Abstract
Aim:
The hygiene hypothesis suggests a lack of bacterial infections would favor the development of allergic diseases. Mycobacterium bovis bacille Calmette–Guérin (BCG) infection can inhibit allergen-induced asthma reactions, but the underlying mechanism of this infection on the immunological responses is unclear. T-regulatory (Treg) cells are thought to play a role as a crucial immunoregulatory cells that are capable of regulating adaptive immune responses. We conducted this study to investigate whether the protective effect of the BCG vaccination on allergic pulmonary inflammation is associated with the alteration of CD4+CD25+ Treg cells in a murine asthma model and the mechanisms of Treg cells.
Methods:
Newborn C57BL/6 mice were vaccinated 3 times with BCG on d 0, 7, and 14 and subsequently sensitized and challenged with ovalbumin. Eosinophil infiltration was investigated. The frequencies of spleen CD4+CD25+ Treg cells and the expression of specific transcriptional factor Foxp3 were assayed. The cytotoxic lymphocyte associated antigen (CTLA)-4 expression and cytokine interleukin-10 (IL-10) and transforming growth factor-β (TGF-β) levels were measured.
Results:
We showed that treatment of mice with BCG inhibited de novo allergic inflammatory response in a mouse model of asthma. BCG treatments are associated with the increase of CD4+CD25+ Treg cells and Foxp3 expression, accompanied by an increased CTLA-4 expression and cytokine IL-10 and TGF-β levels (P<0.05).
Conclusion:
Neonatal BCG vaccinations ameliorate de novo local eosinophilic inflammation induced by allergen and increase the numbers of CD4+CD25+ Treg cells and Foxp3 expression. The cell–cell contact inhibition and regulatory cytokine production may be involved in the regulatory mechanism.
Similar content being viewed by others
Introduction
Allergic asthma is a chronic inflammatory disease characterized by reversible airway obstruction, airway hyperreactivity, airway infiltration by inflammatory cells — particularly eosinophils and T lymphocytes — and high levels of allergen-specific immunoglobulin (Ig)E1, 2, 3. Although current asthma therapy is effective and well-tolerated in most patients4, these drugs do not change the underlying immunological mechanisms, and there is a clear medical need for the development of new therapies capable of changing or reprogramming the underlying immune processes to produce a long-term protective immune response.
Epidemiological and clinical studies have provided compelling evidence that suggest a link between the relative lack of infectious diseases and the increase in allergic disorders. It has been demonstrated that a positive tuberculin test result suggestive of past infections with mycobacterium tuberculosis was inversely related to the subsequent development of atopy and asthma5. Several animal studies showed that vaccination with mycobacteria prevented asthma development or treated asthmatic manifestations6, 7, 8, 9. However, the mechanism by which mycobacteria inhibits the inflammatory response remains unclear. Induction of Th1 cytokines, such as interferon-γ (IFN-γ), and thus reversion of the imbalance between Th1/Th2, has been proposed7, 8. However, Zuany-Amorim demonstrated that mycobacterium vaccae blocked allergic inflammation by a mechanism independent of IFN-γ9. Our previous study also demonstrated that the neonatal bacille Calmette–Guérin (BCG) vaccination significantly inhibited allergic inflammation, but did not significantly change IFN-γ, interleukin (IL)-4, and IL-5 responses in late-challenged mice10. We thought that mechanisms other than simple changes in the balance between Th1 and Th2 may account for the observed effects of vaccination with mycobacteria.
T regulatory (Treg) cells are thought to be the crucial immunoregulatory cells that are capable of suppressing adaptive immune responses. CD4+CD25+ Treg cells are a distinct population of CD4+ Treg cells that constitutively express CD25, with suppressive properties inhibiting the functions of effector CD4+ and CD8+ T cells11, 12, 13. Foxp3 is a master gene governing the development and function of Treg cells14, 15. The suppressive mechanisms of CD4+CD25+ Treg cells are not clear, but there is evidence that cell–cell contact by cytotoxic lymphocyte associated antigen (CTLA)-4 is required16. There is growing and conflicting evidence concerning the roles of IL-10 and transforming growth factor (TGF)-β17, 18. A number of studies have shown that CD4+CD25+ Treg cells play an essential role in limiting the development of immune response in allergy and asthma19, 20, 21. Therapeutic opportunities to exploit the immunosuppressive effects of Treg cells for prophylactic or therapeutic treatment may become feasible. Treg cells may be involved in the inhibitory effect of BCG upon asthma inflammatory response.
Vaccination with mycobacteria is associated with persistent chronic intracellular infection in vivo that maintains a sustained immune response. The immune system has regulatory mechanisms for suppressing the effector response to persistent infection. It has been demonstrated that tuberculosis (TB) infection and the BCG vaccination induced increased Foxp3 expression and CD4+CD25+ Treg cells in human peripheral blood22, 23, 24.
