Abstract
Aim:
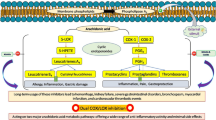
To design and synthesize a series of benzenesulfonamide derivatives, 4-[2-alkylthio-5(4)-(4-substitutedphenyl)imidazole-4(5)-yl]benzenesulfonamides(4a-4j), which are intended to act as cyclooxygenase-2 (COX-2) inhibitors with good COX-2 inhibitor activity, and which will exert anti-inflammatory activities in vivo.
Methods:
Benzenesulfonamide derivatives were designed and synthesized through multi-step chemical reactions. All the synthesized compounds were evaluated in an in vitro assay. The active compound 4a-4f was selected for further evaluation in a carrageenan-induced rat paw edema model.
Results:
Docking studies showed that compound 4 bind into the primary binding site of COX-2 with the sulfonamide SO2NH2 moiety interacting with the secondary pocket amino acid residues. In the in vitro assay, compound 4 inhibited COX-2 with an inhibition concentration IC50 value of 1.23-8 nmol/L, compared to celecoxib with IC50 value of 1.5 nmol/L. Compound 4b and 4c had good potency and selectivity in comparison to the celecoxib. In the in vivo model, compound 4a-4f exhibited a moderate potency to inhibit 50% carrageenan-induced paw edema with value of 1.58-4.3 mg/kg. In the latter experiment, compound 4c was the most active compound.
Conclusion:
The anti-inflammatory effects obtained for compound 4a–4j could be due to the presence of fluorine or hydrogen substituents in the para position of the phenyl ring of these compounds.
Similar content being viewed by others
Article PDF
References
Hansch C, Sammes PG, Taylor JB . The rational design, mechanistic study and therapeutic application of chemical compounds. Comprehensive medicinal chemistry; v 6. Oxford: Pergamon Press; 1990.
Leblanc Y, Black WC, Chan CC, Charleson S, Delorme D, Denis D, et al. Synthesis and biological evaluation of both enantiomers of L-761,000 as inhibitors of cyclooxygenase. Bioorg Med Chem Lett 1996; 6: 731–6.
Kalgutkar AS . Selective cyclooxygenase-2 inhibitors as non-ulcerogenic anti-inflammatory agents. Exp Opin Ther Pat 1999; 9: 831–49.
Reitz DB, Isakson PC . Cyclooxygenase-2 inhibitors. Curr Pharm Des 1995; 1: 211–20.
Penning T, Talley J, Bertenshaw S, Carter J, Collins P, Docter S, et al. Synthesis and biological evaluation of 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors: Identification of 4-[5-(4-methylphenyl)-3 -(trifluoromethyl)- 1H-pyrazole-1-yl] benzene-sulfonamide (SC-58635, Celecoxib). J Med Chem 1997; 40: 1347–65.
Prasit P, Wang Z, Brideau C, Chan CC, Charleson S, Cromlish W, et al. The discovery of rofecoxib, [MK 966, Vioxx (R), 4-(4′-methylsulfonylphenyl)-3-phenyl-2(5H)-furanone], an orally active cyclooxygenase-2 inhibitor. Bioorg Med Chem Lett 1999; 9: 1773–8.
Talley JJ, Brown DL, Carter JS, Graneto MJ, Koboldt CM, Masferrer JL, et al. 4-[5-methyl-3-phenylisoxazol-4-yl]-ben-zenesulfonamide, valdecoxib: a potent and selective inhibitor of COX-2. J Med Chem 2000; 43: 775–7.
Riendeau D, Percival MD, Brideau C, Charleson S, Dube D, Ethier D, et al. Preclinical profile and comparison with other agents that selectively inhibit cyclooxygenase-2. J Pharmacol Exp Ther 2001; 296: 558–66.
Arico S, Pattingre S, Baury C, Gane P, Barbat A, Codogno P, et al. Celecoxib induces apoptosis by inhibiting 3-phosphoinositide dependent protein-kinase-1 activity in the human colon cancer. J Biol Chem 2002; 277: 27613–21.
Davies G, Martin LA, Sacks N, Dowsett M . Cyclooxygenase-2 (COX-2), aromatase and breast cancer: a possible role for COX-2 inhibitors in breast cancer chemoprevention. Ann Oncol 2002; 13: 669–78.
Liu HX, Kirschenbaum A, Yao S, Lee R, Holland FJ, Levine CAJ . Inhibition of cyclooxygenase-2 suppresses angiogenesis and the growth of prostate cancer in vivo. J Urol 2000; 164: 820–5.
Sawaoka H, Kawano S, Tsuji S, Tsuji M, Gunawan ES, Takei Y, et al. Cyclooxygenase-2 inhibitors suppress the growth of gastric cancer xenografts via induction of apoptosis in nude mice. Am J Physiol 1998; 274: G1061–7.
