Abstract
The importance of tacrolimus in the treatment of myasthenia gravis (MG) as a substitute for corticosteroid-dependent immunosuppressive therapy is increasing. Thus far, however, no population pharmacokinetic (PopPK) analysis of tacrolimus in treating MG patients has been published. This article aimed to construct a PopPK model of tacrolimus for Chinese MG patients with the goal of improving its performance in MG treatment. A total of 253 trough concentration records were obtained from 83 Chinese MG patients. The effects of demographics, lifestyle and health status, biochemical test data, disease progression and treatment-related information (including co-administered medications) as covariates on the various parameters were investigated. The covariate selection was based on biological plausibility, clinical significance, statistical significance and reduction in inter-individual variability (IIV). Bootstrap and normalized prediction distribution error (NPDE) analysis were performed to validate the final model. A one-compartment PopPK model with first-order elimination and a fixed absorption phase was constructed. The estimated apparent oral clearance (CL/F) and apparent oral volume of distribution (V/F) were 3.6 L/h and 1700 L, respectively, in the MG patients. Hematocrit and blood urea nitrogen were identified as two covariates that significantly influenced the CL/F. Immunoglobulin treatment (PRO) also had the potential to influence V/F, which was consistent with the clinical observations and the high protein-binding property of tacrolimus. Other covariates including age, weight, gender and co-administered medications had no obvious influence on CL/F or V/F. The first PopPK model of tacrolimus in MG patients was established. The identified covariates were of biological plausibility and clinical importance to help individualize the dosing schedule in MG patients.
Similar content being viewed by others
Introduction
Tacrolimus is a potent immunosuppressant that is used to prevent organ rejection after transplantation. It inhibits T-lymphocyte activation, thereby suppressing humoral immunity1. In addition to being a first-line drug in organ rejection treatment, the importance of tacrolimus in the treatment of myasthenia gravis (MG) is also increasing on the basis of its potential capacity to alleviate MG symptoms and to relieve steroid burden2. MG is an autoimmune disease for which corticosteroids are the main therapeutic option for immune suppression3. However, long-term use of steroids is known to cause severe adverse effects. As a potent immune-suppressant, a low dose of tacrolimus was reported to be effective for steroid-dependent MG patients4. Therefore, tacrolimus has been approved in Japan for MG patients who have not responded well to prednisolone or have experienced substantial adverse effects from hormone therapies. Later, a randomized, double-blind, placebo-controlled clinical trial that included 80 patients further confirmed the steroid-sparing effect of tacrolimus at a dose of 3 mg/d5. In China, tacrolimus is recommended as an immunosuppressant to replace hormone therapy for the treatment of MG. Because it is a potent immunosuppressant with relatively few side effects, low-dose tacrolimus has become an important therapeutic option for Chinese MG patients. In addition, the empirical therapeutic window for MG in Chinese hospitals is 4–8 ng/mL6. The dosing regimen and therapeutic window of tacrolimus for MG patients is very different from those for transplant patients. Previous literature has described some nonlinear PK properties of tacrolimus7. Therefore, with a different dosing regimen and patient population, a different PK profile is very likely to occur when tacrolimus is administered to MG patients.
Because tacrolimus is known for its narrow therapeutic window8 and acute over-dosage of tacrolimus is accompanied by severe side effects9, it is important to investigate the drug's PK behaviors in the context of the treatment of MG. A novel approach to achieve this is to exploit the strong predictive capacity of population pharmacokinetic (PopPK) models and routinely collected therapeutic monitoring (TDM) data. Although various population models have been developed for tacrolimus in patients receiving organ transplantation10, no PopPK model of tacrolimus focusing on MG patients under such low dose levels has been reported. Therefore, we aimed to construct a PopPK model of tacrolimus for MG patients at a lower dose, with the goal of improving its performance in MG treatment and to optimize the dosing regimen.
