Abstract
Aim:
To investigate the effects of piperine, a major pungent alkaloid present in Piper nigrum and Piper longum, on the tumor growth and metastasis of mouse 4T1 mammary carcinoma in vitro and in vivo, and elucidate the underlying mechanisms.
Methods:
Methods: Growth of 4T1 cells was assessed using MTT assay. Apoptosis and cell cycle of 4T1 cells were evaluated with flow cytometry, and the related proteins were examined using Western blotting. Real-time quantitative PCR was applied to detect the expression of matrix metalloproteinases (MMPs). A highly malignant, spontaneously metastasizing 4T1 mouse mammary carcinoma model was used to evaluate the in vivo antitumor activity. Piperine was injected into tumors every 3 d for 3 times.
Results:
Results: Piperine (35–280 μmol/L) inhibited the growth of 4T1 cells in time- and dose-dependent manners (the IC50 values were 105±1.08 and 78.52±1.06 μmol/L, respectively, at 48 and 72 h). Treatment of 4T1 cells with piperine (70–280 μmol/L) dose-dependently induced apoptosis of 4T1 cells, accompanying activation of caspase 3. The cells treated with piperine (140 and 280 μmol/L) significantly increased the percentage of cells in G2/M phase with a reduction in the expression of cyclin B1. Piperine (140 and 280 μmol/L) significantly decreased the expression of MMP-9 and MMP-13, and inhibited 4T1 cell migration in vitro. Injection of piperine (2.5 and 5 mg/kg) dose-dependently suppressed the primary 4T1 tumor growth and injection of piperine (5 mg/kg) significantly inhibited the lung metastasis.
Conclusion:
Conclusion: These results demonstrated that piperine is an effective antitumor compound in vitro and in vivo, and has the potential to be developed as a new anticancer drug.
Similar content being viewed by others
Introduction
Piperine (Figure 1) is a major pungent alkaloid present in black pepper (Piper nigrum) and long pepper (Piper longum). It exhibits a wide variety of biological effects. Piperine could inhibit both the drug transporter P-glycoprotein and the major drug-metabolizing enzyme CYP3A41, 2, so it has been used as a bioavailability enhancer with various structurally and therapeutically diverse drugs3. Piperine also had anti-inflammatory, anti-nociceptive, and anti-arthritic effects by inhibiting the expression of interleukin 6 (IL-6), matrix metalloproteinases 13 (MMP-13) and prostaglandin E2 (PGE2) in an arthritis animal model4 or by inhibiting tumor necrosis factor-α (TNF-α) induced activation of NF-κB via blocking IκBα kinase activation5. Additionally, piperine also possessed anti-depression like activity, cognitive enhancing effect6, a blood pressure-lowering effect7, anti-oxidative, anti-apoptotic and chemo-protective ability in blastogenesis8. More of the diverse physiological effects of piperine have been reported in recent decades9.
Concerning the antitumor effects, piperine has been reported to inhibit lung metastasis induced by B16F-10 melanoma cells10, and the anti-invasive effects of piperine on fibrosarcoma cells also have been demonstrated11. The therapeutic properties of piperine with other antitumor drugs were tested in various cell types12, 13. But there is little known about the anticancer activities of piperine on mammary cancer cells and the underlying mechanism.
The 4T1 tumors closely mimics human breast cancer in its anatomical site, immunogenicity and growth characteristics14. After sc inoculation in the abdominal mammary fat pad, the primary tumor grows into a nodule with the histology of a high-grade breast cancer and sheds spontaneous systemic metastases. Metastatic growth in the lungs is usually the main cause of death of mice. The pattern and histological appearance of such metastases are similar to what is seen in humans, therefore, this model is suitable for testing the effects of experimental therapies on metastatic disease. In the present study, we observed the effects of piperine on the tumor growth and metastasis of 4T1 breast cancer in vitro and in vivo. And the possible mechanism for the inhibitory effect of piperine on tumor cell growth and migration were investigated. The anti-tumor efficacy makes piperine a potential candidate for future cancer therapy.
Materials and methods
Materials
Piperine (molecular weight, 285.35) was purchased from Fluka (St Louis, MO, USA). A 50 mg/mL stock solution of piperine was prepared in DMSO and then further diluted in cell culture medium or PBS. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was purchased from Sigma (St Louis, MO, USA). Annexin-V FITC Apoptosis Detection Kit was purchased from R&D systerms (Minneapolis, MN, USA). Reverse transcription enzymes and SYBR Green detection chemistry were obtained from Takara Bio Inc (Japan). Anti-Bcl-2 (sc-7382), anti-Bax (sc-526), anti-cyclin D1 (sc-8396), anti-cyclin B1 (sc-595), anti-β-actin (sc-4778) and IgG-HRP were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Anti-caspase 3 (1087-1) was purchased from Epitomics (Epitomics, CA, USA). All chemicals and reagents were of analytical grade.
