Abstract
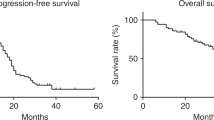
This study describes the efficacy and toxicity of a combination regimen consisting of cyclophosphamide, vincristine (oncovin) and carboplatin (COC) for advanced seminoma on an outpatient basis. Twenty-seven patients (mean age 43 years, range 28-63 years) were classified as stage IIC (n = 5), stage IID (n = 12), stage III (n = 9) or stage IV (n = 1). Six had been treated with prior radiotherapy; elevated beta-HCG and elevated LDH serum levels were observed in 15 and 25 patients respectively. Patients were treated with four cycles of 750 mg m-2 cyclophosphamide intravenously (i.v.), 1.4 mg m-2 vincristine i.v. (maximum 2 mg) and carboplatin adjusted to creatinine clearance. Cycles were given at 3 week intervals. The median dose of carboplatin administered was 400 mg m-2 (range 300-450 mg m-2). Six patients [22%; 95% confidence interval (CI), 6-38%] achieved a complete response (CR), 19 (70%; 95% CI, 51-88%) a partial response and two (8%; 95% CI, 0 18%) showed only a response in tumour markers but not a reduction of retroperitoneal mass (NR). Post-chemotherapeutic masses were not removed surgically or irradiated. After a median follow-up of 26 months (range 5-69 months), two patients have died, one from cardiac arrest 2 years after achieving CR, the other with relapsed seminoma 5 months after therapy. None of the other patients relapsed. Main toxicity was haematological, with 22 patients (81%) experiencing thrombocytopenia WHO grade III/IV and 27 (100%) leucocytopenia WHO grade III/IV, requiring dose reduction in five patients. Seven patients experienced granulocytopenic fever. Non-haematological toxicity was rare. Peripheral neuropathy grade I was observed in four patients and grade III in one. Haemorrhagic cystitis occurred once. In conclusion, despite considerable haematological toxicity, COC is feasible on an outpatient basis, even after prior radiotherapy, and is an effective regimen for advanced seminoma with only 1/27 treatment failures after a median follow-up of 26 months.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sleijfer, S., Willemse, P., de Vries, E. et al. Treatment of advanced seminoma with cyclophosphamide, vincristine and carboplatin on an outpatient basis. Br J Cancer 74, 947–950 (1996). https://doi.org/10.1038/bjc.1996.462
Issue Date:
DOI: https://doi.org/10.1038/bjc.1996.462