Materials and methods
Subjects
This study retrospectively investigated 83 MG patients receiving tacrolimus as a treatment in the General Hospital of PLA Rocket Force, Beijiing, China, from January 2011 to May 2015. All subjects provided written informed consent. The study was approved by the Ethics Committee of the General Hospital of PLA Rocket Force. The certificate number is KY2016023. All included subjects had been clinically diagnosed with MG and were currently receiving orally administered tacrolimus as the main immunosuppressant. Pregnant women and sexually active women who were not willing to practice contraception were excluded. We also excluded one individual with highly unlikely dosing records, which were likely due to human errors, and three observation records for one individual in which all the covariate information was missing.
MG treatment and drug therapy
Most patients were admitted to the hospital and discharged several times over the investigated time range. The patients were administered tacrolimus orally during the hospital stays. Once admitted, the patients underwent routine blood tests and biochemical tests for renal function and hepatic function. TDM was performed four days after the initial dose. The doses were then adjusted based on the TDM results and clinical symptoms. Patients were discharged after the disease was under control. Most patients were discharged after one or two rounds of four-day drug administration; ie, most patients had one or two observation points per admission. On average, the patients were admitted for four times over the investigated time range.
Tacrolimus (Prograf®, FK506) capsules, 0.5 mg (Astellas Ireland Co, Ltd, Killorglin, Co, Kerry, Ireland), was used. The dosing regimens were quite variable. For patients who had no previous tacrolimus dosing records, the initial dose was 1 mg/d for adults and 0.5 mg/d for pediatric patients. For those with previous tacrolimus dosing records, the initial dose was based on those records. Doses were raised or lowered by 0.5 mg/d. All scheduled dosing intervals were 12 h. The actual dosing times varied.
To alleviate MG symptoms, half of tacrolimus dosing events were accompanied by the administration of pyridostigmine. Around 12% of the patients were switching from hormone-dependent immunotherapy to tacrolimus-dependent immunotherapy. For those patients, dosing events were accompanied by corticosteroids (prednisone or methylprednisolone). Patients who were co-administered antihypertensive drug or hypoglycemic drugs accounted for less than 5% of the total investigated population. These drugs were therefore not included in the drug drug interactions (DDI) covariate model.
Bioanalytics
The method for determining tacrolimus concentrations in whole-blood samples has been previously reported11. Briefly, tacrolimus and the internal standard were extracted from whole blood using acetonitrile and then separated on a Shim-pack VP-ODS column (5 μm, 150 mm×4.6 mm). A 20 μL aliquot of the residue was injected into the LC–MS/MS system. The assay was confirmed to be linear over the range of 0.1–25 ng/mL and had a lower limit of quantification of 0.1 ng/mL and a limit of detection of 0.05 ng/mL. With respect to assay precision, the intra-day CV (coefficient of variation) was 3.37% and the inter-day CV was 4.09%. The assay accuracy assessment showed a relative error in the range of -2.99% to 2.23%. The tacrolimus concentrations in the supplementary dataset were measured using the same system.
Covariates
The carefully recorded population information that was subsequently investigated in the covariate model included demographic information, lifestyle information, health status information, lab test results and treatment-related information (Table 1). The demographic information included age (AGE), gender (SEX), weight (WT), and body mass index (BMI). The lifestyle information revealed whether the subjects drank alcohol (ALCO) or smoked cigarettes (TOBA). The health status information indicated whether the subjects had diabetes (DIAB) or hypertension (HYPE). The biochemical test data included the following items: total bilirubin (TBIL), alkaline phosphatase (ALP), aspartate aminotransferase (AST), alanine aminotransferase (ALT), blood urea nitrogen (BUN), serum creatinine (SCR), white blood cells (WBC), red blood cells (RBC), hemoglobin (HGB), hematocrit (HCT), mean corpuscular volume (MCV), percent neutrophilic granulocytes (NP), and percent lymphocytes (LP). The treatments that the patients had received, which included thymectomy (THYM) and high-dose intravenous immunoglobulin treatment (PRO), were also investigated as important covariates that could influence the parameters. The disease status (DIAG) was also included as a potential covariate. Osserman and Genkins' modified classification standard12 divides MG severity into 5 levels (6 subtypes): Class I, Class IIA, Class IIB, Class III, Class IV and Class V. The categorical covariate DIAG was designated 1, 2, 2.5, 3, 4 and 5 for convenience. Since the patients were classified into different subtypes according to clinical symptoms and the difference between adjacent subtypes was unmeasurable, DIAG was included as a categorical covariate instead of a continuous covariate. To investigate potential drug-drug interactions, pyridostigmine and corticosteroids were chosen as two drugs that could potentially influence the PK of tacrolimus. The corresponding dichotomous covariates DDIA and DDIB were assigned the value of 1 if the patients were co-administered pyridostigmine and corticosteroids, respectively; otherwise the value was 0.
