Abstract
Aim:
To study the growth inhibitory and apoptotic effects of adenosine triphosphate (ATP) and adenosine (ADO) on human gastric carcinoma (HGC)-27 cells in vitro and the mechanisms related to the actions of ATP and ADO.
Methods:
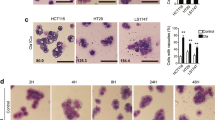
MTT assay was used to determine the reduction of cell viability. The morphological changes of HGC-27 cells induced by ATP or ADO were observed under fluorescence light microscope by acridine orange/ethidium bromide double-stained cells. The internucleosomal fragmentation of genomic DNA was detected by agarose gel electrophoresis. The apoptotic rate and cell-cycle analysis after treatment with ATP or ADO was determined by flow cytometry.
Results:
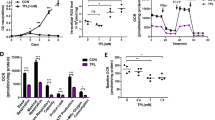
ATP, ADO and the intermediate metabolites, ADP and AMP, and the agonist of purinergic receptors, reduced cell viability of HGC-27 cells at doses of 0.3 and 1.0 mmol·L−1. The distribution of cell cycle phase and proliferation index (PI) value of HGC-27 cells changed when exposed to ATP or ADO at the concentrations of 0.1, 0.3 and 1 mmol/L for 48 h. ATP and ADO both altered the distribution of cell cycle phase via G0/G1-phase arrest and significantly decreased PI value. Under light microscope, the tumor cells exposed to 0.3 mmol·L−1 ATP or ADO displayed morphological changes of apoptosis; a ladder-like pattern of DNA fragmentation obtained from HGC-27 cells treated with 0.1–1 mmol·L−1 ATP or ADO appeared in agarose gel electrophoresis; ATP and ADO induced the apoptosis of HGC-27 cells in a dose-dependent manner at concentrations between 0.03–1 mmol–L−1. The maximum apoptotic rate of HGC-27 cells exposed to ATP or ADO for 48 h was 13.53% or 15.9%, respectively. HGC-27 cell death induced by ATP or ADO was significantly inhibited by dipyridamole (10 mmol·L−1), an inhibitor of adenosine transporter, but was not affected by aminophylline, a broad inhibitor of P1 receptors and pyridoxal-phosphate-6-azophenyl-2, 4-disulphonic acid tetrasodium salt (30 μmol·L−1), a non-selective antagonist of P2 receptors.
Conclusion:
Extracellular ATP and ADO reduced the cell viability, arrested cell cycle and induced apoptosis in HGC-27 cell line by intracellular uptake of ADO. One of the main routes of ATP-induced apoptosis in HGC-27 cells is through the breakdown to adenosine.
Similar content being viewed by others
Article PDF
References
Nihei OK, de Carvalho AC, Savino W, Alves LA . Pharmacologic properties of P2Z/P2X7 receptor characterized in murine dendritic cells: role on the induction of apoptosis. Blood 2000; 96: 996–1005.
Schneider C, Wiendl H, Ogilvie A . Biphasic cytotoxic mechanism of extracellular ATP on U-937 human histiocytic leukemia cells: involvement of adenosine generation. Biochim Biophys Acta 2001; 1538: 190–205.
Schrier SM, Van Tilburg EW, van der Meulen H, Ijzerman AP, Mulder GJ, Nagelkerke JF . Extracellular adenosine-induced apoptosis in mouse neuroblastoma cells: studies on involvement of adenosine receptors and adenosine uptake. Biochem Pharmacol 2001; 61: 417–25.
Schrier SM, Florea BI, Mulder GJ, Nagelkerke JF, Ijzerman AP . Apoptosis induced by extracellular ATP in the mouse neuroblastoma cell line N1E-115: studies on involvement of P2 receptors and adenosine. Biochem Pharmacol 2002; 63: 1119–26.
Yamada T, Okajima F, Akbar M, Tomura H, Narita T, Yamada T, et al. Cell cycle arrest and the induction of apoptosis in pancreatic cancer cells exposed to adenosine triphosphate in vitro. Oncol Rep 2002; 9: 113–7.
Hopfner M, Maaser K, Barthel B, von Lampe B, Hanski C, Riecken EO, et al. Growth inhibition and apoptosis induced by P2Y2 receptors in human colorectal carcinoma cells: involvement of intracellular calcium and cyclic adenosine monophosphate. Int J Colorectal Dis 2001; 16: 154–66.
Janssens R, Boeynaems JM . Effects of extracellular nucleotides and nucleosides on prostate carcinoma cells. Br J Pharmacol 2001; 132: 536–46.
Sun AY, Chen YM . Extracellular ATP-induced apoptosis in PC 12 cells. Adv Exp Med Biol 1998; 446: 73–83.
Bronte V, Macino B, Zambon A, Rosato A, Mandruzzato S, Zanovello P, et al. Protein tyrosine kinases and phosphatases control apoptosis induced by extracellular adenosine 5′-triphosphate. Biochem Biophys Res Commun 1996; 218: 344–51.
Ferre S, Fredholm BB, Morelli M, Popoli P, Fuxe K . Adeno-sinedopamine receptor-receptor interactions as an integrative mechanism in the basal ganglia. Trends Neurosci 1997; 20: 482–7.
Khoa ND, Montesinos MC, Reiss AB, Delano D, Awadallah N, Cronstein BN . Inflammatory cytokines regulate function and expression of adenosine A2A receptors in human monocytic THP-1 cells. J Immunol 2001; 167: 4026–32.