Cell lines
4T1 mouse mammary carcinoma cells were a kind gift from Prof Yang-xin FU (Chicago University, Chicago, IL, USA). NIH3T3 (murine fibroblast cell line) cells were obtained from American Type Culture Collection (ATCC, Manassas, VT, USA). Cells were cultured in RPMI-1640 supplemented with 10% fetal bovine serum (FBS). Cultures were maintained in a humidified atmosphere with 5% CO2 at 37 °C.
Animals
Female BALB/c mice syngeneic to 4T1 cells, aged 5–6 weeks, were purchased from Joint Ventures Sipper BK Experimental Animal Co (Shanghai, China) and housed in a room maintained at constant temperature and humidity under 12-h light and darkness cycle and fed with regular autoclave chow diet with water. All animal usage was conducted according to protocols approved by the Zhejiang University Institutional Animal Care and Use Committee.
MTT assay
The effect of piperine on the growth of 4T1 and NIH3T3 cells was examined using a MTT assay. Cells were subcultured in 96-well plates at a density of 103 cells per well with or without piperine (35, 70, 100, 140, and 280 μmol/L) for 24 h, 48 h, or 72 h in a final volume of 200 μL. Then, the medium was removed and 20 μL of MTT (5 mg/mL in PBS) was added to the fresh medium. After 2 h incubation at 37 °C, 100 μL DMSO was added to each well and plates were agitated for 1 min. Spectrophotometric absorbance at 570 nm was measured. The percentage of viability was calculated as the following formula: (viable cells)%=(OD of drug-treated sample/OD of untreated sample)×100.
Apoptosis detection assay
After the treatment with piperine 0, 70, 140, and 280 μmol/L for 24 h, harvested cells were suspended in 100 μL binding buffer (1×) including 1 μL Annexin V-FITC and 10 μL PI for 15 min in dark at room temperature and then 400 μL binding buffer (1×) was added to each sample. The FITC and PI fluorescence were measured through FL-1 filter (530 nm) and FL-2 filter (585 nm) respectively, and 10 000 events were acquired.
Cell cycle analysis
After the treatment with piperine 0, 70, 140, and 280 μmol/L for 24 h, 4T1 cells were collected, washed with cold PBS, fixed in cold 70% ethanol and stored at -20 °C overnight. Cells were then washed again with PBS before staining (100 μg/mL of RNase A, 25 μg/mL of propidium iodide and 0.1% of Triton X-100 in PBS), and incubated at 37 °C for 30 min. Cell cycle analysis was performed on flow cytometer.
Western blot analysis
4T1 cells were lysed in lysis buffer (Cell Signaling Technology, Danvers, MA, USA) for 30 min on ice after the treatment with piperine 0, 70, 140, and 280 μmol/L for 24 h. Lysates were then centrifuged at 14 000×g for 10 min to remove insoluble material. Then a BCA kit (Pierce Biotechnology, Rockford, IL, USA) was used to determine the concentration of lysates. Cell extracts were separated by 10% sodium dodecylsulfate–polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane, blocked with 5% nonfat milk. The blot was probed with each antibody against Bcl-2, Bax, cyclin D1, cyclin B1, caspase 3, and β-actin. Then the blot was washed, exposed to horseradish peroxidase conjugated secondary antibodies for 1 h at room temperature. Signals were detected as we previously described15.
Wound healing assay
To study the effects of piperine on cell migration in vitro, a wound healing assay was performed following standard methods16. 4T1 cells were seeded in 6-well plates and grown until 90% confluent. Cells were then serum starved for 24 h, and a linear wound was created in the confluent monolayer using a 200 μL pipette tip. Cells were then washed with PBS and diluted in RPMI-1640 containing 1% FBS with piperine at a concentration of 70 or 140 μmol/L. Images of the wound surfaces were recorded after 24 h.
RNA isolation and real-time quantitative PCR analysis
Total RNA from cells was extracted using TRIzol reagent in accordance with the manufacturer's instructions. Real-time quantitative PCR, using SYBR Green detection chemistry, was performed on a 7500 Real-Time PCR System (Applied Biosystems). Quantitative measurements were determined using the ΔΔCt method and expression of GAPDH was used as the internal control. The sequences of the primers are as shown in Table 1.