We performed imputation for 35 missing records of BMI and weight information via the following method. For the individuals whose information was missing for certain admission times but available for other admission times, the imputed missing values were assigned the nearest value available. For individuals for whom there was no information for any admission time, the missing values were imputed as the median value of BMI or weight.
Population model construction
The model was constructed using NONMEM (version 7.2.0, ICON Development Solutions, Ellicott, MD, USA), using PsN toolkit 3.7.613 and Piranã version 2.8.114 as the modeling environment. The basic data analysis was performed using the statistical software package R (v3.2.3). The first-order conditional estimation with ɛ–η interaction (FOCE-I) was used to estimate the parameters including typical population pharmacokinetic parameters, random inter-individual variability in these parameters, and residual variability between observed and predicted concentrations. Correlations between the components of the variability were also tested.
One- and two-compartment models with first-order elimination were fitted to the patients' data. Since the patients' data were not informative regarding the absorption process, the parameters describing absorption were set to the fixed values obtained from the supplementary dataset of healthy volunteers, assuming a similar absorption process. The rationale of fixing the absorption process is further described in the discussion section. The inter-individual variability (IIV) for all parameters was modeled using exponential variance models. A constant coefficient of variation (CCV) error model, an additive model and mixed model were included successively to try to describe the residual variability.
To perform covariate analysis, we considered the four main criteria proposed by Duffull and his colleagues: (1) biological plausibility, (2) clinical significance, (3) statistical significance (e g, from a likelihood ratio test) and (4) a reduction in IIV15. Therefore, we first calculated the statistical significance via a likelihood ratio test (LRT) and IIV reduction after one covariate was added into the model. After statistical calculation, we obtained a full model with several covariates; we then manually excluded those that were biologically irrelevant to the PK process or clinically inapplicable. Statistical analysis was automatically performed using the SCM function in PsN toolkit 3.7.6, through which all the covariates mentioned above were screened in a stepwise manner that included the processes of forward inclusion and backward elimination. The statistical effects of the covariates were evaluated using the difference in the OFV. A difference greater than 3.84 (P<0.05, df=1) in forward inclusion and larger than 6.64 (P<0.01, df=1) in backward elimination was considered significant. During the modeling, the categorical covariates were transformed into several flag dichotomous covariates. The influence of continuous covariates and that of dichotomous covariates on various parameters were included via the power model described by Equation 1 and the shift model described in Equation 2, respectively, where Pi represents a parameter of the ith subject; θTV represents the typical value of the parameter; Covariatei represents the value of the covariate for the ith subject; CovariateMedian represents the median value of one covariate; ηi represents the individual value of the parameter's IIV effect and θCOV is the parameter to be estimated in the covariate model.


