Abstract
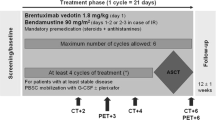
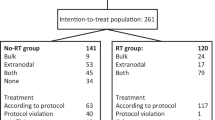
Sixty previously untreated patients with high grade non-Hodgkin's lymphomas stages II-IV received cyclophosphamide 750 mg m2 i.v., doxorubicin 50 mg m2 i.v., and vincristine 2 mg i.v. on day 1, prednisolone 100 mg p.o. on days 1-5 and etoposide 100 mg m2 i.v. on days 3-5 (CHOP-VP16). After four courses an involved field irradiation with a total dose of 25 Gy was employed and followed by two additional courses of CHOP-VP16. The overall response rate was 93%, with 49 patients (82%) achieving a complete remission (CR). Seven patients had a partial response and four patients showed no response. During a median follow-up period of 55 months, 22 of the 49 patients with CR relapsed, seven of them achieving a second complete remission with the same drug regimen. A maintained complete remission of up to 68 months was seen in 55% of all patients initially achieving CR. The median survival is 43 months. Mean side-effects of this drug regimen were alopecia (89%), nausea/vomiting (76%) and leukopenia (61%). No therapy-related deaths were seen. The results of this study demonstrate that this combined modality treatment produces high complete remission rates and that more than half of these patients achieve long-term disease-free survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Köppler, H., Pflüger, K., Eschenbach, I. et al. CHOP-VP16 chemotherapy and involved field irradiation for high grade non-Hodgkin's lymphomas: a phase II multicentre study. Br J Cancer 60, 79–82 (1989). https://doi.org/10.1038/bjc.1989.224
Issue Date:
DOI: https://doi.org/10.1038/bjc.1989.224
This article is cited by
-
Significant dose Escalation of Idarubicin in the treatment of aggressive Non- Hodgkin Lymphoma leads to increased hematotoxicity without improvement in efficacy in comparison to standard CHOEP-14: 9-year follow up results of the CIVEP trial of the DSHNHL
SpringerPlus (2014)
-
Pharmacokinetic comparison of oral and intravenous etoposide in patients treated with the CHOEP-regimen for malignant lymphomas
Cancer Chemotherapy and Pharmacology (2008)
-
CHOP plus etoposide and gemcitabine (CHOP-EG) as front-line chemotherapy for patients with peripheral T cell lymphomas
Cancer Chemotherapy and Pharmacology (2006)
-
Stellenwert der Strahlentherapie beim disseminierten hochmalignen Non-Hodgkin-Lymphom
Strahlentherapie und Onkologie (1997)
-
A phase I trial of high-dose oral tamoxifen and CHOPE
Cancer Chemotherapy and Pharmacology (1995)