Evaluation of antitumor activity of piperine in 4T1 mammary carcinoma models
4T1 harvested from subconfluent cultures were washed once in serum-free medium and resuspended in PBS. Cells (5×105) in 0.1 mL PBS were implanted subcutaneously into the female BALB/c mice in the abdominal mammary fat pad. After 3 d of implantation, piperines (0, 2.5, and 5 mg/kg) were dissolved in 0.2% DMSO and injected into tumors every three days for three times. The tumor volume was measured every two or three days using the formula V=0.5236×d12×d2, where d1 is the shortest diameter, and d2 is the longest diameter. Tumor tissues for histology examination were collected on d 20, fixed in 10% formalin, and embedded in paraffin. Sections (5 μm thick) were prepared for hematoxylin–eosin staining.
Examination of lung metastases by colonogenic assay
Cells (5×105) in 0.1 mL PBS were implanted subcutaneously into the female BALB/c mice. After 3 d of implantation, piperine (0 and 5 mg/kg) were dissolved in 0.2% DMSO and injected into tumors every three days for three times. Five days after the last therapy, primary 4T1 tumors were eliminated from the experiments. For surgical excision of primary 4T1 tumors, mice were anesthetized, and tumors were resected with sterilized instruments. Wounds were closed with metallic clips. Mice in which primary tumors recurred at the site of the surgical excision were eliminated from the experiments. A colonogenic assay was used to evaluate metastases by 4T1 tumors as we previously described15. Briefly, lungs were collected and chopped before being dissociated in RPMI-1640 supplemented with 10% FBS containing 1.5 mg/mL collagenase type D (Sigma, St Louis, MO, USA) for 30 min in 37 °C shaking incubator at 178 r/min speed. Lungs were then plated at various dilutions in the RPMI-1640 supplemented with 10% FBS and 60 μmol/L 6-thioguanine. Individual colonies representing micrometastases were counted after 5–10 d.
Statistical analysis
All experiments were repeated two or three times. Data was described as the mean±SD, and statistical analysis was carried out using Student's t-test or the log-rank test (for survival analysis). The difference was considered statistically significant when the P-value was less than 0.05.
Results
Piperine inhibited the growth of 4T1 cells in vitro
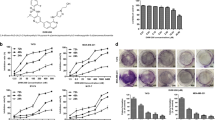
Inhibitory effects of piperine on the growth of 4T1 cells were examined using the MTT assay. We also tested the effects of piperine on murine NIH3T3 fibroblast cell growth. Compared to the untreated cells (taken as 100% viable), different concentrations of piperine showed a time- and dose-dependent inhibition on 4T1 cell growth in the concentration range of 35∼280 μmol/L (Figure 2A). Exposure of 4T1 cells to piperine at 140 μmol/L and 280 μmol/L for 24 h caused a marked decrease in the viability from 100% to 78% and 48% of untreated control levels, respectively. And 48 h of piperine treatment at 140 μmol/L and 280 μmol/L caused a decrease in the viability from 100% to 33% and 18%, respectively. The IC50 values for piperine were 105±1.08 μmol/L for 48 h, and 78.52±1.06 μmol/L for 72 h treatment (Figure 2C), suggesting a dose-dependent inhibition of growth, while piperine was not so toxic to NIH3T3 cells (Figure 2B), for the IC50 values was 232±1.15 μmol/L for 48 h treatment (Figure 2C).
Inhibition of growth by piperine in 4T1 and NIH3T3 cells. 4T1 (A) and NIH3T3 (B) cells were treated with 0, 35, 70, 100, 140, and 280 μmol/L piperine for 24 h, 48 h, and 72 h. Cell viability was monitored by MTT assay. The percentage of viability was calculated as the following formula: (viable cells)%=(OD of drug-treated sample/OD of untreated sample)×100. Mean±SD. n=3. (C) The IC50 values for piperine on 4T1 and NIH3T3 cells.
Piperine induced apoptosis of 4T1 cells and increased the caspase 3 activity
In order to evaluate the mechanism of growth inhibition of 4T1 cells by piperine, apoptosis of 4T1 cells treated with various concentrations of piperine (0, 140, and 280 μmol/L) for 24 h was analyzed by Annexin V-FITC/PI double-labeled flow cytometry. The apoptosis rate was the sum of early apoptosis and late apoptosis. There was little binding of Annexin-V in untreated and 70 μmol/L piperine treated 4T1 cells (Figure 3A). But after treatment with piperine at 140 and 280 μmol/L for 24 h, the apoptosis rate was about 24.2%, 23.6%, respectively (Figure 3B).