Model selection and evaluation
Model selection was based on several criteria, including OFV, goodness-of-fit (GOF) diagnostic plots and the precision of the parameter estimates. The GOF diagnostic plots included the observations (OBS) versus individual predictions (IPRED), OBS versus population predictions (PRED), conditional weighted residuals (CWRES) versus PRED and CWRES versus time since the last dose (TSLD). The precision of the parameter estimates was described as the percent relative standard error (%RSE). To further evaluate the parameter precision and stability, a non-parametric bootstrap analysis was conducted, during which the model was fitted to 1000 resampled datasets. To evaluate the model's predictive performance, a normalized prediction distribution errors (NPDE) test was also performed. For NPDE, 3000 simulations were performed for each observation based on the final model, then the NPDE value for each observation was calculated. The distribution of NPDEs is expected to follow a standard normal distribution if the final model is appropriate. The simulation was performed in NONMEM, and three statistical tests were performed using the add-on R package NPDE (v2.0)16 to test the null hypothesis.
Results
Data for analysis
The tacrolimus whole-blood concentration data (253 observations) and patients' basic information were available for a total of 83 MG patients (Table 1). The whole-blood concentration-time curves of tacrolimus are presented in Figure 1. Since the dosing regimen and sample collection schedule were highly variable, both time since the first dose (TSFD) (Figure 1A, 1B) and time since the last dose (TSLD) (Figure 1C, 1D) were used as the x-axes. The tacrolimus concentrations were normalized to the accumulated dose (mg) before sample collection. The dataset used to supplement the absorption phase is shown in the supplementary information (Supplementary Figure S1).
Dose-normalized tacrolimus concentration-time profiles. The tacrolimus concentration was normalized by the accumulated dose (mg) before sample collection.
Population pharmacokinetic model
The MG patients' data were best described by a one-compartment model with first-order elimination and absorption. Although tacrolimus is usually modeled using a two-compartment model7, this model failed to converge due to a limited number of observations. Therefore, the one-compartment model was finally adopted as the model for the MG patients. The model was finally parameterized in terms of apparent clearance (CL/F, where F is bioavailability), apparent volume of distribution (V/F), absorption rate constant (Ka) and lag time (Tlag). Ka and Tlag were fixed to the values obtained from the supplementary dataset (Table 2).
After automatic forward inclusion and backward elimination, HCT, LP and BUN on CL/F were included as statistically significant covariates (Table 3). We then investigated the biological plausibility and clinical importance of these three included covariates. Because no previously published findings were found to link the percent lymphocytes (LP) to a drug's pharmacokinetic behaviors, LP was eliminated as a covariate after this investigation. In addition, the final model with HCT and BUN as two covariates actually had a lower IIV for the CL/F than the three-covariate model (Table 3), which further supported this decision. After the covariate analysis, CL/F was expressed as Equation 3, where (CL/F)i represents the CL/F value of the ith individual; θCL/F represents the typical value of CL/F; ηi represents the individual value of the parameter's IIV effects; HCTi, BUNi, HCTMedian, BUNMedian, θHCT and θBUN represent the HCT and BUN values of the ith individual, the median values for HCT and BUN and the covariate coefficient for HCT and BUN, respectively. The estimates of the parameters of the final model after covariate inclusion are presented in Table 2.

There was a correlation between the variability of CL/F and V/F in the base model. However, this correlation disappeared after the inclusion of covariates, possibly because the correlation was explained by the addition of covariates17.
The results of non-parametric bootstrap analysis are presented in Table 2. In the bootstrap procedure, the percentage of successful minimization rate was 99%. The pharmacokinetic parameters from the final population model were all included in the 90% confidence interval (CI) of the bootstrap procedure. The bootstrap procedure proved the model parameters' reliability and stability.
The goodness-of-fit plots of the final model showed a good fit to most of the data and no obvious bias (Figure 2). Although some under-predictions for the high concentration points were observed (Figure 2B, 2C), the plot of the conditional weighted residuals (CWRES) versus time since last dose (TSLD) (Figure 2D) was symmetrical, and the absolute values of CWRES were almost all within 3 units, which indicated a good fit of the model to the data.
Goodness-of-fit plots for the final tacrolimus model in MG patients. The solid lines in (A) and (B) are identity lines and the solid lines in (C) and (D) are zero lines. The dashed lines represent regression lines via locally weighted polynomial regression (LOESS).