Given the role of Treg cells in asthma and the effect of mycobacteria infection on Treg cells, we conducted this study to investigate whether the protective effects on allergic pulmonary inflammation of the BCG vaccination are associated with the alteration of CD4+CD25+ Treg cells in a murine asthma model and the mechanisms of Treg cells.
Materials and methods
Animals, immunization, and treatment
Newborn C57BL/6 mice were purchased from the Experimental Animal Center of Zhejiang University (Grade CL, Certificate No 22-010014 conferred by Zhejiang Medical Laboratory Animal Administration Committee; Hangzhou, China). Mice receiving the following treatments were studied: (1) sensitizations and airway challenges with ovalbumin (OVA group); (2) treatment with BCG plus sensitizations and challenges with OVA (BCG-treated group); and (3) the control group. Mice in the BCG-treated group were subcutaneously injected in the back 3 times with 25 μL BCG (D2-BP302 strain; Biological Institute of Shanghai, Shanghai, China; 105CFU) on d 0, 7, and 14; the mice in the OVA and control groups were injected by the same volume of saline solution. The mice in the OVA and BCG-treated groups were sensitized intraperitoneally with 0.1 mL of 0.1% OVA (Sigma–Aldrich, St Louis, MO, USA) plus 0.1 mL aluminum hydroxide on d 42 and 56. On d 66–68, the mice were killed by inhaling OVA (10 mg/mL) aerosols in a Plexiglas exposure chamber for 40 min each day. The mice in the control group were administered saline solution. In each experiment, each group consisted of 12 mice.
Bronchoalveolar lavage fluid and histological analysis
Animals were intraperitoneally anesthetized with 100 mg/kg pentobarbitone sodium 48 h after the last OVA or saline challenge. The trachea was cannulated and bronchoalveolar lavage fluid (BALF) was collected by injecting 0.3 mL phosphate-buffered saline 3 times into the left lung. Total leukocyte numbers were counted under microscopy. Differential cell counts were performed on cytospin slides stained with Wright–Giemsa in a blinded fashion by counting at least 400 cells under immersion oil at ×1000 magnification. The absolute number of eosinophils was calculated. After obtaining the BALF, the left lungs were inflated and fixed with 10% phosphate-buffered formalin, embedded, and sectioned. Tissue sections were stained with hematoxylin-eosin (HE) for identification of inflammation. The sections were examined in a blinded fashion under light microscopy.
Flow cytometry
Spleens from mice were collected and single-cell suspensions were prepared by passing the cells through a cell strainer. Red blood cells (RBC) were removed by hypotonic lysis. The cells were stained for surface markers CD4 [GK1.5, fluorescein isothiocyanate (FITC) labeled] and CD25 [3C7, phycoerythrin(PE)-labeled]. The isotype controls used were FITC-rat IgG2b (A95-1) and PE-rat IgG2b (A95-1). All antibodies were purchased from BD Biosciences (San Jose, CA, USA). The frequency of CD25 was determined by gating on CD4 cells. For each sample, at least 10 000 cells were analyzed. The data were collected and analyzed using CellQuest 3.1f software and a FACSCalibur flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA).
RT-PCR reaction
RNA was extracted from frozen spleen tissue using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). In total, 2 μg total RNA was then reverse transcribed into cDNA using MMLV reverse transcriptase (Promega, Madison, WI, USA) and oligo(dT) as a primer. PCR consisted of 94 °C for 3 min followed by 10 cycles of 94 °C for 10 s, 70 °C for 20 s, 72 °C for 20 s, and then 30 cycles at 94 °C for 10 s, 60 °C for 20 s, and 72 °C for 20 s, terminated by 72 °C for 10 min. All PCR were performed on a Life Express programmable thermal controller (Bioer Technology, Hangzhou, China). As a control, cDNA samples were also amplified with GAPDH-specific primers. The specific primers sequences are as follows: Foxp3: 5′-GAGCCAGAAGAGTTTCTCAAGC-3′ and 5′-GCTACGATGCAGCAAGAGC-3′, CTLA-4: 5′-CTGCTCACTCTTCTTTTCATCC-3′ and 5′-CAATGACATAAATCYGCGTCC-3′, and GAPDH: 5′-AAGGTCGGTGTGAACGGATTT-3′ and 5′-ATGACCTTGCCCACAGCCT-3′. After separation on agarose gels, the specific bands were quantified with Biosens gel imaging system (Shanghai Bio-Tech, Shanghai, China).
Western blot analysis
Frozen spleen tissue was lysed in protein extraction buffer containing protease inhibitors. Lysates were boiled, and equal amounts of proteins were separated on 10% SDS-polyacrylamide gel and transferred to a nitrocellulose membrane. After blocking, the membrane was reacted with a Foxp3 monoclonal antibody (1:500; eBio7979; eBioscience, San Diego, CA, USA) and a GAPDH monoclonal antibody and further incubated with the donkey antimouse IgG horseradish peroxidase secondary antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) and washed. Detection was carried out with enhanced chemiluminescence (Pierce, Rockford, IL, USA). Protein expression was determined by photodensitometry with the Biosens gel imaging system.