Khanna IK, Weier RM, Yu Y, Xu XD, Koszyk FJ, Collins PW, et al. 1,2-Diarylimidazoles as potent, cycloxygenase-2 selective, and orally active anti-inflammatory agents. J Med Chem 1997; 40: 1634–47.
Wright JM . The double-edged sword of COX-2 selective NSAIDs. CMAJ 2002; 167: 1131–7.
Kontogiorgis CA, Hadjipavlou-Litina DJ . Synthesis and anti-inflammatory activity of coumarin derivatives. J Med Chem 2005; 48: 6400–8.
Niedballa U, Bottcher I . Antiinflammatory 4,5-diphenyl-2-substituted-thio-imidazoles and their corresponding sulfoxides and sulfones. US patent 4 440 776. Apr 03 1984.
Navidpour L, Shafaroodi H, Abdi KH, Amini M, Ghahremani MH, Dehpour AR, et al. Design, synthesis, and biological evaluation of substituted 3-alkylthio-4,5-diaryl-4H-1,2,4-triazoles as selective COX-2 inhibitors. Biorg Med Chem 2006; 14: 2507–17.
Navidpour L, Amini M, Shafaroodi H, Abdi KH, Ghahremani MH, Dehpour AR, et al. Design and synthesis of new water-soluble tetrazolide derivatives of celecoxib and rofecoxib as selective cyclooxygenase-2 (COX-2) inhibitors. Biorg Med Chem Lett 2006; 16: 4483–7.
Navidpour L, Karimi L, Amini M, Vosooghi M, Shafiee A . Syntheses of 5-alkylthio-1,3-diaryl-1,2,4-triazoles. J Heterocyclic Chem 2004; 41: 201–4.
Karimi L, Navidpour L, Amini M, Shafiee A . Synthesis of 4,5-Diaryl-1,2,3-thiadiazoles. Phosphorus Sulfur and Silicon and the Related Elements 2005; 180: 1593–600.
Johari Daha F, Matloubi H, Tabatabai SA, Shafiee B, Shafiee A . Synthesis of 1-(4-methylsulfonylphenyl)-5-aryl-1,2,3-triazoles and 1-(4-aminosulfonylphenyl)-5-aryl-1,2,3-triazoles. J Heterocyclic Chem 2005; 42: 33–7.
Salimi M, Amini M, Shafiee A . Syntheses of 2-alkylthio-(4,5-diaryl) imidazoles. Phosphorus Sulfur Silicon 2005; 180: 1587–92.
Goodsell DS, Olson AJ . Automated docking of substrates to proteins by simulate annealing. Proteins: structure function and genetics 1990; 8: 195–202.
Morris GM, Goodsell DS, Huey R, Olson AJ . Distributed automated docking of flexible ligands to proteins: Parallel applications of autodock 2.4. J Computre-aided Mol Design 1996; 10: 293–304.
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, et al. Automated docking using Lamarckian genetic algorithm and an empirical binding free energy function. J Comp Chem 1998; 19: 1639–62.
Kulmacz RJ, Lands WEM . Requirements for hydroperoxide by the cyclooxygenase and peroxidase activities of prostaglandin H synthase. Prostaglandins 1983; 25: 531–40.
Winter CA, Rislet EA, Nuss GW . Carrageenan-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Proc Soc Exp Biol Med 1962; 111: 544–7.
Ahmadiani A, Fereidoni M, Semnanian S, Kamalinejad M, Saremi S . Antinociceptive and anti-inflammatory effects of sambucus ebulus rhysome extracts in rats. J Ethnopharmacol 1998; 61: 229–35.
Luong F, Miller A, Barnett J, Chow J, Ramesha C, Browner MF . Flexibility of the NSAIDs binding site in the structure of human cyclooxygenase-2. Nat Struct Biol 1996; 3: 927–33.
Gierse JK, McDonald JJ, Hauser SD, Rangwala SH, Koboldt CM, Seibert K . A single amino acid difference between cyclooxygenase-1 (COX-1) and 2- (COX-2) reverse the selectivity of COX-2 specific inhibitors. J Biol Chem 1996; 271: 15810–4.
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegman RA, Pak JY, et al. Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 1996; 384: 644–8.
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from the research council of Tehran University of Medical Sciences and Iran Chapter of TWAS (The Developing World of Academy of Sciences), and INSF (Iran National Science Foundation).
Rights and permissions
About this article
Cite this article
Salimi, M., Ghahremani, M., Naderi, N. et al. Design, synthesis and pharmacological evaluation of 4-[2-alkylthio-5(4)-(4-substitutedphenyl)imidazole-4(5)yl]benzenesulfonamides as selective COX-2 inhibitors. Acta Pharmacol Sin 28, 1254–1260 (2007). https://doi.org/10.1111/j.1745-7254.2007.00619.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00619.x
Keywords
This article is cited by
-
Design and synthesis of new 1,2-diaryl-4,5,6,7-tetrahydro-1H-benzo[d] imidazoles as selective cyclooxygenase (COX-2) inhibitors
Medicinal Chemistry Research (2012)