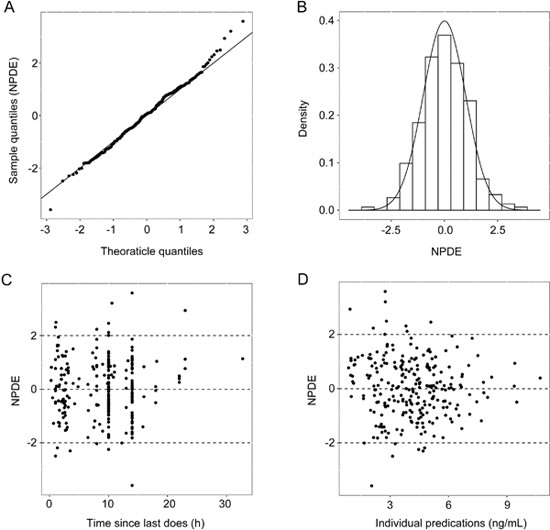
Additional model validation was conducted using the NPDE test (Figure 3). Statistical evaluation of the NPDE distribution showed that the mean of the NPDE was not significantly different from 0 (Wilcoxon signed rank test, P=0.59), the variance was not significantly different from 1 (Fisher test, P=0.0563) and the NPDE distribution was not significantly different from a normal distribution (Shapiro-Wilks test of normality, P=0.609), thereby confirming that the model adequately described the observed data. No trends were visible for the NPDE versus time (Figure 3C) or the predicted concentration (Figure 3D). The NPDE test confirmed the good predictive performance of the final model.
Normalized prediction distribution error (NPDE) plots of the final model. (A) Quantile-quantile plot of the distribution of the NPDE against the theoretical distribution (the identity line); (B) Histogram of the distribution of the NPDE against the theoretical distribution (the normal distribution curve); (C) NPDE vs time since first dose and (D) NPDE vs individual predictions.
Discussion
The pharmacokinetic behavior of tacrolimus in MG patients was described using a population model that consisted of a one-compartment model with first-order elimination and a fixed absorption phase. Although tacrolimus is usually modeled as a two-compartment model7, this model failed to converge due to the limited informative nature of the dataset. Given that the two-compartment model can be simplified to a one-compartment model by integrating the central compartment and the peripheral compartment, the one-compartment model was ultimately used to describe the drug's pharmacokinetic behaviors. Since tacrolimus is used as a long-term medication and its half-life is more than ten-fold longer than the peak time, the elimination phase of the drug is of greater clinical importance than the distribution phase. In addition, although we fixed the absorption phase, a previous methodological study reported that the misspecification of absorption process has little effect on the estimation of subsequent parameters in the one-compartment model18. Therefore, the population pharmacokinetic study presented here is appropriate and of clinical importance.
The elimination process deserves the most attention. Compared with other PopPK analysis of tacrolimus in various subpopulations using the same structural model and quantification method, the clearance of tacrolimus in the MG patients appeared to be lower than that in liver transplant patients19, kidney transplant recipients20 and even healthy volunteers9 (Table 4). Although inaccuracy in the calculation of the clearance due to the limitation of sparse data is a possible explanation, our team managed to identify some clues to another physiologically possible explanation. A previous study confirmed the weakness of the myocardium in some MG patients via clinical indications, ECG, vectorcardiography and autopsy data21. Further studies demonstrated that the striated cardiac muscle could be a target for an immune attack manifesting as heart failure, arrhythmia, and sudden death22. In addition, a prospective clinical study at Johns Hopkins Hospital indicated that patients with heart failure had a significantly lower clearance of lidocaine than patients without heart failure (P<0.005)23. The reduced blood flow in the eliminating organs was probably the cause of the lower clearance value in the heart failure patients. Therefore, we inferred that the lower CL/F value in MG patients could be attributed to reduced blood flow in the eliminating organs due to myocardial fatigue in that population.
