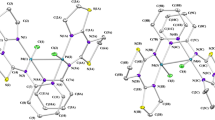
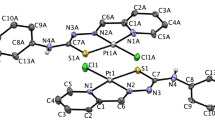
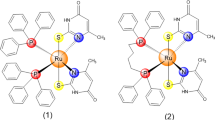
Abstract
Antimony, a natural element that has been used as a drug for over more than 100 years, has remarkable therapeutic efficacy in patients with acute promyelocytic leukemia. This review focuses on recent advances in developing antimony anticancer agents with an emphasis on antimony coordination complexes, Sb (III) and Sb (V). These complexes, which include many organometallic complexes, may provide a broader spectrum of antitumoral activity. They were compared with classical platinum anticancer drugs. The review covers the literature data published up to 2007. A number of antimonials with different antitumoral activities are known and have diverse applications, even though little research has been done on their possibilities. It might be feasible to develop more specific and effective inhibitors for phosphatase-targeted, anticancer therapeutics through the screening of sodium stibogluconate (SSG) and potassium antimonyltartrate-related compounds, which are comprised of antimony conjugated to different organic moieties. For example, SSG appears to be a better inhibitor than suramin which is a compound known for its antineoplastic activity against several types of cancers.
Similar content being viewed by others
Article PDF
References
Desoize B . Metals and metal compounds in cancer treatment. Anticancer Res 2004; 24: 1529–44.
Rosenberg B, Van Camp L, Trosko JE, Mansour VH . Platinum compounds: a new class of potent antitumour agents. Nature 1969; 222: 385–6.
Allen TM . Ligand-targeted therapeutics in anticancer therapy. Nat Rev Cancer 2002; 2: 750–63.
Gielen M, editor. Metal based antitumour drugs. London: Freund Publishing House Ltd; 1988.
Keppler BK, editor. Metal complexes in cancer chemotherapy. Weinheim Germany: VCH; 1993. p 1–8.
Köpf-Maier P . Complexes of metals other than platinum as antitumour agents. Eur J Clin Pharmacol 1994; 47: 1–16.
Barnard CFJ, Fricker SP, Vaughan OJ . Medical applications of inorganic chemicals. In Thompson D, editor. Insights into specialty inorganic chemicals. Cambridge: Royal Society of Chemistry; 1995. p 35–60.
Sadler PJ, Guo Z . Metal complexes in medicine: design and mechanism of action. Pure Appl Chem 1998; 70: 863–71.
Guo Z, Sadler PJ . Metals in medicine. Angew Chem Int Ed Engl 1999; 38: 1512–31.
Meriem A, Willem R, Biesemans M, Mahieu B, Vos de D, Lelievald P, et al. Synthesis, characterization and in vitro antitumour activity of dibutyltin(IV) derivatives of some aromatic carboxylic acids, including aspirin. Appl Organomet Chem 1991; 5: 195–201.
Brutkiewicz RR, Suzuki F . Biological activities and antitumour mechanism of an immunopotentiating organogermanium compound, Ge-132 (review). In Vivo 1987; 1: 189–204.
Celaries B, Gielen M, Vos de D, Rima G . In vitro antitumour activity of some organogermanium radioprotectors. Appl Organomet Chem 2003; 17: 191–3.
Gielen M, Willem R, Bouhdid A, Vos de D, Inventors; Pharmachemie B.V., asignee., Preparation of Aaromatic fluorine-containing organotin compounds as anti-tumour agents. Eur Patent 5 700 921 1996 Mar 13.
Gielen M, Willem R, Biesemans M, Kemmer M, Vos de D, Inventors; Pharmachemie B.V., asignee, Antitumour tin poly oxaalkanecarboxylates, preparation thereof, and compositions containing them. PCT Int Appl 17 006 583 2000 Feb 10.
Tabassum S, Pettinari CJ . Chemical and biotechnological developments in organotin cancerchemotherapy. J Organomet Chem 2006; 691: 1761–6.
Duffin J, Camping BJ . Therapy and disease concepts: the history (and future) of antimony in cancer. J Hist Med 2002; 57: 61–78.