Cytokine assays
Splenocytes were prepared as described earlier, plated in 24-well plates (2×106cells/mL) in triplicate, and cultured with medium containing OVA (40 μg/mL) at 37 °C in 5% CO2 for 48 h. The supernatants were then collected and the cytokine levels were determined by ELISA using commercial kits for mouse IL-10 and TGF-β (R&D Systems, Minneapolis, MN, USA). ELISA were performed according to the manufacturer's directions. The limits of detection were 4 pg/mL.
Statistical analysis
For all of the groups, data are presented as mean±SEM. Results were analyzed by one-way ANOVA to identify significant differences between groups. The levels of statistical significance were set at P<0.05, and all statistical calculations were done using SPSS software (version 10.1; SPSS, Chicago, IL, USA).
Results
BCG treatment abolishes eosinophil accumulation in airway and peribronchial/perivascular tissue
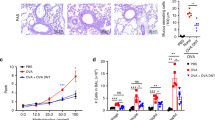
To determine the effects of BCG on the eosinophilic airway inflammation following allergic sensitization and challenge, the cellular content of BALF and the histological examination of tissue stained with HE were assessed. The mice in the OVA group developed a predominance of eosinophils in BALF. The administration of BCG at birth prevented the increase of eosinophil numbers. The control mice had almost no eosinophils in BALF (Figure 1A). Sensitization and subsequent airway challenge resulted in significant increases in numbers of peribronchial and perivascular eosinophils. Treatment with BCG abolished the eosinophil accumulation. Few eosinophils were detected in the control group mice (Figure 1B).
(A) treatment with BCG prevents eosinophil airway infiltration. Cellular content of BALF was assessed. Absolute number of total cells and eosinophils is expressed. Data represent mean±SEM of the number of the different cell types from 36 mice in 3 groups. Eosino, eosinophil; bP<0.05 OVA group vs BCG-treated group or control group. (B) BCG treatment abolishes eosinophil accumulation in the peribronchial-perivascular tissue. Histological examination of lung sections stained with HE was performed. Representative tissue section from a mouse in OVA group (B1), BCG-treated group (B2), and control group (B3) is shown. Final magnification: ×100.
Treatment with BCG increases frequencies of spleen CD4+CD25+ Treg cells and Foxp3 expression
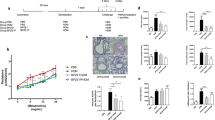
We evaluated the effect of the BCG on the changes of CD4+CD25+ T cells by flow cytometry present in mouse splenocytes (Figure 2A). The mice in the OVA group had a lower percentage of CD25+ T cells within the CD4+ T-cell population in splenocytes compared with the control group (P<0.05). The mice in the BCG-treated group had almost twice the number of CD4+CD25+ T cells in the splenocytes compared with the control mice (P<0.05; Figure 2B).
Increased percentage of spleen Treg cells in BCG-treated mice. (A) representative flow cytometric analysis of CD4 and CD25 on splenocytes of 3 mice from 3 different groups. Numbers represent percentages of events within the CD4+CD25+ T-cell gate in the total CD4+ population. (B) percentage of CD25+ T cells within the CD4+ T-cell population in splenocytes from mice of 3 groups are shown. Results of each group are expressed as mean±SEM (%) of 12 mice. bP<0.05 BCG treated group vs OVA group or control group. eP<0.05 OVA group vs control group.
The transcription factor Foxp3 that is specifically confined to CD4+CD25+ Treg cells25 was used as a definitive marker for Treg cells. The relative level of Foxp3 mRNA was assessed by RT-PCR in mouse spleens. The relative level of Foxp3 mRNA in the mouse spleens was 0.60±0.18 in the BCG-treated group, 0.38±0.11 in the OVA group, and 0.23±0.12 in the control group, respectively. Consistent with the increased percentage of CD4+CD25+ T cells, there was a significant 1.6-fold increase in the Foxp3 mRNA expression in the mouse spleens of the BCG-treated group compared with the OVA group (P=0.000), and a 2.7-fold increase compared with the control group (P=0.000). There was also a significant increase in the OVA group compared with control group (P=0.011; Figure 3A).
Expression of Foxp3 is upregulated in the spleen of BCG-treated mice. (A) relative level of Foxp3 mRNA normalized to GAPDH was assessed by RT-PCR in mouse spleens (12 per group) and is presented as the percentage of the levels. (B) relative level of Foxp3 protein normalized to GAPDH was assessed by Western blot in mouse spleens (12 per group) and presented as the percentage of the levels. Results are expressed as mean±SEM (%) of 12 mice. bP<0.05, cP<0.01 BCG treated group vs OVA group or control group. eP<0.05 OVA group vs control group.