Hematocrit (HCT) was the most commonly included covariate on CL/F according to the previously published PopPK analysis of tacrolimus7. Because tacrolimus has a strong association with erythrocytes, it is logical to expect that hematocrit influences drug clearance. Blood urea nitrogen (BUN) is a conventional value to express the urea level in blood. Reidenberg and his colleagues reported that unbound drug (diphenylhydantoin) levels in plasma were positively correlated with individual BUN values24. Subsequently, decreased binding in uremic (high BUN value) plasma has been demonstrated for several other drugs25. Later, a possible molecular mechanism for this phenomenon was published: blood urea was believed to promote protein carbamylation26,27, which was reported to increase the free drug level28. Considering that the protein binding ratio of tacrolimus is approximately 99%, it is reasonable to expect more unbound tacrolimus to be present in the plasma of the patients with higher BUN values. In this way, it is physiologically plausible that clearance will increase as a function of the blood urea level.
In addition to the two important covariates mentioned above, the most frequently prescribed medicine for the treatment of MG, pyridostigmine, was evaluated as a covariate, and this drug showed no significant influence on the PK behavior of tacrolimus in this study. In addition, in patients who were switching from hormone therapy to tacrolimus, corticosteroids did not affect the drug's PK. This result may further simplify the administration of tacrolimus in MG management.
Another interesting result from the covariate screening was the effect of high-dose intravenous immunoglobulin treatment (PRO) on the V/F. Although the inclusion of PRO as a covariate led to a reduction of only 5.9 units in OFV (P=0.015, chi-squared test), which failed to meet the criterion for backward elimination, the OFV reduction did indicate some effect of PRO on V/F. This phenomenon further confirmed the high protein-binding property of tacrolimus. However, the frequency of PRO as a covariate was only 7.9% (Table 1). When the frequency of a covariate is low (<10%), the type I error can be inflated, which makes it difficult to determine the significance via the likelihood ratio test. The significance of PRO as a covariate on V/F may be greater than that calculated in this study29. In addition, patients whose tacrolimus administration was accompanied by the PRO treatment demonstrated a significantly higher dose-normalized concentration (P=0.01078, t-test) compared with those who did not receive the PRO treatment (Figure 4). This clinical observation may suggest a different pharmacokinetic behavior in PRO-treated patients. Therefore, PRO has a potential to become an influential covariate on V/F.
Boxplot of normalized concentration vs PRO (immunoglobulin treatment). The upper, middle and lower lines of the box represent the 75% quantile, median and 25% quantile of the data, respectively. The black points represent the outliers beyond the end of the whiskers. **P=0.01078. t-test.
Because the age span of the study cohort was large, we paid specific attention to the covariate AGE. Based on the final model, the pediatric group (AGE<16 years) and non-pediatric group (AGE≥16 years) had significantly different CL/F values (Table 5). However, we have tried to add AGE as a continuous covariate and age group as a categorical covariate in the models for CL/F and V/F. None of the models produced a significant decrease in OFV (dOFV<2). In addition, none of them produced a better GOF plot than that produced by the original model. This result may be attributable to the low number of individuals in the pediatric group. Specifically, adults accounted for 75% of the total 253 observation points.
Although we evaluated a large number of covariates, data for the genetic polymorphisms of the CYP enzymes were not collected. We attempted to model the influence of polymorphisms using a mixture model, but the estimated parameters were unstable and near the boundary of zero. Therefore, we decided to abandon the mixture model. The failure to include genetic polymorphisms was one possible cause of the IIV in our model. A future study may evaluate the influence of genetic polymorphisms.
In conclusion, this was the first PopPK study of tacrolimus in MG patients. We demonstrated that MG patients had a lower clearance rate than that in other subpopulations. We further identified HCT (hematocrit) and BUN (blood urea nitrogen) as two important covariates and PRO (high-dose intravenous immunoglobulin treatment) as a potential covariate. Given the emerging role of tacrolimus in the treatment of MG, this study serves as a useful reference for physicians in determining an individual-specific dosing regimen.
