Abstract
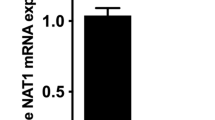
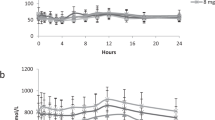
The urinary excretion during 24 h of aminoglutethimide (AG) its major metabolite (N-acetylAG) and two minor metabolites (N-formylAG and nitroG) were measured in 10 volunteers given AG who had been typed for acetylator phenotype using sulphadimidine. The slow acetylators of sulphadimidine excreted more AG (mean 28% of the administered dose) than did the fast acetylators (12%), but the latter excreted more of the dose as N-acetylAG (8.8%) than did the former (3.9%). NitroG and N-formylAG were minor urinary metabolites of AG in humans. The former was more abundant in the urine of slow acetylators (0.10% of the dose) than in that of fast acetylators (0.047%), whereas the respective proportions of doses excreted as the N-formyl derivative (0.475 and 0.465%) were not significantly different for the two acetylator phenotypes. These results show that AG is among those drugs that are polymorphically acetylated in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Rights and permissions
About this article
Cite this article
Coombes, R., Foster, A., Harland, S. et al. Polymorphically acetylated aminoglutethimide in humans. Br J Cancer 46, 340–345 (1982). https://doi.org/10.1038/bjc.1982.209
Issue Date:
DOI: https://doi.org/10.1038/bjc.1982.209
This article is cited by
-
Aminoglutethimide in advanced breast cancer: plasma levels and clinical results after low and high doses
Cancer Chemotherapy and Pharmacology (1991)
-
Could aminoglutethimide replace adrenalectomy?
Breast Cancer Research and Treatment (1985)