The Foxp3 protein expression was also determined. Parallel with the mRNA level, the Foxp3 protein was also upregulated in the mouse spleens of the BCG-treated group compared with the OVA group (P<0.05) and the control group (P<0.05). There was also a significant increase in the OVA group compared with control group (P<0.05, Figure 3B).
Treatment with BCG increases CTLA-4 expression
The suppressive mechanisms of CD4+CD25+ Treg cells are unclear, but there is evidence that cell–cell contact is required and the inhibitory costimulatory molecule CTLA-4 might be involved15. The expressions of CTLA-4 in the spleen tissue of each group of mice were assessed by RT-PCR. The expression of CTLA-4 mRNA in the BCG-treated group was higher than that in the OVA group and the control group (P<0.05) and there was no significant difference between the OVA group and the control group (Figure 4).
Expressions of CTLA-4 are upregulated in the spleens of BCG-treated mice. Relative level of CTLA-4 mRNA normalized to GAPDH was assessed by RT-PCR in mouse spleens (12 per group) and presented as the percentage of the levels. Results of each group are expressed as mean±SEM (%) of 12 mice. bP<0.05 BCG treated group vs OVA group or control group.
Treatment with BCG alters cytokine production in splenocyte culture
There is conflicting evidence concerning the roles of IL-10 and TGF-β in the suppressive mechanisms of CD4+CD25+ Treg cells16, 17. We therefore examined the levels of regulatory cytokines IL-10 and TGF-β in the cultured splenocyte supernatant. The cultures of splenocytes from BCG-treated mice contained significantly higher levels of IL-10 and TGF-β compared with the mice in the OVA and control groups (P<0.05). There was also an increase of IL-10 in the OVA mice compared with control mice (P<0.05). No significant differences were found in the TGF-β levels between the OVA and control mice (Figure 5).
BCG treatment upregulates the production of IL-10 and TGF-β by OVA-stimulated splenocytes in vitro. Culture cell-free supernatants were harvested and analyzed for cytokine release by ELISA. Results are expressed in pg/mL. Each condition was cultured in triplicate wells in each experiment. Data represent mean±SEM from 12 mice for each group. bP<0.05 BCG-treated groups vs OVA group or control group. eP<0.05 OVA group vs control group.
Discussion
The present study demonstrates that a neonatal subcutaneously injection of BCG can prevent the development of de novo allergen-induced bronchial inflammation in a murine model of allergic pulmonary inflammation. The reduction of the allergic response was associated with an induction of CD4+CD25+ Treg cells and Foxp3 expression. The cell–cell contact inhibition and regulatory cytokine production may be involved in the regulatory mechanism.
In this study, we selected C57BL/6 mice as the animal model. Previous studies reported that BALB/c mice were more prone to a Th2 allergic response than C57BL/6 mice. However, C57BL/6 mice were also selected to make asthma models and used in a series of studies on asthma in our laboratory and those of others10, 26, 27. In this study, the asthma model was proven to be successful based on the eosinophil accumulation in the airway and peribronchial/perivascular tissue pathology.
An inverse relationship between the reduced incidence of infection and increased allergy has been observed in the worldwide over the past 2–3 decades, which has led to the “hygiene hypothesis”, that is, that the existence of microbial infections may prevent or inhibit the development of allergic diseases28, 29. Consistent with previous studies6, 7, 8, 9, this experiment suggests that the neonatal BCG vaccination can inhibit the development of a de novo allergic response characterized by a significant reduction of local eosinophilic inflammation. Also, in the previous study10, we investigated the effects of neonatal BCG treatment on allergic inflammation in mice of different ages and found that BCG treatment could attenuate airway hyperresponsiveness and eosinophilia not only in early-challenged (9 weeks age) mice, but also in late-challenged (45 weeks age) mice. We concluded that the neonatal BCG vaccination had a long-term effect on inhibiting airway allergic inflammation.
