Abstract
Aim:
To investigate the effects of lithium (Li) and prostaglandin A1 (PGA1) on the expression of heat shock factor 1 (HSF-1), heat shock proteins (HSP), and apoptosis protease activating factor-1 (Apaf-1) induced by permanent focal ischemia in rats.
Methods:
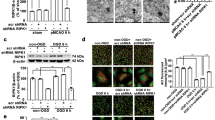
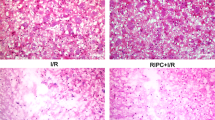
The rats were pretreated with a subcutaneous (sc) injection of Li for 2 d or a single intracerebral ventricle (icv) administration of PGA1 for 15 min before ischemic insult, or a combination of Li (sc, 1 mEq/kg, 2 d) and PGA1 (icv, 15 min prior to ischemic insult). Brain ischemia was induced by the permanent middle cerebral artery occlusion (pMCAO). Twenty-four hours after the occlusion, the expression of HSF-1, HSP, andApaf-1 in the ischemic striatum were examined with Western blot analysis.
Results:
The expression of HSF-1, heme oxygenase-1 (HO-1), HSP90α, and Apaf-1 were significantly increased, but the expression of HSP90β was significantly decreased 24 h after the pMCAO. PGA1 and Li and their combination significantly enhanced the ischemia-induced elevation in the levels of HSF-1, HO-1, and HSP90α, and recovered HSP90β expression, but decreased Apaf-1 levels in the ischemic striatum.
Conclusion:
The present study demonstrates that PGA1 and Li have synergistic effects on the enhancement of the expression of HSP, suggesting that the synergistic effects of PGA1 and Li in the rat model of permanent focal cerebral ischemia may be mediated by the enhancement expression of HSP expression and the downregulation of Apaf-1. Our studies suggest that combined PGA1 and Li may have potential clinical value for the treatment of stroke.
Similar content being viewed by others
Article PDF
References
Stoll G, Jander S, Schroeter SM . Inflammation and glial responses in ischemic brain lesions. Prog Neurobiol 1998; 56: 149–71.
White BC, Sullivan JM, DeGracia DJ, O'Neil BJ, Neumar RW, Grossman LI, et al. Brain ischemia and reperfusion: molecular mechanisms of neuronal injury. J Neurol Sci 2000; 179: 1–33.
Qin ZH, Chen RW, Wang Y, Nakai M, Chuang DM, Chase TN . Nuclear factor kappaB nuclear translocation upregulates c-Myc and p53 expression during NMDA receptor-mediated apoptosis in rat striatum. J Neurosci 1999; 19: 4023–33.
Wang X, Qin ZH, Leng Y, Wang Y, Jin X, Chase TN, et al. Prostaglandin A1 inhibits rotenone-induced apoptosis in SH-SY5Y cells. J Neurochem 2002; 83: 1094–102.
Chen RW, Qin ZH, Ren M, Kanai H, Chalecka-Franaszek E, Leeds P, et al. Regulation of c-Jun N-terminal kinase, p38 kinase and AP-1 DNA binding in cultured brain neurons: roles in glutamate excitotoxicity and lithium neuroprotection. J Neurochem 2003; 84: 566–75.
Nonaka S, Chuang DM . Neuroprotective effects of chronic lithium on focal cerebral ischemia in rats. Neuroreport 1998; 9: 2081–4.
Wei H, Qin ZH, Senatorov VV, Wei W, Wang Y, Qian Y, et al. Lithium supresses excitotoxicity-induced striatal lesions in a rat model of Huntington's disease. Neuroscience 2001; 106: 603–12.
Ren M, Senatotov VV, Chen RW, Chuang DM . Postinsult treatment with lithium reduces brain damage and facilitates neurological recovery in a rat ischemia/reperfusion model. Proc Natl Acad Sci USA 2003; 100: 6210–5.
Xu J, Culman J, Blume A, Brecht S, Gohlke P . Chronic treatment with a low dose of lithium protects the brain against ischemic injury by reducing apoptotic death. Stroke 2003; 34: 1287–92.
Zhang HL, Huang ZH, Zhu Y, Liang ZQ, Han R, Wang X, et al. Neuroprotective effects of prostaglandin A1 in animal models of focal ischemia. Brain Res 2005; 1039: 203–6.
Xu XH, Zhang HL, Han R, Gu ZL, Qin ZH . Enhancement of neuroprotection and heat shock protein induction by combined prostaglandin A1 and lithium in rodent models of focal ischemia. Brain Res 2006; 1102: 154–62.