Piperine induced the apoptosis in 4T1 cells with the up-regulation of caspase 3. (A) 4T1 cells were treated with piperine (0, 70, 140, and 280 μmol/L) for 24 h. The induction of apoptosis was detected by Annexin V-FITC/PI double staining assay. (B) The apoptotic cell death was quantified as Annexin V+ (both PI-negative and PI-positive) cells. Mean±SD. n=3. bP<0.05, cP<0.01. (C) Western blot analysis of caspase 3, Bcl-2, and Bax in 4T1 cells treated with or without piperine. Similar results were obtained in four independent experiments.
Apoptosis is characterized by changes in the expression and activity of several apoptotic markers. The control and regulation of the apoptotic mitochondrial events occur through the members of the Bcl-2 family of proteins which can be pro-apoptotic (Bcl-10, Bax, etc) or anti-apoptotic (Bcl-2, Bcl-xL, etc). The expression levels of Bax and Bcl-2 are detected by Western blot. The results showed no change in the expression of Bcl-2 and Bax in 4T1 cells after piperine treatment (Figure 3C). Caspases are responsible for cell apoptosis. Therefore we determined the effect of piperine on the expression of caspases by Western blot analysis. The results indicated that caspase 3 activity was up-regulated after piperine treatment (Figure 3C).
Piperine caused the accumulation of the cells in G2/M phase
After incubation with various concentrations of piperine (0, 70, 140, and 280 μmol/L) for 24 h, cell cycle distributions were analyzed by FCM. We found that with the dose of piperine increased, the percentage of cells in G2/M phase was increased, and there were slight changes in G0/G1 phase cell population (Figure 4A). After incubation with 140 and 280 μmol/L piperine for 24 h, percentage of cells in G2/M phase was increased to 7.3%, 15.5%, respectively (Figure 4B).
Piperine caused the accumulation of the cells in the G2/M phase possibly by reducing the expression of cyclin B1. (A) 4T1 cells were treated with piperine 0, 70, 140, 280 μmol/L for 24 h. Then the cells were washed, fixed, stained with propidiumiodide, and analyzed for DNA content by flow cytometry. One representative result was shown. (B) The percentage of cells in different phases of cell cycle after piperine treatment. Mean±SD. n=4. bP<0.05, cP<0.01. (C) Western blot analysis of cyclin D1 and cyclin B1 expression in 4T1 cells treated with or without piperine. One representative result of three independent experiments was shown.
Piperine down-regulated the expression of cyclin B1
Given that cyclins are required for the cell cycle progression, the protein levels of cyclin D1 and cyclin B1 in 4T1 cells were detected by Western blot. The result showed that piperine treatment down-regulated the expression of cyclin B1 in a dose-dependent manner, with maximum suppression observed at 280 μmol/L (Figure 4C), while there was no change of cyclin D1 expression after piperine treatment with various concentrations.
Piperine inhibited the migration ability of 4T1 cells in vitro
We investigated the effects of piperine on the migration properties of 4T1 cells in vitro by wound healing assay. Cells were treated by piperine at a concentration of 0, 70, and 140 μmol/L, and the results showed that piperine inhibited the migration of 4T1 cells in a dose-dependent manner (Figure 5A).
Piperine inhibited 4T1 cell migration in vitro and down-regulated the expression of MMP-9 and 13. (A) Cell migration was evaluated by wound healing assay after 24 h incubation with 0, 70, 140 μmol/L piperine. (B) The mRNA expression of MMP-9, 13 and 14 in 4T1 cells treated by piperine at different concentrations (0, 70, and 140 μmol/L) for 24 h was detected by real-time PCR and normalized to the expression of GAPDH in each sample. Mean±SD. n=4. bP<0.05, cP<0.01.
Piperine decreased the mRNA expression of MMP-9 and 13
Matrix metalloproteinases (MMPs), comprise a family of zinc-containing endopeptidases that share common structural domains. These proteins have the capacity to degrade extracellular matrix (ECM) components, as well as alter their biological functions. Therefore, we examined the mRNA expression of MMP-9, 13, and 14 in 4T1 cells by real-time quantitative PCR after piperine treatment. The mRNA expression of MMP-9 and 13 in 4T1 cells, but not MMP-14, were decreased in a dose-dependent manner (Figure 5B).