However, the mechanism by which BCG inhibits the inflammatory response is debatable. The induction of Th1 cytokines, such as IFN-γ, and thus the reversion of the imbalance between Th1/Th2, has been proposed7, 8. It has been shown that repeated BCG vaccinations improve lung function and increase the IFN-γ/IL-4 ratio in the peripheral blood in asthmatic patients30. Similarly, our previous study demonstrated that the neonatal BCG vaccination induces a strong Th1 type response, which downregulated allergen-specific Th2 responses in an early-challenged asthmatic model. Surprisingly, we found that the neonatal BCG vaccination significantly inhibited allergic inflammation, but did not significantly change IFN-γ, IL-4, and IL-5 responses in late-challenged mice10. Zuany-Amorim demonstrated that mycobacterium vaccae was effective in blocking allergic inflammation by a mechanism independent of IFN-γ9. The mechanisms other than simple changes in the balance between Th1 and Th2 must be at play. A novel finding in this study is that the neonatal BCG vaccination gives rise to CD4+CD25+ Treg cells and Foxp3 expression in the experimental asthma mice. CD4+CD25+ cells represent 5%–10% of periphery CD4+ T cells, expressing CD25, the IL-2 receptor α-chain11. However, because CD25 is also a marker of activation of T cells, its specificity as a marker of Treg is limited. Foxp3 is a master gene governing the development and function of Treg cells14, 15, and Foxp3 expression distinguishes Treg from activated effector cells within the CD25+ T-cell population31. Thus the Foxp3 expression is considered to be the most definite marker of Treg cell activity. We demonstrated that the increases in both the cell surface CD25 expression and Foxp3 expression were the 2 most accurate available markers for the activation of Treg. Therefore, we can conclude that the neonatal BCG vaccination induces the increase of CD4+CD25+ Treg cells in mice.
The 3 BCG vaccinations induced a systemic infection in the mice, and the immune responses were not limited to a regional lymph node. The spleen is the important peripheral lymph organ and abundant in lymphocytes. It plays a key role in the immune response to BCG. The tissue order of the proportion of CD4+CD25+/CD4+ T cells from high to low is spleen, lymph node, peripheral blood, and thymus. The total number of CD4+CD25+ Treg cells in 1 mouse lung is approximately 5×104–7×104 after being intranasally infected with BCG32. The low number of Treg cells can not ensure stable experiment results. Considering all the facts, we selected the spleen as our target tissue.
The BCG vaccination is associated with persistent antigen stimulation and chronic infection in vivo. Many pathogens that cause persistent or chronic infections have evolved strategies to subvert the immune responses of the host. One important immune-subversion strategy is the generation of Treg cells. A number of bacterial, viral, and protozoal infections have been shown to maintain the involvement of CD4+CD25+ Treg cells, such as Leishmania major17, Plasmodium yoeli33, polymicrobial sepsis34, and hepatitis C35. Treg cells are activated by infections. Microbial products can evolve to favor Treg cells' priming, recruitment, and survival. Regarding mycobacteria infection, Guyot-Revol found that in patients with TB, percentages of CD4+CD25 high T cells and levels of Foxp3 mRNA expression in peripheral blood mononuclear cells were both significantly higher compared with controls22. In blood samples from 5675 10-week-old South African infants routinely vaccinated with BCG at birth, the Foxp3 mRNA expression is inducted, which suggests the presence of BCG-induced CD4+ Treg cells23. CD4+CD25 high Foxp3+ Treg cells can be expanded or induced in the peripheral blood of mycobacterium tuberculosis latently infected individuals in conditions known to be predisposed to progression towards active tuberculosis and may therefore play an important role in the pathogenesis of the disease24.
Once activated by the antigen-specific stimulation via the T-cell receptor, CD4+CD25+ cells can suppress the activation and proliferation of other effector CD4+ and CD8+ T cells. The suppression acts in an Ag-non-specific bystander manner36, 37, 38. Allergic asthma is characterized by inflammation mediated by CD4+ Th2 cells. CD4+CD25+ T cells can suppress Th2 maturation39, possibly by inhibiting IL-4 production40. Jaffar demonstrated that CD4+CD25+ T cells limited the development of a CD4+ Th2 phenotype by reduced cytokine production41. There is increasing evidence that Treg play an essential role in limiting the development of immune response in allergy and asthma. It has been shown that CD4+CD25+ Treg cells isolated from hay fever patients have an impaired ability to suppress proliferation and IL-5 production of CD4+CD25– T cells compared with healthy controls19. Lewkowich et al. used an anti-CD25 antibody to deplete CD25+ cells in vivo and showed that the depletion of CD25+ cells before delivery of a house dust mite allergen led to significantly increased Th2 cytokine responses, IgE levels, eosinophilia, and allergen-induced airway hyperresponsiveness (AHR) in allergy-resistant mice20. Using a complementary approach, Kearley et al transferred Treg cells to the immunocompetent mice previously sensitized with OVA and thus inhibited AHR, eosinophil recruitment, and Th2 cytokine production21. Collectively, we speculated that the activated CD4+CD25+ Treg cells by BCG vaccination suppressed the polarized Th2 response in the asthma mice and thus inhibited the allergic inflammatory response.
The mechanism of the suppressive function of Treg cells is still unclear. Suppressive activity has been shown to be mediated through cell–cell contact essentially42 or through secretion of immunosuppressive cytokines equivocally. Cell–cell contact is mediated by CTLA-4 expressed by CD4+CD25+ Treg cells, which interacts with CD80 and/or CD86 on the surface of antigen-presenting cells (APC). This interaction delivers a negative signal for effector T-cell activation16. Read et al found that the blockade of CTLA-4 on CD4+CD25+ Treg cells abrogated their function in vivo43. Our data demonstrate that the CTLA-4 mRNA expression was increased in the spleens of the BCG-treated mice. Although CTLA-4 is not absolutely expressed on Treg, considering the activation of Treg and the essential role of CTLA-4 in the suppressive mechanism, we can reason that the suppressive effects of the activated Treg were at least partially conducted through cell–cell contact mediated by CTLA-4. Transwell experiments are needed to definitely elucidate whether direct cell–cell contact is required and whether CTLA-4 actually works.