Hendrick JP, Hartl FU . Molecular chaperone functions of heat-shock proteins. Annu Rev Biochem 1993; 62: 349–84.
Fu R, Zhao ZQ, Zhao HY, Zhao JS, Zhu XL . Expression of heme oxygenase-1 protein and messenger RNA in permanent cerebral ischemia in rats. Neurological Res 2006; 28: 38–45.
Fredduzzi S, Mariucci G, Tantucci M, Ambrosini MV . Generalized induction of 72-kDa heat-shock protein after transient focal ischemia in rat brain. Exp Brain Res 2001; 136: 19–24.
Izaki K, Kinouchi H, Watanabe K, Owada Y, Okubo A, Itoh H, et al. Induction of mitochondrial heat shock protein 60 and 10 mRNAs following transient focal cerebral ischemia in the rat. Brain Res Mol Brain Res 2001; 88: 14–25.
Majda BT, Meloni BP, Rixon N, Knuckey NW . Suppression subtraction hybridization and northern analysis reveal upregula-tion of heat shock, trkB, and sodium calcium exchanger genes following global cerebral ischemia in the rat. Brain Res Mol Brain Res 2001; 93: 173–9.
Takaki E, Fujimoto M, Sugahara K, Nakahari T, Yonemura S, Tanaka Y, et al. Maintenance of olfactory neurogenesis requires HSF-1, a major heat shock transcription factor in mice. J Biol Chem 2006; 281: 4931–7.
Csermely P, Schnaider T, Soti C, Prohaszka Z, Nardai G . The 90-kDa molecular chaperone family: Structure, function, and clinical applications. A comprehensive review. Pharmacol Ther 1998; 79: 129–68.
Minami Y, Kawasaki H, Miyata Y, Suzuki K, Yahara I . Analysis of native forms and isoform compositions of the mouse 90–kDa heat shock protein, HSP90. J Biol Chem 1991; 266: 10099–103.
Pandey P, Saleh A, Nakazawa A, Kumar S, Srinivasula SM, Kumar V, et al. Negative regulation of cytochrome c-mediated oligo-merization of Apaf-1 and activation of procaspase-9 by heat shock protein 90. EMBO J 2000; 19: 4310–22.
Longa EZ, Weinstein PR, Carlson S, Cummins R . Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989; 20: 84–91.
Dwyer BE, Nishimura RN, Lu SY, Alcaraz A . Transient induction of heme oxygenase after cortical stab wound injury. Mol Brain Res 1996; 38: 251–9.
Siesjo BK, Katsura K, Kristian T . The biological basis of cerebral ischemic damage. J Neurosurg Anesthesiol 1995; 7: 47–52.
Takeda A, Kimpara T, Onodera H, Itoyama Y, Shibahara S, Kogure K . Regional difference in induction of heme oxygenase-1 protein following rat transient forebrain ischemia. Neurosci Lett 1996; 205: 169–72.
Hara ED, Takahashi K, Tominaga T, Kumabe T, Kayama T, Suzuki H, et al. Expression of heme oxygenase and inducible nitric oxide synthase mRNA in human brain tumours. Biochem Biophys Res Commun 1996; 224: 153–8.
Premkumar DR, Smith MA, Richey PL, Petersen RB, Castellani R, Kutty RK, et al. Induction of heme oxygenase-1 mRNA and protein in neocortex and cerebral vessels in Alzheimer's disease. J Neurochem 1995; 65: 1399–402.
Geddes JW, Pettigrew LC, Holtz ML, Craddock SD, Maines MD . Permanent focal and transient global cerebral ischemia increase glial and neuronal expression of heme oxygenase-1, but not heme oxygenase-2, protein in rat brain. Neurosci Lett 1996; 210: 205–8.
Shih AY, Li P, Murphy TH . A small-molecule-inducible Nrf2-mediated antioxidant response provides effective prophylaxis against cerebral ischemia in vivo. J Neurosci 2005; 25: 10321–35.
Elbirt KK, Bonkovsky HL . Heme oxygenase: recent advances in understanding its regulation and role. Proc Assoc Am Physicians 1999; 111: 438–47.
Suttner DM, Dennery PA . Reversal of HO-1 related cytoprotection with increased expression is due to reactive iron. FASEB J 1999; 13: 1800–9.
Abraham NG, Lavrosvky Y, Schwartzman ML, Stoltz RA, Levere RD, Gerritsen ME, et al. Transfection of the human hemeoxygenase gene into rabbit coronary microvessel endothelial cells: protective effect against heme and hemoglobin toxicity. Proc Natl Acad Sci USA 1995; 92: 6798–802.