Piperine suppressed 4T1 tumor growth in vivo
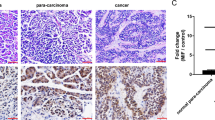
To test the antitumor effect of piperine, female BALB/c mice were inoculated sc with 5×105 4T1 cells. Piperines (0, 2.5, and 5 mg/kg) were injected intratumorally every three days for three times. The result showed that piperine exhibited a significant inhibition of tumor growth especially at a high dose (5 mg/kg) (Figure 6A). To determine whether the regulation of apoptotic proteins and cell cycle-related regulatory proteins was also involved in the inhibition of tumor growth by piperine in vivo, the tumors were harvested from each group and the expression level of these proteins were analyzed by Western blot. As shown in Figure 6B, the level of active caspase 3 was increased and cyclin B1 was reduced in tumors from the mice received piperine therapy, which was similar to the effect of piperine in vitro. Histopathological analysis of the tumor tissues excised from the control mice showed groups of large, round and polygonal cells, with pleomorphic shapes, hyperchromatic nuclei and binucleation. Several degrees of cellular and nuclear pleomorphism were seen. In the tumors excised from the mice treated with piperine, extensive areas of coagulative necrosis were observed (Figure 6C).
Piperine suppressed 4T1 tumor growth in vivo. (A) 5×105 cells in 0.1 mL PBS were implanted subcutaneously into the BALB/c mice. After 3 d of implantation, piperine (0, 2.5, and 5 mg/kg) were dissolved in 0.2% DMSO and injected into tumors every three days for three times. The tumor volume was measured every two or three days using the formula V=0.5236×d12×d2, where d1 is the shortest diameter, and d2 is the longest diameter (n=5). Data are the mean±SD of one representative experiment. Similar results were obtained in at least three independent experiments. (B) Western blot analysis of cleaved caspase 3 and cyclin B1 expression in tumor tissues that were collected on d 20. One representative result of three independent experiments was shown. (C) Hematoxylin-eosin staining of tumor mass after various therapies. Tumor tissues from different groups were collected on d 20, fixed in 10% formalin, and embedded in paraffin. Sections (5 μm thick) were prepared for HE staining.
Piperine also inhibited the lung metastasis of 4T1 tumors
4T1 cells can metastasize to various organs, such as lung, as early as d 10 after inoculation of 1×105 4T1 tumor cells17. To study the potential inhibitory effect of piperine on metastasis, we used a colonogenic assay to evaluate the metastases in the lung. We detected less metastasis in the lungs of the mice that had been given 5 mg/kg piperine treatment compared to the lungs of the control mice (Figure 7). The results indicated that piperine not only suppressed the local primary tumor growth, but also effectively controlled the occurrence of spontaneous metastases.
Piperine inhibited the metastasis of 4T1 tumors in vivo. 4T1 cells were implanted subcutaneously into the female BALB/c mice. After 3 days of implantation, piperine (0 and 5 mg/kg) was dissolved in 0.2% DMSO and intratumorally injected every three days for three times. Primary tumor was surgically removed on d 15 and mice were sacrificed on d 30 post-tumor inoculation for analysis of lung metastases by colonogenic assay. Mean±SD. n=3. bP<0.05.
Discussion
Piperine is a potent anticancer compound that has been demonstrated in various cell types. However, the antitumor activity and action mechanism of piperine on mammary cancer cells still remain unknown. Our results firstly indicated that piperine possessed significant cytoxicity against 4T1 cells both in vitro and in vivo. It was shown piperine induced the apoptosis of 4T1 cells in a dose-dependent manner. We investigated the effect of piperine on the expression levels of the Bcl-2 family, the pro-apoptotic Bax and the anti-apoptotic Bcl-2, which regulate mitochondrial apoptosis. When Bax is overexpressed in cells, apoptotic death in response to death signals is accelerated, while Bcl-2 is overexpressed, it heterodimerized with Bax and the cell death is repressed. Following the exposure to piperine, we found no change of Bax and Bcl-2 at protein level in the 4T1 cells, indicating that the apoptosis induced by piperine is not dependent of the Bcl-2 pathway. We found that the up-regulation of caspase 3 activity may lead to the apoptosis in 4T1 cells. Members of the cyclin family of proteins are key regulators of the cell cycle. Cyclins bind and activate members of the cyclin-dependent kinase (Cdk) family to control cell cycle progression. During the G1 phase, cyclins D1, D2, and D3 form complexes with cdk4 or cdk6, and cyclin E with cdk2, to modulate the expression of proliferative genes. Cyclin A associates with cdk2 during the S phase, and with cdc2 (cdk1) at the S-G2 boundary and into G2. Progression through G2, culminating in mitosis, further requires that cdc2 form complexes with cyclins B1 and B218. We found that piperine treatment down-regulated the expression of cyclin B1 in 4T1 cells with a dose-dependent manner, while there was no change of cyclin D1 expression after piperine treatment. It is consistent with the observation that the percentage of 4T1 cells in G2/M phase was increased after piperine treatment, while no significant change in G0/G1 phase cell population.
