upregulation of the KIAA1199 gene is associated with cellular mortality. Cancer Lett 239 (1): 71–77
Nakayama M, Kikuno R, Ohara O (2002) Protein-protein interactions between large proteins: two-hybrid screening using a functionally classified library composed of long cDNAs. Genome Res 12 (11): 1773–1784
Olesen SH, Christensen LL, Sorensen FB, Cabezon T, Laurberg S, Orntoft TF, Birkenkamp-Demtroder K (2005) Human FK506 binding protein 65 is associated with colorectal cancer. Mol Cell Proteomics 4 (4): 534–544
Øster B, Thorsen K, Lamy P, wojdacz TK, hansen LL, Birkenkamp-Demtroder K, Sørensen KD, Laurberg S, Orntoft TF, Andersen C (2011) Identification and validation of highly frequent CpG island hypermethylation in colorectal adenomas and carcinomas. Int J Cancer; e-pub ahead of print 1 April 2011; doi: 10.1002/ijc.25951
Polyak K (1996) Negative regulation of cell growth by TGF beta. Biochim Biophys Acta 1242 (3): 185–199
Sabates-Bellver J, Van der Flier LG, de PM, Cattaneo E, Maake C, Rehrauer H, Laczko E, Kurowski MA, Bujnicki JM, Menigatti M, Luz J, Ranalli TV, Gomes V, Pastorelli A, Faggiani R, Anti M, Jiricny J, Clevers H, Marra G (2007) Transcriptome profile of human colorectal adenomas. Mol Cancer Res 5 (12): 1263–1275
Sidle A, Palaty C, Dirks P, Wiggan O, Kiess M, Gill RM, Wong AK, Hamel PA (1996) Activity of the retinoblastoma family proteins, pRB, p107, and p130, during cellular proliferation and differentiation. Crit Rev Biochem Mol Biol 31 (3): 237–271
Stewart SA, Dykxhoorn DM, Palliser D, Mizuno H, Yu EY, An DS, Sabatini DM, Chen IS, Hahn WC, Sharp PA, Weinberg RA, Novina CD (2003) Lentivirus-delivered stable gene silencing by RNAi in primary cells. RNA 9 (4): 493–501
Stuhler K, Koper K, Pfeiffer K, Tagariello A, Souquet M, Schwarte-Waldhoff I, Hahn SA, Schmiegel W, Meyer HE (2006) Differential proteome analysis of colon carcinoma cell line SW480 after reconstitution of the tumour suppressor Smad4. Anal Bioanal Chem 386 (6): 1603–1612
Thorsen K, Sorensen KD, Brems-Eskildsen AS, Modin C, Gaustadnes M, Hein AM, Kruhoffer M, Laurberg S, Borre M, Wang K, Brunak S, Krainer AR, Torring N, Dyrskjot L, Andersen CL, Orntoft TF (2008) Alternative splicing in colon, bladder, and prostate cancer identified by exon array analysis. Mol Cell Proteomics 7 (7): 1214–1224
Tian X, Du H, Fu X, Li K, Li A, Zhang Y (2009) Smad4 restoration leads to a suppression of Wnt/beta-catenin signaling activity and migration capacity in human colon carcinoma cells. Biochem Biophys Res Commun 380 (3): 478–483
Van der Flier LG, Van Gijn ME, Hatzis P, Kujala P, Haegebarth A, Stange DE, Begthel H, van den Born M, Guryev V, Oving I, van Es JH, Barker N, Peters PJ, van de Wetering M, Clevers H (2009) Transcription factor achaete scute-like 2 controls intestinal stem cell fate. Cell 136 (5): 903–912
Weinacker AS, Chen AS, Agrez MQ, Cones R, Nishimura SS, Wayne E, Pytela SR, Sheppard DS (1994) Role of the integrin alpha v beta 6 in cell attachment to fibronectin. Heterologous expression of intact and secreted forms of the receptor. J Biol Biochem 269 (9): 6940–6948
Zachary IC, Frankel P, Evans IM, Pellet-Many C (2009) The role of neuropilins in cell signalling. Biochem Soc Trans 37 (Part 6): 1171–1178
Acknowledgements
We are grateful to Susanne Bruun and Pamela Celis for excellent technical assistance. We thank Asger Østerberg-Eller for providing the TMA and Laurens van der Flier and Hans Clevers, The Netherlands, for the generous gift of the anti-ASCL2 antibody.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Supplementary Information accompanies the paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Birkenkamp-Demtroder, K., Maghnouj, A., Mansilla, F. et al. Repression of KIAA1199 attenuates Wnt-signalling and decreases the proliferation of colon cancer cells. Br J Cancer 105, 552–561 (2011). https://doi.org/10.1038/bjc.2011.268
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2011.268
Keywords
This article is cited by
-
Downregulation of CEMIP enhances radiosensitivity by promoting DNA damage and apoptosis in colorectal cancer
Medical Oncology (2023)
-
Hypoxia increases KIAA1199/CEMIP expression and enhances cell migration in pancreatic cancer
Scientific Reports (2021)
-
CEMIP, a novel adaptor protein of OGT, promotes colorectal cancer metastasis through glutamine metabolic reprogramming via reciprocal regulation of β-catenin
Oncogene (2021)
-
Secreted KIAA1199 promotes the progression of rheumatoid arthritis by mediating hyaluronic acid degradation in an ANXA1-dependent manner
Cell Death & Disease (2021)
-
A novel metastatic promoter CEMIP and its downstream molecular targets and signaling pathway of cellular migration and invasion in SCLC cells based on proteome analysis
Journal of Cancer Research and Clinical Oncology (2020)