In the present study, we demonstrate that cultures of splenocytes from BCG-treated mice contain significantly higher levels of IL-10 and TGF-β compared with that in OVA and control mice. The production of IL-1044 and TGF-β45 have been shown to be required in the suppression of effector T cells by CD4+CD25+ T cells. In allergies, IL-10 and TGF-β skew the production of IgE towards non-inflammatory isotypes IgG4 and IgA, respectively46, 47. However, most studies suggest that CD4+CD25+ Treg cells are not major producers of IL-10 and TGF-β48. Pathogens have been shown to induce the production of either one or both of these cytokines by immature or semimature APC and dendritic cells (DC), and to facilitate the induction of aTreg cells in this way49, 50. CD4+CD25+ Treg cells were also found to aid the generation of aTreg that mediated suppression by these cytokines51, 52. Taken together, we hypothesized that the involvement of the inhibitory cytokines in the suppression effect of CD4+CD25+ Treg cells induced by BCG-treatment was indirect.
We propose in this study that neonatal repeat BCG vaccinations inhibited the de novo allergic inflammatory response in mice via the alteration of the CD4+CD25+ Treg cell pathway. It is interesting to further investigate other immunoregulatory mechanisms that underlie the inhibitory effects of BCG vaccinations. We also examined the contents of IL-5, IL-13, and IL-17E in BALF by ELISA10 and found that higher levels of IL-5 were observed in the BALF of mice in the OVA group while the levels of IL-5 in the BCG group were greatly reduced. Furthermore, low levels of IL-13 and IL-17E were detected from different groups of mice, but were indistinguishable among these groups. These data were consistent with the conclusion and suggested IL-13 and IL-17E may be not involved in the protection of BCG treatment in our experimental models, although they have been shown to be crucial for asthmatic inflammation in other experimental models.
In this study, we propose that neonatal BCG vaccinations ameliorate de novo local eosinophilic inflammation induced by allergens and increase the numbers of CD4+CD25+ Treg cells and Foxp3 expression. The cell–cell contact inhibition and regulatory cytokine production may be involved in the regulatory mechanism.
Author contribution
Hua-hao SHEN designed research; Qian LI performed research; Qian LI contributed new analytical tools and reagents; Hua-hao SHEN analyzed data; Qian LI and Hua-hao SHEN wrote the paper.
References
Moore WC. Update in asthma 2007. Am J Respir Crit Care Med 2008; 177: 1068–73.
Gould HJ, Sutton BJ. IgE in allergy and asthma today. Nat Rev Immunol 2008; 8: 205–17.
Meyer EH, DeKruyff RH, Umetsu DT. T cells and NKT cells in the pathogenesis of asthma. Annu Rev Med 2008; 59: 281–92.
Holgate ST, Polosa R. Treatment strategies for allergy and asthma. Nat Rev Immunol 2008; 8: 218–30.
Shirakawa T, Enomoto T, Shimazu S, Hopkin JM. The inverse association between tuberculin response and atopic disorders. Science 1997; 275: 77–9.
Cui Y, Choi IS, Koh YA, Lin XH, Cho YB, Won YH. Effects of combined BCG and DHEA treatment in preventing the development of asthma. Immunol Invest 2008; 37: 191–202.
Erb KJ, Holloway JW, Sobeck A, Moll H, Gros GL. Infection of mice with Mycobacterium bovis-bacillus Calmette-Guerin (BCG) suppresses allergen-induced airway eosinophilia. J Exp Med 1998; 187: 561–9.
Herz U, Gerhold K, Gruber C, Braun A, Wahn U, Renz H, et al. BCG infection suppresses allergic sensitization and development of increased airway reactivity in an animal model. J Allergy Clin Immunol 1998; 102: 867–74.
Zuany-Amorim C, Manlius C, Trifilieff A, Brunet LR, Rook G, Bowen G, et al. Long-term protective and antigen-specific effect of heat-killed Mycobacterium vaccae in a murine model of allergic pulmonary inflammation. J Immunol 2002; 169: 1492–9.
Shen H, Huang H, Wang J, Ye S, Li W, Wang K, et al. Neonatal vaccination with Bacillus Calmette-Guérin elicits long-term protection in mouse-allergic responses. Allergy 2008; 63: 555–63.
Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 1995; 155: 1151–64.