The results of the present study provide the potential application of piperine in tumor therapy. Bhat BG et al reported the metabolism of piperine including absorption, tissue distribution and excretion of urinary conjugates in rats19. Upon administration of piperine to male albino rats by gavage or intraperitoneally, about 97% was absorbed irrespective of the mode of dosing. Examination of the passage of piperine through the gut indicated that the highest concentration in the stomach and small intestine was attained at about 6 h19.
The anticancer activity of piperine in vivo was probably partly due to its direct antiproliferative effect on 4T1 cells possibly by increasing the caspase 3 activity and down-regulated the expression of cyclin B1. In this study, we also provided the evidence that piperine can suppress the metastasis of 4T1 tumor in vitro and in vivo. It is well known the process of tumor cell metastasis requires the degradation of ECM molecules in the basement membrane, which is the largest barrier between cancer cells and the bloodstream. The key proteases that are involved in ECM degradation contain MMPs. Our results showed that Piperine significantly reduced the mRNA expression of MMP-9 and MMP-13. It had been reported that suppression of the expression of MMP-9 in tumor cells by piperine is through the inhibition of PKCα and ERK phosphorylation and reduction of NF-κB and AP-1 activation11. MMP-13 was shown to be expressed in more invasive breast carcinoma cells20, and some evidence demonstrated that increased tumor-derived MMP-13 expression independently predicts poor prognoses21. In breast cancer, MMP-9 seems to be expressed in cancer tissue. Malignant breast tumors have increased MMP-9 activity compared to the benign tumors22. Nevertheless, MMP-9 expression has also been described as a positive prognostic marker in node-negative breast cancer23. In another study, positive stromal MMP-9 expression predicts poor survival in the hormone-responsive small tumors, whereas MMP-9 expression in carcinoma cells favors survival24. Thus, MMP-9 expression is associated with both inhibition and stimulation of tumor growth and progression. So, the exact mechanism of the inhibitory effect of piperine on tumor metastasis in vivo needs to be further elucidated. It have been supposed that the inhibition effects of piperine on oxygenase, p450 isoenzyme and cycloxygenase-1 expression may contribute to the antimetastatic qualities25.
According to some authors, the antitumor activity of piperine may be related to its immunomodulatory properties, which involves the activation of cellular and humoral immune responses26, 27. It was proved to be a potent inhibitor of NF-κB, c-Fos, CREB, ATF-2, and proinflammatory cytokine gene expression in B16F-10 melanoma cells10. Other researches documented the chemopreventive efficacy of Piperine is probably due to their antilipidperoxidative and antioxidant potential as well as its modulating effect on the carcinogen detoxification process28, 29. Piperine is also known to enhance the bioavailability of some drugs by inhibiting drug metabolism or by increasing absorption12, 30, 31. Piperine with other antitumor drugs such as 5-FU12, piplartine13 augmented the antitumor activity and also decreased the side effects.
In conclusion, piperine can inhibit tumor growth by inducing cell apoptosis and cell cycle blockage. Also it can suppress 4T1 tumor growth and metastasis in vivo. Further studies will need to be carried out to determine the exact mechanisms of the antitumor action of piperine, and to investigate how it can be used in cancer therapy alone or combination with other antitumor drugs.
Author contribution
Jian-gen SHEN, Qing-qing WANG, and Li-hua LAI designed research; Li-hua LAI, Qi-hong FU, Yang LIU, Kai JIANG, Qing-ming GUO, Qing-yun CHEN, and Bin YAN performed research; Li-hua LAI and Qi-hong FU analyzed data; Li-hua LAI and Qing-qing WANG wrote the paper.
References
Bhardwaj RK, Glaeser H, Becquemont L, Klotz U, Gupta SK, Fromm MF . Piperine, a major constituent of black pepper, inhibits human P-glycoprotein and CYP3A4. J Pharmacol Exp Ther 2002; 302: 645–50.
