Abstract
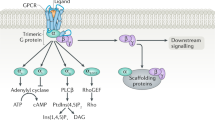
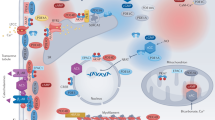
RhoA and Rho-kinase (ROCK) participate in a wide variety of cell signal functions such as cell growth, smooth and cardiac muscle contraction, cytoskeleton rearrangement, cell migration and proliferation. In vascular smooth muscle cells, RhoA and ROCK play an important role in Ca2+ sensitization and regulate vascular smooth muscle tone. In the heart, RhoA and ROCK mediate hypertrophic response leading to cardiac hypertrophy. Recent cellular and molecular biology studies using ROCK inhibitors such as Y-27632 and fasudil have indicated a pivotal role of the RhoA-ROCK cascade in many aspects of cardiovascular function such as cardiac hypertrophy and ventricular remodeling following myocardial infarction. Inhibition of the RhoA-ROCK signaling pathway may be a suitable target for a number of cardiovascular diseases including hypertension, atherosclerosis, diabetes and hypertrophic heart failure. This review focuses on the current understanding of the RhoA-ROCK signal pathway in heart diseases and discusses the use of ROCK inhibitors as therapeutic agents for heart diseases ranging from hypertensive cardiomyopathy to heart failure.
Similar content being viewed by others
Article PDF
References
Amano M, Chihara K, Kimura K, Fukata Y, Nakamura N, Matsuura Y, et al. Formation of actin stress fibers and focal adhesions enhanced by Rho-kinase. Science 1997; 275: 1308–11.
Yasui Y, Amano M, Nagata K, Inagaki N, Nakamura H, Saya H, et al. Roles of Rho-associated kinase in cytokinesis: mutations to Rho-associated kinase phosphorylation sites impair cytokinetic segregation of glial filaments. J Cell Biol 1998; 143: 1249–58.
Hoshijima M, Sah VP, Wang Y, Chien KR, Brown JH . The low molecular weight GTPase Rho regulates myofibril formation and organization in neonatal rat ventricular myocytes: involvement of Rho kinase. J Biol Chem 1998; 273: 7725–30.
Majumdar M, Seasholtz TM, Goldstein D, de Lanerolle P, Brown JH . Requirement for Rho-mediated myosin light chain phosphorylation in thrombin-stimulated cell rounding and its dissociation from mitogenesis. J Biol Chem 1998; 273: 10099–106.
Seasholtz TM, Majumdar M, Kaplan DD, Brown JH . Rho and Rho kinase mediate thrombin-stimulated vascular smooth muscle cell DNA synthesis and migration. Circ Res 1999; 84: 1186–93.
Clerk A, Sugden PH . Small guanine nucleotide-binding proteins and myocardial hypertrophy. Circ Res 2000; 86: 1019–23.
Ito K, Hirooka Y, Sakai K, Kishi T, Kaibuchi K, Shimokawa H, et al. Rho/Rho-kinase pathway in brain stem contributes to blood pressure regulation via sympathetic nervous system: possible involvement in neural mechanisms of hypertension. Circ Res 2003; 92: 1337–43.
Ito K, Hirooka Y, Kishi T, Kimura Y, Kaibuchi K, Shimokawa H, et al. Rho/Rho-kinase pathway in the brainstem contributes to hypertension caused by chronic nitric oxide synthase inhibition. Hypertension 2004; 43: 156–62.
Yamakawa T, Tanaka S, Numaguchi K, Yamakawa Y, Motley ED, Ichihara S, et al. Involvement of Rho-kinase in angiotensin II-induced hypertrophy of rat vascular smooth muscle cells. Hypertension 2000; 35: 313–8.
Mukai Y, Shimokawa H, Matoba T, Kandabashi T, Satoh S, Hiroki J, et al. Involvement of Rho-kinase in hypertensive vascular disease: a novel therapeutic target in hypertension. FASEB J 2001; 15: 1062–4.
Nirro N, Nishimura J, Sakihara C, Nakano H, Kanaide H . Upregulation of Rho A and Rho-kinase mRNAs in the rat myometrium during pregnancy. Biochem Biophys Res Commun 1997; 230: 356–9.
