Abstract
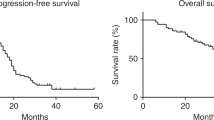
A total of 161 previously untreated patients with FIGO stage III or IV epithelial ovarian cancer were randomised after surgery to receive six courses of either carboplatin 400 mg m-2 alone (Arm A) or carboplatin 300 mg m-2 with chlorambucil 10 mg day-1 for 7 days (Arm B). The median progression free survival (PFS) was similar: arm A: 45 weeks; arm B: 61 weeks (P = 0.830). Multivariate Cox regression analysis showed that the extent of residual disease and performance status were the most important prognostic factors for PFS. Fifty-two per cent of patients received dose escalations based on nadir blood counts, and 89% of all dose adjustments were made according to protocol. Failure to achieve a significant degree of leucopenia was associated with worse progression free survival (P less than 0.001). A total of 29.4% of patients fall into this category. The median survival was similar in both arms, i.e. 75 weeks. It is unlikely that there is any major clinical advantage to adding chlorambucil to single agent carboplatin for the management of advanced ovarian cancer, but whether used in combination or a single agent, the dose of carboplatin should be sufficient to cause at least grade I leucopenia. This may best be achieved by determining the initial dose based on renal function, and then adjusting subsequent doses according to nadir blood counts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rankin, E., Mill, L., Kaye, S. et al. A randomised study comparing standard dose carboplatin with chlorambucil and carboplatin in advanced ovarian cancer. Br J Cancer 65, 275–281 (1992). https://doi.org/10.1038/bjc.1992.55
Issue Date:
DOI: https://doi.org/10.1038/bjc.1992.55
This article is cited by
-
Randomised phase II study comparing dose-escalated weekly paclitaxel vs standard-dose weekly paclitaxel for patients with previously treated advanced gastric cancer
British Journal of Cancer (2014)
-
Timing of chemotherapy-induced neutropenia is a prognostic factor in patients with metastatic non-small-cell lung cancer: a retrospective analysis in gemcitabine-plus-platinum-treated patients
Journal of Cancer Research and Clinical Oncology (2013)
-
First-line treatment for advanced ovarian cancer: paclitaxel, platinum and the evidence
British Journal of Cancer (2002)
-
How to calculate the dose of chemotherapy
British Journal of Cancer (2002)