Tiekink ERT . Antimony and bismuth compounds in oncology. Crit Rev Onco Hemat 2002; 42: 217–24.
Tracy JW, Webster LT . Drugs used in the chemotherapy of protozoal infections. In: Hardman JG, Limbird LE, Molinoff PB, Ruddon RW, Gilman AG. editors. The pharmacological basis of therapeutics, 9th ed. New York: Mcgraw-Hill; 1995; p 987–1008.
Berman DJ . Chemotherapy for Leishmaniasis: boichemical mechanisms, clinical efficacy, and future strategies. Rev Infect Dis 1988; 10: 560–86.
Tester-Dalderup CBM . Antiprotozoal drugs. In: Dukes MNG, editor. Meyler′s side effects of drugs, 13th ed. Amsterdam: Elsevier; 1996; p 799–842.
Lee MB, Gilbert HM . Current approaches to Leishmaniasis. Infect Med 1999; 16: 37–45.
Lugo A, Anez N, Petit de Pena Y, Burguera JL, Burguera M . Antimony determination in tissues and blood serum of hamsters infected with Leishmania garnhami and treated with meglumine antimoniate. Annals Trop Med Parasitol 1994; 88: 37–41.
Demicheli C, Frézard F, Lecouvey M, Garnier-Suillerot A . Antimony(V) complex formation with adenine nucleosides in aqueous solution. Biochim Biophys Acta 2002; 1570: 192–8.
Hsu B, Chou CH, Chen JT, Shen ML . Studies on antitumour action of antimony-complexones. Chin Med J 1963; 82: 155–63.
Hsu B, Chou CH, Chen JT, Shen ML . Studies on antitumour action of antimony complexones. Acta Unio Intern Contra Cancrum 1964; 20: 245–8.
Hsu B, Kao YS, Tsai JS, Chou CH, Liu MC, Shen ML, et al. Pharmacological studies of several new antitumour agents. Sci Sin 1964; 13: 789–800.
Jasmin C, Cherman JC, Herve G, Teze A, Souchay P, Boy- Loustau C, et al. In vivo inhibition of murine leukemia and sarcoma viruses by the heteropolyanion 5-tungsto-2-antimoniate J Natl Cancer Inst 1974; 53: 469–74.
Lidereau R, Bouchet C, Sinoussi F, Saracino R, Cherman JC . Current Chemotherapy Proc 1978; 2: 1323–6.
Joesten MD, Najjar R, Hebrank G . Metal complexes of alkylating agents-(III). Complexes by cyclophosphamides. Polyhedron 1982; 1: 637–9.
Hu SZ, Tu LD, Huang YQ, Li ZX . Studies on the antitumour antimony (III) aminopolycarboxylic acid chelates. Crystal structures of M[Sb(pdta)].H2O(M=Na+,NH4+., pdta=propylenediaminetetraacetic acid). Inorg Chim Acta 1995; 232: 161–5.
Hu SZ, Fu YM, Xu B, Tang WD, Yu WJ . Studies on the antitumour antimony (III) triaminocarboxylic complexonates. Crystal structures of NH4[Sb(Hdtpa)]. H2O and Na[Sb(Hdtpa)]. 4.5H2O (dtpa=diethylenetriaminepenta acetic acid). Main Group Metal Chem 1997; 20: 169–80.
Popov AM, Davidovich RL, Li IA, Skulbeda AV, Hu SZ . Cytotoxic and antitumour activity of antimony (III) nitrilotriacetate complexes M2Sb(Nta) (HNta) nH2O (M=NH4, Na., n=1,2). Pharm Chem J 2005; 39: 119–21.
Tofazzal M, Tarafder H, Ali AM, Elias MS, Crouse K, Silong S . Coordination chemistry and biological activity of bidentate and quadridentate nitrogen- sulphur donor ligands and their complexes. Trans Met Chem 2000; 25: 706–10.
Hadjikakou SK, Antoniadis CD, Hadjiliadis N, Kubicki M, Binolis J, Karkaounas S, et al. Synthesis and characterization of new water stable antimony(III) complex with pyrimidine-2-thione and in vitro biological study. Inorg Chim Acta 2005; 358: 2861–6.