Volak LP, Ghirmai S, Cashman JR, Court MH . Curcuminoids inhibit multiple human cytochromes P450, UDP-glucuronosyltransferase, and sulfotransferase enzymes, whereas piperine is a relatively selective CYP3A4 inhibitor. Drug Metab Dispos 2008; 36: 1594–605.
Bhutani MK, Bishnoi M, Kulkarni SK . Anti-depressant like effect of curcumin and its combination with piperine in unpredictable chronic stress-induced behavioral, biochemical and neurochemical changes. Pharmacol Biochem Behav 2009; 92: 39–43.
Bang JS, Oh DH, Choi HM, Sur BJ, Lim SJ, Kim JY, et al. Anti-inflammatory and anti-arthritic effects of piperine in human interleukin-1beta-stimulated fibroblast-like synoviocytes and in rat arthritis models. Arthritis Res Ther 2009; 11: R49.
Kumar S, Singhal V, Roshan R, Sharma A, Rembhotkar GW, Ghosh B . Piperine inhibits TNF-alpha induced adhesion of neutrophils to endothelial monolayer through suppression of NF-kappaB and IkappaB kinase activation. Eur J Pharmacol 2007; 575: 177–86.
Wattanathorn J, Chonpathompikunlert P, Muchimapura S, Priprem A, Tankamnerdthai O . Piperine, the potential functional food for mood and cognitive disorders. Food Chem Toxicol 2008; 46: 3106–10.
Taqvi SI, Shah AJ, Gilani AH . Blood pressure lowering and vasomodulator effects of piperine. J Cardiovasc Pharmacol 2008; 52: 452–8.
Pathak N, Khandelwal S . Cytoprotective and immunomodulating properties of piperine on murine splenocytes: an in vitro study. Eur J Pharmacol 2007; 576: 160–70.
Srinivasan K . Black pepper and its pungent principle-piperine: a review of diverse physiological effects. Crit Rev Food Sci Nutr 2007; 47: 735–48.
Pradeep CR, Kuttan G . Effect of piperine on the inhibition of lung metastasis induced B16F-10 melanoma cells in mice. Clin Exp Metastasis 2002; 19: 703–8.
Hwang YP, Yun HJ, Kim HG, Han EH, Choi JH, Chung YC, et al. Suppression of phorbol-12-myristate-13-acetate-induced tumor cell invasion by piperine via the inhibition of PKCalpha/ERK1/2-dependent matrix metalloproteinase-9 expression. Toxicol Lett 2011; 203: 9–19.
Bezerra DP, de Castro FO, Alves AP, Pessoa C, de Moraes MO, Silveira ER, et al. In vitro and in vivo antitumor effect of 5-FU combined with piplartine and piperine. J Appl Toxicol 2008; 28: 156–63.
Bezerra DP, Castro FO, Alves AP, Pessoa C, Moraes MO, Silveira ER, et al. In vivo growth-inhibition of Sarcoma 180 by piplartine and piperine, two alkaloid amides from Piper. Braz J Med Biol Res 2006; 39: 801–7.
Pulaski BA, Ostrand-Rosenberg S . Reduction of established spontaneous mammary carcinoma metastases following immunotherapy with major histocompatibility complex class II and B7.1 cell-based tumor vaccines. Cancer Res 1998; 58: 1486–93.
Hong X, Liu Y, Hu G, Zhao D, Shen J, Shen F, et al. EBAG9 inducing hyporesponsiveness of T cells promotes tumor growth and metastasis in 4T1 murine mammary carcinoma. Cancer Sci 2009; 100: 961–9.
Valster A, Tran NL, Nakada M, Berens ME, Chan AY, Symons M . Cell migration and invasion assays. Methods 2005; 37: 208–15.
Yu P, Lee Y, Wang Y, Liu X, Auh S, Gajewski TF, et al. Targeting the primary tumor to generate CTL for the effective eradication of spontaneous metastases. J Immunol 2007; 179: 1960–8.
Koepp DM, Harper JW, Elledge SJ . How the cyclin became a cyclin: regulated proteolysis in the cell cycle. Cell 1999; 97: 431–4.
Bhat BG, Chandrasekhara N . Studies on the metabolism of piperine: absorption, tissue distribution and excretion of urinary conjugates in rats. Toxicology 1986; 40: 83–92.
Balduyck M, Zerimech F, Gouyer V, Lemaire R, Hemon B, Grard G, et al. Specific expression of matrix metalloproteinases 1, 3, 9 and 13 associated with invasiveness of breast cancer cells in vitro. Clin Exp Metastasis 2000; 18: 171–8.