Maines MD . Theheme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol 1997; 37: 517–54.
Otterbein LE, Kolls JK, Mantell LL, Cook JL, Alam J, Choi AMK . Exogenous administration of heme oxygenase-1 by gene transfer provides protection against hyperoxia-induced lung injury. J Clin Invest 1999; 103: 1047–54.
Jakob U, Buchner J . Assisting spontaneity: the role of Hsp90 and small Hsps as molecular chaperones. Trends Biochem Sci 1994; 19: 205–11.
Parsell DA, Lindquist S . The function of heat-shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu Rev Genet 1993; 27: 437–96.
Ali A, Krone PH, Pearson DS, Heikkila JJ . Evaluation of stress-inducible hsp90 gene expression as potential molecular biomarker in Xenopus laevis. Cell Stress Chaperones 1996; 1: 62–9.
Yang SH, Liu R, Wen Y, Perez E, Cutright J, Brun-Zinkernagel AM, et al. Neuroendocrine mechanism for tolerance to cerebral ischemia-reperfusion injury in male rats. J Neurobiol 2005; 62: 341–51.
Lewis J, Devin A, Miller A, Lin Y, Rodriguez Y, Neckers L, et al. Disruption of hsp90 function results in degradation of the death domain kinase, receptor-interacting protein (RIP), and blockage of tumor necrosis factor-induced nuclear factor-kappaB activation. J Biol Chem 2000; 275: 10519–26.
Sato S, Fujita N, Tsuruo T . Modulation of Akt kinase activity by binding to Hsp90. Proc Natl Acad Sci USA 2000; 97: 10832–7.
Sun N, Shi J, Chen L, Liu X, Guan X . Influence of electro-acupuncture on the mRNA of heat shock protein 70 and 90 in brain after cerebral ischemia/reperfusion of rats. J Huazhong Univ Sci Tech Med Sci 2003; 23: 112–5.
Bergeron M, Mivechi NF, Giaccia AJ, Giffard RG . Mechanism of heat shock protein 72 induction in primary cultured astrocytes after oxygen-glucose deprivation. Neurol Res 1996; 18: 64–72.
Choi DW . Cerebral hypoxia: Some new approaches and unanswered questions. J Neurosci 1990; 10: 2493–501.
Sciandra JJ, Subjeck JR, Hughes CS . Induction of glucose-regulated proteins during anacrobic exposure and of heat-shock proteins after reoxygenation. Proc Natl Acad Sci USA 1984; 81: 4843–7.
Mestril R, Chi SH, Sayen MR, Dillmann WH . Isolation of a novel inducible rat heat-shock protein (HSP70) gene and its expression during ischemia/hypoxia and heat shock. Biochem J 1994; 298: 561–9.
Mosser DD, Kotzbauer PT, Sarge KD, Morimoto RI . In vitro activation of heat shock transcription factor DNA-binding by calcium and biochemical conditions that affect protein conformation. Proc Natl Acad Sci USA 1990; 87: 3748–52.
Amici C, Sistonen L, Santoro MG, Morinoto RI . Antiproliferative prostaglandins activate heat shock transcription factor. Proc Natl Acad Sci USA 1992; 89: 6277–81.
Jurivich DA, Sistonen L, Sarge KD, Morinoto RI . Arachidonate is a potent modulator of human heat shock gene transcription. Proc Natl Acad Sci USA 1994; 91: 2280–4.
Higashi T, Nakai A, Uemura Y, Kikuchi H, Nagata K . Activation of heat shock factor 1 in rat brain during cerebral ischemia or after heat shock. Brain Res Mol Brain Res 1995; 34: 262–70.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from the Department of Education of Jiangsu Province (No 03KJB310123), the Natural Science Foundation of Jiangsu Province (No BK2004037), and the National Natural Science Foundation of China (No 30470587).
Rights and permissions
About this article
Cite this article
Xu, Xh., Hua, Yn., Zhang, Hl. et al. Greater stress protein expression enhanced by combined prostaglandin A1 and lithium in a rat model of focal ischemia. Acta Pharmacol Sin 28, 1097–1104 (2007). https://doi.org/10.1111/j.1745-7254.2007.00624.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00624.x
Keywords
This article is cited by
-
PGA2-induced HO-1 attenuates G2M arrest by modulating GADD45α expression
Molecular & Cellular Toxicology (2015)
-
GSK-3β inhibition protects mesothelial cells during experimental peritoneal dialysis through upregulation of the heat shock response
Cell Stress and Chaperones (2013)