Ozturk II, Hadjikakou SK, Hadjiliadis N, Kourkoumelis N, Kubicki M, Baril M, et al. Synthesis, structural characterization, and biological studies of new antimony (III) complexes with thiones. The influence of the solvent on the geometry of the complexes. Inorg Chem 2007; 46: 8652–61.
Xu S, Guo G, Wang J, Huang Y . Effects of potassium antimonyl tartrate on proliferation and apoptosis in vitro of human gastric cancer. Disi Junyi Daxue Xuebao 2004; 25: 1464–6.
Salerno M, Suillerot AG . Resistance to arsenic-and antimony based drugs. Bioinorg Chem Appl 2003; 1: 189–98.
Yu W, Sun B, Chai Y, Zhao Z, Liu X, Ji Z, et al. Apoptosis of acute promyelocytic leukemia cell NB4 induced by different kinds of antimonials. Disi Junyi Daxue Xuebao 2003; 24: 338–41.
Pathak MK, Hu X, Yi T . Effects of sodium stibogluconate on differenciation and proliferation of human myeloid leukemia in vitro. Leukemia 2002; 16: 2285–91.
Muller S, Miller WH, Dejean A . Trivalent antimonials induce degradation of the PML-RARa oncoprotein and reorganization of the promyelocytic leukemia nuclear bodies in acute promyelocytic leukemia NB4 cells. Blood 1998; 92: 4308–16.
Yi T, Pathak MK, Lindner DJ, Ketterer MF, Farver C, Borden EC . Anticancer activity of sodium stibogluconate in synergy with IFNs. J Immunol 2002; 169: 5978–85.
Fan K, Zhou M, Pathak MK, Lidner DJ, Aluntas CJ, Tuohy VK, et al. Sodium Stibogluconate interacts with IL-2 in anti-Renca tumour action via T cell-dependent mechanism in connection with induction of tumour-infiltrating macrophages. J Immunol 2005; 169: 7003–8.
Yu W, Sun B, Chai Y, Zhao Z, Liu X, Ji Z, et al. Apoptotic induction of acute promyelocytic leukemia cell NB4 by antimony trioxide. Xibao Yu Fenzi Mianyixue Zazhi 2002; 18: 592–4.
Yi T . Inhibition of protein tyrosine phosphatase by leishmaniasis agents and their use in combination with cytokines for treatment of cancer and other diseases. PCT Int Appl 2003; 144.
Silvestru C, Socaciu C, Bara A, Haiduc I . The first organoantimony (III) compounds possessing antitumour properties: diphenylantimony(III) derivatives of dithiophosphorus ligands. Anticancer Res 1990; 10: 803–4.
Bara A, Socaciu C, Silvestru C, Haiduc I . Activity of some diphenyltin(IV) and diphenylantimony(III) derivatives on in vitro and in vivo Ehrlich ascites tumour Antitumour organometallics. Anticancer Res 1991; 11: 1651–6.
Socaciu C, Bara A, Silvestru C, Haiduc I . Antitumour organometallics. II. Inhibitory effects of two diphenyl-anti-mony (III) dithiophosphorus derivatives on in vitro and in vivo Ehrlich ascites tumour. In Vivo 1991; 5: 425–8.
Keppler BK, Silvestru C, Haiduc I . Antitumour organometallics. III. In vivo activity of diphenylantimony(III) and diorganotin (IV) dithiophosphorus derivatives against P388 leukemia. Metal-Based Drugs 1994; 1: 73–7.
Socaciu C, Pasca I, Silvestru C, Bara A, Haiduc I . Antitumour organometallics. IV. The mutagenic potential of some diphenylantimony(III) dithiophosphorus derivatives. Metal-Based Drugs 1994; 1: 291–7.
Sharma P, Rosas N, Cabrera A, Toscano A, Silva MJ, Pérez D, et al. First synthesis and structural report on selenophen-2-yl containing pnictogens: Biological Activities of tris (selenophen-2-yl)stibine. J Organomet Chem 2005; 690: 3286–91.