Author contribution
Ping MA and Wei LU designed the research; Ping MA, Zi-qi LIU, and Ling HUANG collected the data and performed the measurements; Yu-si CHEN, Rong CHEN, Xiao ZHU, and Zi-qi LIU graphically explored the data and conducted the PopPK analysis; Yu-si CHEN drafted the manuscript; Ping MA, Wei LU, Rong CHEN, and Yu-si CHEN revised the manuscript; Lei WANG provided clinical consultations; Tian-yan ZHOU provided technical support.
References
Thomson AW, Bonham CA, Zeevi A . Mode of action of tacrolimus (FK506): molecular and cellular mechanisms. Ther Drug Monit 1995; 17: 584–91.
Cruz JL, Wolff ML, Vanderman AJ, Brown JN . The emerging role of tacrolimus in myasthenia gravis. Ther Adv Neurol Disord 2015; 8: 92–103.
Skeie G, Apostolski S, Evoli A, Gilhus N, Hart I, Harms L, et al. Guidelines for the treatment of autoimmune neuromuscular transmission disorders. Eur J Neurol 2006; 13: 691–9.
Ponseti JM, Gamez J, Azem J, López-Cano M, Vilallonga R, Armengol M . Tacrolimus for myasthenia gravis. Ann N Y Acad Sci 2008; 1132: 254–63.
Yoshikawa H, Kiuchi T, Saida T, Takamori M . Randomised, double-blind, placebo-controlled study of tacrolimus in myasthenia gravis. J Neurol Neurosurg Psychiatry 2011; 82: 970–7.
Group of Neuro-Immunity DoN, Chinese Medical Association. Consensus on the treatment and diagnosis of myasthenia gravis in China. Chin J Neuroimmunol Neurol 2011; 18: 386–72.
Zhao CY, Jiao Z, Mao JJ, Qiu XY . External evaluation of published population pharmacokinetic models of tacrolimus in adult renal transplant recipients. Br J Clin Pharmacol 2016; 81: 891–907.
Beckebaum S, Cicinnati VR, Radtke A, Kabar I . Calcineurin inhibitors in liver transplantation – still champions or threatened by serious competitors? Liver Int 2013; 33: 656–65.
(FDA) UFaDA. Prograf®: tacrolimus capsules/ tacrolimus injection (for intravenous infusion only). ed. Available from http://www.accessdata.fda.gov/drugsatfda_docs/label/2009/050708s027,050709s021lbl.pdf [revised May 2009; cited August 2016].
Brooks E, Tett SE, Isbel NM, Staatz CE . Population pharmacokinetic modelling and bayesian estimation of tacrolimus exposure: is this clinically useful for dosage prediction yet? Clin Pharmacokinet 2016; 55: 1295–335.
Li P, Liu L, Ma P . Ding Cl, Tong WH, Zhang XF, et al. LC-MS/MS determination of tacrolimus in human blood and application in its bioequivalence study. Chin J Pharm Anal 2009; 29: 237–42.
Osserman KE, Genkins G . Studies in myasthenia gravis: review of a twenty-year experience in over 1200 patients. Mt Sinai J Med 1971; 38: 497–537.
Lindbom L, Pihlgren P, Jonsson N . PsN-Toolkit — a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Programs Biomed 2005; 79: 241–57.
Keizer RJ, Van Benten M, Beijnen JH, Schellens JH, Huitema AD . Pirana and PCluster: a modeling environment and cluster infrastructure for NONMEM. Comput Methods Programs Biomed 2011; 101: 72–9.
Duffull SB, Wright DF, Winter HR . Interpreting population pharmacokinetic pharmacodynamic analyses — a clinical viewpoint. Br J Clin Pharmacol 2011; 71: 807–14.
Comets E, Brendel K, Mentré F . Computing normalised prediction distribution errors to evaluate nonlinear mixed-effect models: the npde add-on package for R. Comput Methods Programs Biomed 2008; 90: 154–66.