Trincavelli ML, Falleni A, Chelli B, Tuscano D, Costa B, Gremigni V, et al. A2A adenosine receptor ligands and proinflammatory cytokines induce PC 12 cell death through apoptosis. Biochem Pharmacol 2003; 66: 1953–62.
Bohm SK, Grady EF, Bunnett NW . Regulatory mechanisms that modulate signalling by G-protein-coupled receptors. Biochem J 1997; 322 ( Pt 1): 1–18.
Wakade TD, Palmer KC, McCauley R, Przywara DA, Wakade AR . Adenosine-induced apoptosis in chick embryonic sympathetic neurons: a new physiological role for adenosine. J Physiol 1995; 488: 123–38.
Tanaka Y, Yoshihara K, Tsuyuki M, Kamiya T . Apoptosis induced by adenosine in human leukemia HL-60 cells. Exp Cell Res 1994; 213: 242–52.
Kohno Y, Sei Y, Koshiba M, Kim HO, Jacobson KA . Induction of apoptosis in HL-60 human promyelocytic leukemia cells by adenosine A3 receptor agonists. Biochem Biophys Res Commun 1996; 219: 904–10.
Coutinho-Silva R, Perfettini JL, Persechini PM, Dautry-Varsat A, Ojcius DM . Modulation of P2Z/P2X7 receptor activity in macrophages infected with Chlamydia psittaci. Am J Physiol Cell Physiol 2001; 280: C81–9.
Maaser K, Hopfner M, Kap H, Sutter AP, Barthel B, von Lampe B, et al. Extracellular nucleotides inhibit growth of human oesophageal cancer cells via P2Y2-receptors. Br J Cancer 2002; 86: 636–44.
Fujita N, Kakimi M, Ikeda Y, Hiramoto T, Suzuki K . Extracellular ATP inhibits starvation-induced apoptosis via P2X2 receptors in differentiated rat pheochromocytoma PC 12 cells. Life Sci 2000; 66: 1849–59.
Burnstock G . Purinergic signaling and vascular cell proliferation and death. Arterioscler Thromb Vasc Biol 2002; 22: 364–73.
Barry CP, Lind SE . Adenosine-mediated killing of cultured epithelial cancer cells. Cancer Res 2000; 60: 1887–94.
Wang MX, Ren LM, Shan BE . Effects of extracellular adenosine triphosphate and adenosine on tumor cell proliferation. Chin Pharmacol Bull 2003; 19: 1242–6.
Situ ZQ, Wu JZ . Cell Culture. Xi-an: World Book's Publishing Company; 1996. 186–8.
Wang CY, Sheng RL, Wang F, Ding XJ, Qiu NL . Fluorescence method of apoptotic morphology studying by acridine orange and ethidium bromide double stained cells. Chin J Pathophysiol 1998; 14: 104–6.
Kim KT, Yeo EJ, Choi H, Park SC . The effect of pyrimidine nucleosides on adenosine-induced apoptosis in HL-60 cells. J Cancer Res Clin Oncol 1998; 124: 471–7.
Fredholm BB, Abbracchio MP, Burnstock G, Daly JW, Harden TK, Jacobson KA, et al. Nomenclature and classification of purinoceptors. Pharmacol Rev 1994; 46: 143–56.
Saitoh M, Nagai K, Nakagawa K, Yamamura T, Yamamoto S, Nishizaki T . Adenosine induces apoptosis in the human gastric cancer cells via an intrinsic pathway relevant to activation of AMP-activated protein kinase. Biochem Pharmacol 2004; 67: 2005–11.
Lu GZ, Lin ZX, Zhang ZQ, Zhou LX, Xu GW . Studies on mechanism about ATP inhibited the proliferation of MGC-803 cells. Shi Yan Sheng Wu Xue Bao 1994; 27: 137–9.
Abbracchio MP, Ceruti S, Brambilla R, Franceschi C, Malorni W, Jacobson KA, et al. Modulation of apoptosis by adenosine in the central nervous system: a possible role for the A3 receptor. Patho-physiological significance and therapeutic implications for neurodegenerative disorders. Ann N Y Acad Sci 1997; 825: 11–22.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Science and Technology Development Project of Hebei Province (No 20042761317).
Rights and permissions
About this article
Cite this article
Wang, Mx., Ren, Lm. Growth inhibitory effect and apoptosis induced by extracellular ATP and adenosine on human gastric carcinoma cells: involvement of intracellular uptake of adenosine. Acta Pharmacol Sin 27, 1085–1092 (2006). https://doi.org/10.1111/j.1745-7254.2006.00342.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00342.x
Keywords
This article is cited by
-
Autocrine and paracrine purinergic signaling in the most lethal types of cancer
Purinergic Signalling (2021)
-
Grifolic acid induces GH3 adenoma cell death by inhibiting ATP production through a GPR120-independent mechanism
BMC Pharmacology and Toxicology (2018)
-
Purinergic signalling in the gastrointestinal tract and related organs in health and disease
Purinergic Signalling (2014)
-
Purinergic signalling and cancer
Purinergic Signalling (2013)
-
Extracellular ATP and Cancer—An Overview with Special Reference to P2 Purinergic Receptors
Pathology & Oncology Research (2008)