Murakami M, Sakamoto A, Bender J, Kappler J, Marrack P. CD25+CD4+ T cells contribute to the control of memory CD8+ T cells. Proc Natl Acad Sci USA 2002; 99: 8832–7.
Thornton AM, Shevach EM. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J Exp Med 1998; 188: 287–96.
Ochs HD, Ziegler SF, Torgerson TR. FOXP3 acts as a rheostat of the immune response. Immunol Rev 2005; 203: 156–64.
Brunkow ME, Jeffery EW, Hjerrild KA, Paeper B, Clark LB, Yasayko SA, et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet 2001; 27: 68–73.
Read S, Malmstrom V, Powrie F. Cytotoxic T lymphocyte associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J Exp Med 2000; 192: 295–302.
Belkaid Y, Piccirillo CA, Mendez S, Shevach EM, Sacks DL. CD4+CD25+ regulatory T cells control Leishmania major persistence and immunity. Nature 2002; 420: 502–7.
Nakamura K, Kitani A, Strober W. Cell contact dependent immunosuppression by CD4+CD25+ regulatory T cells is mediated by cell surface-bound transforming growth factor-β. J Exp Med 2001; 194: 629–44.
Ling EM, Smith T, Nguyen XD, Pridgeon C, Dallman M, Arbery J, et al. Relation of CD4+CD25+ regulatory T-cell suppression of allergen-driven T-cell activation to atopic status and expression of allergic disease. Lancet 2004; 363: 608–15.
Lewkowich IP, Herman NS, Schleifer KW, Dance MP, Chen BL, Dienger KM, et al. CD4+CD25+ T cells protect against experimentally-induced asthma by altering the dendritic cell phenotype and activation state. J Exp Med 2005; 202: 1549–61.
Kearley J, Barker JE, Robinson DS, Lloyd CM. Resolution of airway inflammation and hyperreactivity after in vivo transfer of CD4+CD25+ regulatory T cells is interleukin 10 dependent. J Exp Med 2005; 202: 1539–47.
Guyot-Revol V, Innes JA, Hackforth S, Hinks T, Lalvani A. Regulatory T cells are expanded in blood and disease sites in tuberculosis patients. Am J Respir Crit Care Med 2006; 173: 803–10.
Hanekom WA. The immune response to BCG vaccination of newborns. Ann N Y Acad Sci 2005; 1062: 69–78.
Hougardy JM, Verscheure V, Locht C, Mascart F. In vitro expansion of CD4+CD25highFOXP3+CD127low/- regulatory T cells from peripheral blood lymphocytes of healthy Mycobacterium tuberculosis-infected humans. Microbes Infect 2007; 9: 1325–32.
Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science 2003; 299: 1057–61.
Shen HH, Ochkur SI, McGarry MP, Crosby JR, Hines EM, Borchers MT, et al. A causative relationship exists between eosinophils and the development of allergic pulmonary pathologies in the mouse. J Immunol 2003; 170: 3296–305.
Wills-Karp M, Karp CL. Biomedicine. Eosinophils in asthma: remodeling a tangled tale. Science 2004; 305: 1726–9.
Ponsonby AL, Kemp A. Investigation of the hygiene hypothesis: current issues and future directions. Allergy 2008; 63: 506–8.
Holgate ST. The epidemic of allergy and asthma. Nature 1999; 402 (6760 Suppl): B2–4
Choi IS, Koh YI. Effects of BCG revaccination on asthma. Allergy 2003; 58: 1114–6.
Fontenot JD, Rasmussen JP, Williams LM, Dooley JL, Farr AG, Rudensky AY. Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity 2005; 22: 329–41.
Quinn KM, Mchugh RS, Rich FJ, Goldsack LM, de Lisle GW, Buddle BM, et al. Inactivation of CD4+CD25+ regulatory T cells during early mycobacterial infection increases cytokine production but does not affect pathogen load. Immunol Cell Biol 2006; 84: 467–74.
Hisaeda H, Maekawa Y, Iwakawa D, Okada H, Himeno K, Kishihara K, et al. Escape of malaria parasites from host immunity requires CD4+CD25+ regulatory T cells. Nat Med 2004; 10: 29–30.
Heuer JG, Zhang T, Zhao J, Ding C, Cramer M, Justen KL, et al. Adoptive transfer of in vitro stimulated CD4+CD25+ regulatory T cells increases bacterial clearance and improves survival in polymicrobial sepsis. J Immunol 2005; 174: 7141–6.
Boettler T, Spangenberg HC, Neumann-Haefelin C, Panther E, Urbani S, Ferrari C, et al. T cells with a CD4+CD25+ regulatory phenotype suppress in vitro proliferation of virus-specific CD8+ T cells during chronic hepatitis C virus infection. J Virol 2005; 79: 7860–7.
Thornton AM, Shevach EM. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin-2 production. J Exp Med 1998; 188: 287–96.