Byon W, Smith M, Chan P, Tortorici M, Riley S, Dai H, et al. Establishing best practices and guidance in population modeling: an experience with an internal population pharmacokinetic analysis guidance. CPT Pharmacometrics Syst Pharmacol 2013; 2: e51.
Wade JR, Kelman AW, Howie CA, Whiting B . Effect of misspecification of the absorption process on subsequent parameter estimation in population analysis. J Pharmacokinet Biopharm 1993; 21: 209–22.
Staatz CE, Willis C, Taylor PJ, Lynch SV, Tett SE . Toward better outcomes with tacrolimus therapy: population pharmacokinetics and individualized dosage prediction in adult liver transplantation. Liver Transpl 2003; 9: 130–7.
Staatz CE, Willis C, Taylor PJ, Tett SE . Population pharmacokinetics of tacrolimus in adult kidney transplant recipients. Clin Pharmacol Ther 2002; 72: 660–9.
Hofstad H, Ohm OJ, Mørk SJ, Aarli JA . Heart disease in myasthenia gravis. Acta Neurol Scand 1984; 70: 176–84.
Shivamurthy P, Parker MW . Cardiac manifestations of myasthenia gravis: A systematic review. IJC Metab Endocr 2014; 5: 3–6.
Zito RA, Reid PR . Lidocaine kinetics predicted by indocyanine green clearance. N Engl J Med 1978; 298: 1160–3.
Reidenberg MM, Odar-Cederlöf I, von Bahr C, Borgå O, Sjöqvist F . Protein binding of diphenylhydantoin and desmethylimipramine in plasma from patients with poor renal function. N Engl J Med 1971; 285: 264–7.
Sjöholm I, Kober A, Odar-Cederlöf I, Borgå O . Protein binding of drugs in uremic and normal serum: The role of endogenous binding inhibitors. Biochem Pharmacol 1976; 25: 1205–13.
Stark GR . Reactions of cyanate with functional groups of proteins. II. Formation, decomposition, and properties of N-carbamylimidazole*. Biochemistry 1965; 4: 588–95.
Stark GR, Stein WH, Moore S . Reactions of the cyanate present in aqueous urea with amino acids and proteins. J Biol Chem 1960; 235: 3177–81.
Erill S, Calvo R, Carlos R . Plasma protein carbamylation and decreased acidic drug protein binding in uremia. Clin Pharmacol Ther 1980; 27: 612–8.
Lagishetty CV, Duffull SB . Evaluation of approaches to deal with low-frequency nuisance covariates in population pharmacokinetic analyses. AAPS J 2015; 17: 1388–94.
Acknowledgements
We thank Dr Zheng JIAO from Huashan Hospital, Shanghai, for his advice on model construction. We also thank Tong-tong JIA for collecting patient information.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary information is available on the website of Acta Pharmacologica Sinica.
Supplementary information
Supplementary Figure 1
Tacrolimus concentration-time curve of the supplementary dataset. (DOC 11 kb)
Rights and permissions
About this article
Cite this article
Chen, Ys., Liu, Zq., Chen, R. et al. Population pharmacokinetic analysis of tacrolimus in Chinese myasthenia gravis patients. Acta Pharmacol Sin 38, 1195–1204 (2017). https://doi.org/10.1038/aps.2016.174
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2016.174
Keywords
This article is cited by
-
Population pharmacokinetic analyses of tacrolimus in non-transplant patients: a systematic review
European Journal of Clinical Pharmacology (2023)
-
Population Pharmacokinetic Evaluation with External Validation of Tacrolimus in Chinese Primary Nephrotic Syndrome Patients
Pharmaceutical Research (2022)
-
Multiple genetic factors affecting the pharmacokinetic and pharmacodynamic processes of tacrolimus in Chinese myasthenia gravis patients
European Journal of Clinical Pharmacology (2020)
-
Population Pharmacokinetic Analysis of Tacrolimus in Adult Chinese Patients with Myasthenia Gravis: A Prospective Study
European Journal of Drug Metabolism and Pharmacokinetics (2020)