Abstract
Aim:
To determine whether all-trans retinoic acid (atRA) acts to modulate angiotensin II (Ang II)-induced cardiac fibroblast cell growth and collagen secretion.
Methods:
Cultured neonatal rat cardiac fibroblasts (CF) were used in the experiment. A 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay was used to detect cell growth of the CF; and immunocytochemistry and Western blotting were used to measure the production and secretion of collagen and the expression of transforming growth factor-β1 (TGF-β1) by the CF.
Results:
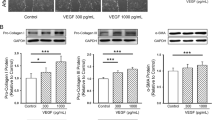
atRA (1 × 10−7 to 1 × 10−5 mol/L) inhibited the Ang II-induced increase in cell growth of CF (P<0.05). Ang II stimulated the secretion of collagen types I and III by the CF. This effect was blocked by AT1 receptor antagonist losartan (1 × 10−6 mol/L), but not by AT2 receptor antagonist PD123319 (up to 1 × 10−6 mol/L). Exposure of CF to atRA (1 × 10−5 mol/L) attenuated the Ang II-induced increase in the secretion of collagen types I and III (P<0.05). atRA (1 × 10−5 mol/L) also blocked the Ang II-induced increase in the expression of TGF-β1
Conclusion:
atRA inhibits the Ang II-induced increase in cell growth and collagen secretion of neonatal rat CF. The effect of atRA is possibly mediated by lowering the TGF-β1 level. These observations support the notion that atRA is a potential candidate for the prevention and therapy of cardiac remodeling.
Similar content being viewed by others
Article PDF
References
Weber KT, Anversa P, Armstrong PW . Remodeling and reparation of the cardiovascular system. J Am Coll Cardiol 1992; 20: 3–16.
Cooper G . Basic determinants of myocardial hypertrophy: a review of molecular mechanisms. Annu Rev Med 1997; 48: 13–23.
Schurch W, Seemayer TA, Gabbiani G . Myofibroblast. In: Sternberg SS, editor. Histology for pathologists. New York: Raven Press; 1992. p 109–4.
Weber KT, Brilla CG . Pathological hypertrophy and cardiac interstitium. Fibrosis and renin-angiotensin-aldosterone system. Circulation 1991; 83: 1849–65.
Fujisaki H, Ito H, Hirata Y . Natriuretic peptides inhibit angio-tensin II-induced proliferation of rat cardiac fibroblasts by blocking endothelin-1 gene expression. J Clin Invest 1995; 96: 1059–65.
Gray MO, Long CS, Kalinyak JE, Li HT, Karliner JS . Angio-tensin II stimulates cardiac myocyte hypertrophy via paracrine release of TGF-beta 1 and endothelin-1 from fibroblasts. Cardiovasc Res 1998; 40: 352–63.
Sano M, Fukuda K, Sato T . ERK and p38 MAPK, but not NF-kB, are critically involved in reactive oxygen species-mediated induction of IL-6 by angiotensin II in cardiac fibroblasts. Circ Res 2001; 89: 661–9.
Dorfman DM, Wilson DB, Bruns GA, Orkin SH . Human transcription factor GATA-2. Evidence for regulation of preproendothelin-1 gene expression in endothelial cells. J Biol Chem 1992; 267: 1279–85.
Griendling KK, Murphy TJ, Alexander RW . Molecular biology of the renin-angiotensin system. Circulation 1993; 87: 1816–28.
Osterrieder W, Muller RK, Powell JS, Clozel JP, Hefti F, Baumgartner HR . Role of angiotensin II in injury-induced neointima formation in rats. Hypertension 1991; 18 Suppl II: II60–II64.
Miano J, Topouzis S, Majesky M, Olson E . Retinoid receptor expression and all-trans retinoic acid-mediated growth inhibition in vascular smooth muscle cells. Circulation 1996; 93: 1886–95.
Datta PK, Lianos EA . Retinoic acids inhibit inducible nitric oxide synthase expression in mesangial cells. Kidney Int 1999; 56: 486–93.