Zhang B, Cao X, Liu Y, Cao W, Zhang F, Zhang S, et al. Tumor-derived matrix metalloproteinase-13 (MMP-13) correlates with poor prognoses of invasive breast cancer. BMC Cancer 2008; 8: 83.
Hanemaaijer R, Verheijen JH, Maguire TM, Visser H, Toet K, McDermott E, et al. Increased gelatinase-A and gelatinase-B activities in malignant vs benign breast tumors. Int J Cancer 2000; 86: 204–7.
Scorilas A, Karameris A, Arnogiannaki N, Ardavanis A, Bassilopoulos P, Trangas T, et al. Overexpression of matrix-metalloproteinase-9 in human breast cancer: a potential favourable indicator in node-negative patients. Br J Cancer 2001; 84: 1488–96.
Pellikainen JM, Ropponen KM, Kataja VV, Kellokoski JK, Eskelinen MJ, Kosma VM . Expression of matrix metalloproteinase (MMP)-2 and MMP-9 in breast cancer with a special reference to activator protein-2, HER2, and prognosis. Clin Cancer Res 2004; 10: 7621–8.
Stohr JR, Xiao PG, Bauer R . Constituents of Chinese Piper species and their inhibitory activity on prostaglandin and leukotriene biosynthesis in vitro. J Ethnopharmacol 2001; 75: 133–9.
Sunila ES, Kuttan G . Immunomodulatory and antitumor activity of Piper longum Linn and piperine. J Ethnopharmacol 2004; 90: 339–46.
Pathak N, Khandelwal S . Modulation of cadmium induced alterations in murine thymocytes by piperine: oxidative stress, apoptosis, phenotyping and blastogenesis. Biochem Pharmacol 2006; 72: 486–97.
Manoharan S, Balakrishnan S, Menon VP, Alias LM, Reena AR . Chemopreventive efficacy of curcumin and piperine during 7,12-dimethylbenz[a]anthracene-induced hamster buccal pouch carcinogenesis. Singapore Med J 2009; 50: 139–46.
Selvendiran K, Banu SM, Sakthisekaran D . Oral supplementation of piperine leads to altered phase II enzymes and reduced DNA damage and DNA-protein cross links in Benzo(a)pyrene induced experimental lung carcinogenesis. Mol Cell Biochem 2005; 268: 141–7.
Zhao JQ, Du GZ, Xiong YC, Wen YF, Bhadauria M, Nirala SK . Attenuation of beryllium induced hepatorenal dysfunction and oxidative stress in rodents by combined effect of gallic acid and piperine. Arch Pharm Res 2007; 30: 1575–83.
Suresh D, Srinivasan K . Studies on the in vitro absorption of spice principles — curcumin, capsaicin and piperine in rat intestines. Food Chem Toxicol 2007; 45: 1437–42.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (30872377), Education of Zhejiang Province (Y200803762), Zhejiang Provincial Natural Science Foundation (R2100528), Ningbo Municipal Bureau of Science and Technology (2010A610058) and also sponsored by Zhejiang Provincial Program for the Cultivation of High-level Innovative Health Talents.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lai, Lh., Fu, Qh., Liu, Y. et al. Piperine suppresses tumor growth and metastasis in vitro and in vivo in a 4T1 murine breast cancer model. Acta Pharmacol Sin 33, 523–530 (2012). https://doi.org/10.1038/aps.2011.209
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2011.209
Keywords
This article is cited by
-
Characterization and in vitro cytotoxicity of piperine-loaded nanoemulsion in breast cancer cells
Chemical Papers (2024)
-
Piperine improves the sensitivity of osteosarcoma cells to doxorubicin by inducing apoptosis and inhibiting the PI3K/AKT/GSK-3β pathway
Journal of Orthopaedic Surgery and Research (2023)
-
Preparation, Characterization, and Anticancer Efficacy of Chitosan, Chitosan Encapsulated Piperine and Probiotics (Lactobacillus plantarum (MTCC-1407), and Lactobacillus rhamnosus (MTCC-1423) Nanoparticles
BioNanoScience (2022)
-
A Review on the Ecology, Evolution and Conservation of Piper (Piperaceae) in India: Future Directions and Opportunities
The Botanical Review (2022)
-
In vitro synergistic potentials of novel antibacterial combination therapies against Salmonella enterica serovar Typhimurium
BMC Microbiology (2020)