Abstract
Aim:
To investigate the expression of sonic hedgehog (SHH) and epidermal growth factor receptor (EGFR) signal molecules in pancreatic cancer cells, and to assess the inhibitory effects through the blockade of the SHH and EGFR signaling pathways by cyclopamine and Iressa, respectively.
Methods:
The expression of SHH and EGFR in pancreatic cancer cell lines (PANC-1, SUIT-2, and ASPC-1) was detected by RT-PCR and Western blot analysis. After treatment with different concentrations of cyclopamine, alone or in combination with Iressa, the antiproliferative effect on pancreatic cancer cells was analyzed by methyl thiazolyl tetrazolium assays. A flow cytometry analysis was used to detect the cellular cycle distribution and apoptosis of pancreatic cancer cells.
Results:
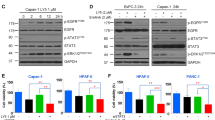
All of the 3 pancreatic cancer cell lines expressed SHH, Smoothened (SMO), and EGFR. Cyclopamine could downregulate the expression of EGFR in all cell lines. Cyclopamine or Iressa could induce a growth inhibitory effect in a dose-dependent manner. Moreover, the combined use of 2.5 μmol/L cyclopamine and 1 μmol/L Iressa induced an enhanced inhibitory effect and a greater apoptosis rate than any agent alone. The percentage of the cell population of the G0/G1 and sub-G1 phases was significantly increased along with the increasing dose of cyclopamine and/or Iressa.
Conclusion:
The blockade of the sonic hedgehog signal pathway enhances the antiproliferative effect of the EGFR inhibitor through the downregulation of its expression in pancreatic cancer cells. The simultaneous blockade of SHH and EGFR signaling represents possible targets of new treatment strategies for pancreatic carcinoma.
Similar content being viewed by others
Article PDF
References
Moon HJ, An JY, Heo JS, Choi SH, Joh JW, Kim YI . Predicting survival after surgical resection for pancreatic ductal adenocarcinoma. Pancreas 2006; 32: 37–43.
Thayer SP, Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 2003; 425: 851–6.
Hebrok M, Kim SK, St Jacques B, McMahon AP, Melton DA . Regulation of pancreas development by hedgehog signaling. Development 2000; 127: 4905–13.
Fu JR, Liu WL, Zhou JF, Sun HY, Xu HZ, Luo L, et al. Sonic hedgehog protein promotes bone marrow-derived endothelial progenitor cell proliferation, migration and VEGF production via PI 3–kinase/Akt signaling pathways. Acta Pharmacol Sin 2006; 27: 685–93.
Berman DM, Karhadkar SS, Maitra A, Montes De Oca R, Gerstenblith MR, Briggs K, et al. Widespread requirement for hedgehog ligand stimulation in growth of digestive tract tumours. Nature 2003; 425: 846–51.
Wang Z, Sengupta R, Banerjee S, Li Y, Zhang Y, Rahman KMW, et al. Epidermal growth factor receptor-related protein inhibits cell growth and invasion in pancreatic cancer. Cancer Res 2006; 66: 7653–60.
Sipos B, Moser S, Kalthoff H, Torok V, Lohr M, Kloppel G . A comprehensive characterization of pancreatic ductal carcinoma cell lines: towards the establishment of an in vitro research platform. Virchows Arch 2003; 442: 444–52.
Carpenter G, Cohen S . Epidermal growth factor. J Biol Chem 1990; 265: 7709–12.
Salomon DS, Brandt R, Ciardiello F, Normanno N . Epidermal growth factor-related peptides and their receptors in human malignancies. Crit Rev Oncol Hematol 1995; 19: 183–232.
Mimeault M, Moore E, Moniaux N, Hénichart JP, Depreux P, Lin MF, et al. Cytotoxic effects induced by a combination of cyclopamine and gefitinib, the selective hedgehog and epidermal growth factor receptor signaling inhibitors, in prostate cancer cells. Int J Cancer 2006; 118: 1022–31.
Sui G, Bonde P, Dhara S, Broor A, Wang J, Marti G, et al. Epidermal growth factor receptor and hedgehog signaling pathways are active in esophageal cancer cells from rat reflux model. J Surg Res 2006; 134: 1–9.