Sharma P, Rosas N, Cabrera A, Toscano A, Silva MJ, Hernández S, et al. Substituted thienyl stibines and bismuthines: syntheses, structures and cytotoxicity. Appl Organomet Chem 2005; 19: 1121–6.
Silvestru C, Haiduc I, Tiekink ERT, de Vos D, Biesemans M, Willem R, et al. Synthesis, structural characterization and in vitro antitumour properties of triorganoantimony(V) disalicylates: crystal and molecular structures of [5-Y-2-(HO)-C6H3COO]2SbMe3(Y=H, Me, MeO). Appl Organomet Chem 1995; 9: 597–607.
Carraher CE Jr, Nass MD, Giron DJ, Cerutis DR . Structrual and biological characterization of antimony(V) polyamines. J Macromol Sci Chem 1983; 19: 1101–20.
Wang GC, Xiao J, Lu Y, Li JS, Cui JR, Wang RQ, et al. Synthesis, crystal structures and in vitro antitumour activities of some arylantimony derivatives of analogues of demethylcantharimide. J Organomet Chem 2004; 689: 1631–8.
Wang GC, Lu YN, Xiao J, Yu L, Song HB, Li JS, et al. Synthesis, Crystal structure and in vitro antitumour activities of some organoantimony arylhydroxamates. J Organomet Chem 2005; 690: 151–6.
Li JS, Ma YQ, Cui JR, Wang RQ . Synthesis and in vitro antitumour activity of some tetraphenylantimony derivatives of exo-7-oxa-bicyclo [2,2,1]heptane(ene)-3- arylamide -2-acid. Appl Organomet Chem 2001; 15: 639–45.
Li JS, Ma YQ, Yu L, Cui JR, Wang RQ . Synthesis, spectroscopic characterization, and in vitro antitumour activity of tetrapheny-lantimony derivatives of analogs of demethylcantharidin and demethyl-dehydrogencantharidin. Synth React Inorg Met Org Chem 2002; 32: 583–93.
Ma YQ, Li JS . Synthesis and in vitro antitumour activity of triarylantomony di (triphenylgermanyl propionates). Main Group Met Chem 2001; 24: 235–8.
Yu L, Ma YQ, Liu RC, Wang GC, Li JS, Du GH, et al. Synthesis, characterization and in vitro antitumour activity of some arylantimony ferrocenylcarboxylate derivatives and the crystal structures of [C5H5FeC5H4C(CH3)=CHCOO]2Sb(C6H4F-4) and (C5H5FeC5H4)C6H4COO]2 Sb(C6H4F-4)3. Polyhedron 2004; 23: 823–9.
Yu L, Ma YQ, Wang GC, Li JS . Synthesis and in vitro antitumour activity of some triarylantimony di(N-phenylglycinates). Heteroatom Chem 2004; 15: 32–6.
Slegmann-Louda DW, Carraher CE Jr, Quinones Q, McBride G . Preliminary evaluation of organoarsenic and organoantimony polymers derived from cephalexin as potential anticancer drugs. PMSE Preprints 2003; 88: 390–2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, P., Perez, D., Cabrera, A. et al. Perspectives of antimony compounds in oncology. Acta Pharmacol Sin 29, 881–890 (2008). https://doi.org/10.1111/j.1745-7254.2008.00818.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00818.x
Keywords
This article is cited by
-
Mechanisms of genotoxicity and proteotoxicity induced by the metalloids arsenic and antimony
Cellular and Molecular Life Sciences (2023)
-
Conjugation of triphenylantimony(V) with carvacrol against human breast cancer cells
JBIC Journal of Biological Inorganic Chemistry (2022)
-
Effect of cobalt(II) chloride hexahydrate on some human cancer cell lines
SpringerPlus (2016)
-
DFT-based prediction of antifungal and insecticidal activities of perfluorophenyl antimony(III) and antimony(V) chlorides
Medicinal Chemistry Research (2014)
-
Small molecule inhibitors of DNA repair nuclease activities of APE1
Cellular and Molecular Life Sciences (2010)