Neuville P, Yan Z, Gidlof A, Pepper MS, Hansson GK, Gabbiani G, et al. Retinoic acid regulates arterial smooth muscle cell proliferation and phenotypic features in vivo and in vitro through an RARa-dependent signaling pathway. Arterioscler Thromb Vasc Biol 1999; 19: 1430–6.
Simonson M . Anti-AP-1 activity of all-trans retinoic acid in glomerular mesangial cells. Am J Physiol 1994; 267: F805–F815.
Wang HJ, Zhu YC, Yao T . Effects of all-trans retinoic acid on angiotensin II-induced myocyte hypertrophy. J Appl Physiol 2002; 92: 2162–8.
Lü L, Yao T, Zhu YZ, Huang GY, Cao YX, Zhu YC . Chronic all-trans retinoic acid treatment prevents medial thickening of intramyocardial and intrarenal arteries in spontaneously hypertensive rats. Am J Physiol Heart Circ Physiol 2003; 285: H1370–H1377.
Mercola M, Wang CY, Kelly J, Brownlee C, Jackson-Grusby L, Stiles C, et al. Selective expression of PDGF A and its receptor during early mouse embryogenesis. Dev Biol 1990; 138: 114–22.
Jakowlew SB, Cubert J, Danielpour D, Sporn MB, Roberts AB . Differential regulation of the expression of transforming growth factor-beta mRNAs by growth factors and retinoic acid in chicken embryo chondrocytes, myocytes, and fibroblasts. J Cell Physiol 1992; 150: 377–85.
Glick AB, McCune BK, Abdulkarem N, Flanders KC, Lumadue A, Smith JM, et al. Complex regulation of TGF-β expression by retinoic acid in the vitamin A-deficient rat. Development 1991; 111: 1081–6.
Haxsen V, Adam-Stitah S, Ritz E, Wagner J . Retinoids inhibit the actions of angiotensin II on vascular smooth muscle cells. Circ Res 2001; 88: 637–45.
Jalil JE, Ebensperger R, Melendez J, Acevedo E, Sapag-Hagar M, Gonzalez-Jara F, et al. Effects of antihypertensive treatment on cardiac IGF-1 during prevention of ventricular hypertrophy in the rat. Life Sci 1999; 64: 1603–12.
Pfeffer MA, Pfeffer JM, Froehlich ED . Pumping ability of the hypertrophying left ventricle of the spontaneously hyperten sive rat. Circ Res 1976; 38: 423–9.
Lijnen PJ, Petrov VV . Role of intracardiac renin-angiotensin-aldosterone system in extracellular matrix remodeling. Methods Find Exp Clin Pharmacol 2003; 25: 541–64.
Strickland S, Mahdavi V . The induction of differentiation in terato carcinoma stem cells by retinoic acid. Cell 1978; 15: 393–403.
Van Giezen JJ, Boon GI, Jansen JW, Bouma BN . Retinoic acid enhances fibrinolytic activity in vivo by enhancing tissue type plasminogen activator (t-PA) activity and inhibits venous thrombosis. Thromb Haemost 1993; 69: 381–6.
Fanjul A, Dawson MI, Hobbs PD, Jong L, Cameron JF, Harlev E, et al. Anew class of retinoids with selective inhibition of AP-1 inhibits proliferation. Nature 1994; 372: 107–10.
James TW, Wagner R, White LA, Zwolak RM, Brinckerhoff CE . Induction of collagenase and stromelysin gene expression by mechanical injury in a vascular smooth muscle-derived cell line. J Cell Physiol 1993; 157: 426–37.
Lijnen PJ, Petrov VV, Fagard RH . Induction of cardiac fibrosis by transforming growth factor-β. Mol Gen Metab 2000; 71: 418–35.
Booz GW, Baker KM . Molecular signaling mechanisms controlling growth and function of cardiac fibroblasts. Cardiovasc Res 1995; 30: 537–43.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 39970241).
Rights and permissions
About this article
Cite this article
He, Y., Huang, Y., Zhou, L. et al. All-trans retinoic acid inhibited angiotensin II-induced increase in cell growth and collagen secretion of neonatal cardiac fibroblasts. Acta Pharmacol Sin 27, 423–429 (2006). https://doi.org/10.1111/j.1745-7254.2006.00296.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00296.x