Kayed H, Kleeff J, Osman T, Keleg S, Buchler MW, Friess H . Hedgehog signaling in the normal and diseased pancreas. Pancreas 2006; 32: 119–29.
Amin A, Li Y, Finkelstein R . Hedgehog activates the EGF receptor pathway during Drosophila head development. Development 1999; 126: 2623–30.
Dahmane N, Sanchez P, Gitton Y, Palma V, Sun T, Beyna M, et al. The Sonic Hedgehog-Gli pathway regulates dorsal brain growth and tumorigenesis. Development 2001; 128: 5201–12.
Altaba A, Sanchez P, Dahmane N . Gli and hedgehog in cancer: tumours, embryos and stem cells. Nat Rev Cancer 2002; 2: 361–72.
Wessells RJ, Grumbling G, Donaldson T, Wang SY, Simcox A . Tissue-specific regulation of vein/EGF receptor signaling in Drosophila. Dev Biol 1999; 216: 243–59.
Altaba A, Stecca B, Sanchez P . Hedgehog-Gli signaling in brain tumors: stem cells and paradevelopmental programs in cancer. Cancer Lett 2004; 204: 145–57.
Incardona JP, Gaffield W, Kapur RP, Roelink H . The teratogenic Veratrum alkaloid cyclopamine inhibits sonic hedgehog signal transduction. Development 1998; 125: 3553–62.
Berman DM, Karhadkar SS, Hallahan AR, Pritchard JI, Eberhart CG, Watkins DN, et al. Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 2002; 297: 1559–61.
Kubo M, Nakamura M, Tasaki A, Yamanaka N, Nakashima H, Nomura M, et al. Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res 2004; 64: 6071–4.
Sanchez P, Hernandez AM, Stecca B, Kahler AJ, DeGueme AM, Barrett A, et al. Inhibition of prostate cancer proliferation by interference with sonic hedgehog-gli1 signaling. Proc Natl Acad Sci USA 2004; 101: 12561–6.
Karhadkar SS, Steven Bova G, Abdallah N, Dhara S, Gardner D, Maitra A, et al. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 2004; 431: 707–12.
Kayed H, Kleeff J, Keleg S, Guo J, Ketterer K, Berberat PO, et al. Indian hedgehog signaling pathway: expression and regulation in pancreatic cancer. Int J Cancer 2004; 110: 668–76.
Ciardiello F, Caputo R, Bianco R, Damiano V, Pomatico G, De Placido S, et al. Antitumor effect and potentiation of cytotoxic drugs activity in human cancer cells by ZD-1839 (Iressa), an epidermal growth factor receptor-selective tyrosine kinase inhibitor. Clin Cancer Res 2000; 6: 2053–63.
Kenney AM, Rowitch DH . Sonic hedgehog promotes G(1) cyclin expression and sustained cell cycle progression in mammalian neuronal precursors. Mol Cell Biol 2000; 20: 9055–67.
Qualtrough D, Buda A, Gaffield W, Williams AC, Paraskeva C . Hedgehog signalling in colorectal tumour cells: induction of apoptosis with cyclopamine treatment. Int J Cancer 2004; 110: 831–7.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30571817).
Rights and permissions
About this article
Cite this article
Hu, Wg., Liu, T., Xiong, Jx. et al. Blockade of sonic hedgehog signal pathway enhances antiproliferative effect of EGFR inhibitor in pancreatic cancer cells. Acta Pharmacol Sin 28, 1224–1230 (2007). https://doi.org/10.1111/j.1745-7254.2007.00620.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00620.x
Keywords
This article is cited by
-
Hedgehog ligand and receptor cooperatively regulate EGFR stability and activity in non-small cell lung cancer
Cellular Oncology (2024)
-
Upregulation of LINC01426 promotes the progression and stemness in lung adenocarcinoma by enhancing the level of SHH protein to activate the hedgehog pathway
Cell Death & Disease (2021)
-
Pancreatic cancer stem cells: new understanding of tumorigenesis, clinical implications
Langenbeck's Archives of Surgery (2010)
-
Adjuvante und palliative Therapie des Pankreaskarzinoms
Der Onkologe (2010)
-
Pancreatic cancer: molecular pathogenesis and new therapeutic targets
Nature Reviews Gastroenterology & Hepatology (2009)