Kursar M, Bonhagen K, Fensterle J, Kohler A, Hurwitz R, Kamradt T, et al. Regulatory CD4+CD25+ T cells restrict memory CD8+T cell responses. J Exp Med 2002; 196: 1585–92.
Thornton AM, Shevach EM. Suppressor effector function of CD4+CD25+immunoregulatory T cells is antigen nonspecific. J Immunol 2000; 164: 183–90.
Xu D, Liu H, Komai-Koma M, Campbell C, McSharry C, Alexander J, et al. CD4+CD25+ regulatory T cells suppress differentiation and functions of Th1 and Th2 cells, Leishmania major infection, and colitis in mice. J Immunol 2003; 170: 394–9.
Aseffa A, Gumy A, Launois P, MacDonald HR, Louis JA, Tacchini-Cottier F. The early IL-4 response to Leishmania major and the resulting Th2 cell maturation steering progressive disease in BALB/c mice are subject to the control of regulatory CD4+CD25+ T cells. J Immunol 2002; 169: 3232–3.
Jaffar Z, Sivakuru T, Roberts K. CD4+CD25+ T cells regulate airway eosinophilic inflammation by modulating the Th2 cell phenotype. J Immunol 2004; 172: 3842–9.
Birebent B, Lorho R, Lechartier H, de Guibert S, Alizadeh M, Vu N, et al. Suppressive properties of human CD4+CD25+regulatory T cells are dependent on CTLA-4 expression. Eur J Immunol 2004; 34: 3485–96.
Read S, Greenwald R, Izcue A, Robinson N, Mandelbrot D, Francisco L, et al. Blockade of CTLA-4 on CD4+CD25+ regulatory T cells abrogates their function in vivo. J Immunol 2006; 177: 4376–83.
Asseman C, Mauze S, Leach MW, Coffman RL, Powrie F. An essential role for interleukin 10 in the function of regulatory T cells that inhibit intestinal inflammation. J Exp Med 1999; 190: 995–1004.
Horwitz DA, Zheng SG, Wang J, Gray JD. Critical role of IL-2 and TGF-beta in generation, function and stabilization of Foxp3+CD4+ Treg. Eur J Immunol 2008; 38: 912–5.
Akdis CA, Blesken T, Akdis M, Wuthrich B, Blaser K. Role of IL-10 in specific immunotherapy. J Clin Invest 1998; 102: 98–106.
McKarns SC, Letterio JJ, Kaminski NE. Concentration-dependent bifunctional effect of TGF-beta 1 on immunoglobulin production: a role for Smad3 in IgA production in vitro. Int Immunopharmacol 2003; 3: 1761–74.
Maloy KJ, Powrie F. Regulatory T cells in the control of immune pathology. Nat Immunol 2001; 2: 816–22.
Kapsenberg ML. Dendritic-cell control of pathogen-driven T-cell polarization. Nat Rev Immunol 2003; 3: 984–93.
van Kooyk Y, Engering A, Lekkerkerker AN, Ludwig IS, Geijtenbeek TB. Pathogens use carbohydrates to escape immunity induced by dendritic cells. Curr Opin Immunol 2004; 16: 488–93.
Foussat A, Cottrez F, Brun V, Fournier N, Breittmayer JP, Groux H. A comparative study between T regulatory type 1 and CD4+CD25+ T cells in the control of inflammation. J Immunol 2003; 171: 5018–26.
Zheng SG, Wang JH, Gray JD, Soucier H, Horwitz DA. Natural and induced CD4+CD25+ cells educate CD4+CD25+ cells to develop suppressive activity: the role of IL-2, TGF-alpha, and IL-10. J Immunol 2004; 172: 5213–21.
Acknowledgements
Project supported by the key programs of the Ministry of Health (WKJ2005-2-039), the key programs of Natural Science Foundation of Zhejiang Province in China (No 2007C24012) and High-level Innovation Personnel Project of Department of Health of Zhejiang Province, China to Hua-hao SHEN.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Q., Shen, Hh. Neonatal bacillus Calmette–Guérin vaccination inhibits de novo allergic inflammatory response in mice via alteration of CD4+CD25+ T-regulatory cells. Acta Pharmacol Sin 30, 125–133 (2009). https://doi.org/10.1038/aps.2008.3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2008.3
Keywords
This article is cited by
-
Facile and controllable synthesis of amino-modified carbon dots for efficient oil displacement
Nano Research (2023)
-
Ginsenoside compound K suppresses the abnormal activation of T lymphocytes in mice with collagen-induced arthritis
Acta Pharmacologica Sinica (2014)
-
Th2 Responses in OVA-Sensitized BALB/c Mice Are Down-Modulated By Mycobacterium bovis BCG Treatment
Journal of Clinical Immunology (2013)
-
The Role of Regulatory T Cells in Respiratory Infections and Allergy and Asthma
Current Allergy and Asthma Reports (2010)