Kaarbo M, Crane DI, Murrell WG . RhoA is highly up-regulated in the process of early heart development of the chick and important for normal embryogenesis. Dev Dyn 2003; 227: 35–47.
Hu E, Lee D . Rho kinase inhibitors as potential therapeutic agents for cardiovascular diseases. Curr Opin Investig Drugs 2003; 4: 1065–75.
Wei L, Wang L, Carson JA, Agan JE, Imanaka-Yoshida K, Schwartz RJ . beta1 Integrin and organized actin filaments facilitate cardiomyocyte-specific RhoA-dependent activation of the skeletal alpha-actin promoter. FASEB J 2001; 15: 785–96.
Kawamura S, Miyamoto S, Brown JH . Initiation and transduction of stretch-induced RhoA and Rac1 activation through caveolae: cytoskeletal regulation of ERK translocation. J Biol Chem 2003; 278: 31111–7.
Charron F, Tsimiklis G, Arcand M, Robitaille L, Liang Q, Molkentin JD, et al. Tissue-specific GATA factors are transcriptional effectors of the small GTPase RhoA. Genes Dev 2001; 15: 2702–19.
Satoh S, Ueda Y, Koyanagi M, Kadokami T, Sugano M, Yoshikawa Y, et al. Chronic inhibition of Rho kinase blunts the process of left ventricular hypertrophy leading to cardiac contractile dysfunction in hypertension-induced heart failure. J Mol Cell Cardiol 2003; 35: 59–70.
Sah VP, Minamisawa S, Tam SP, Wu TH, Dorn GW, Ross J Jr, et al. Cardiac-specific overexpression of RhoA results in sinus and atrioventricular nodal dysfunction and contractile failure. J Clin Invest 1999; 103: 1627–34.
Nakagami H, Liao JK . Statins and myocardial hypertrophy. Coron Artery Dis 2004; 15: 247–50.
Kobayashi N, Horinaka S, Mita S, Nakano S, Honda T, Yoshida K, et al. Critical role of Rho-kinase pathway for cardiac performance and remodeling in failing rat hearts. Cardiovasc Res 2002; 55: 757–67.
Wei L, Taffet GE, Khoury DS, Bo J, Li Y, Yatani A, et al. Disruption of Rho signaling results in progressive atrioventricular conduction defects while ventricular function remains preserved. FASEB J 2004; 18: 857–9.
Somlyo AP, Somlyo AV . Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: modulated by G proteins, kinases, and myosin phosphatase. Physiol Rev 2003; 83: 1325–58.
Surks HK, Richards CT, Mendelsohn ME . Myosin phosphatase-Rho interacting protein: a new member of the myosin phosphatase complex that directly binds RhoA. J Biol Chem 2003; 278: 51484–93.
Ren J, Davidoff AJ . Diabetes rapidly induces contractile dysfunctions in isolated ventricular myocytes. Am J Physiol Heart Circ Physiol 1997; 272: H148–H158.
Ren J, Bode AM . Altered cardiac excitation-contraction coupling in ventricular myocytes from spontaneously diabetic BB rats. Am J Physiol Heart Circ Physiol 2000; 279: H238–44.
Duan J, Zhang HY, Adkins SD, Ren BH, Norby FL, Zhang X, et al. Impaired cardiac function and IGF-I response in myocytes from calmodulin-diabetic mice: role of Akt and RhoA. Am J Physiol Endocrinol Metab 2003; 284: E366–76.
Dillmann WH . Diabetes and thyroid-hormone-induced changes in cardiac function and their molecular basis. Annu Rev Med 1989; 40: 373–94.
Hofmann PA, Menon V, Gannaway KF . Effects of diabetes on isometric tension as a function of [Ca2+] and pH in rat skinned cardiac myocytes. Am J Physiol Heart Circ Physiol 1995; 269: H1656–63.
Chattou S, Diacono J, Feuvray D . Decrease in sodium-calcium exchange and calcium currents in diabetic rat ventricular myocytes. Acta Physiol Scand 1999; 166: 137–44.
Miao L, Calvert JW, Tang J, Parent AD, Zhang JH . Age-related RhoA expression in blood vessels of rats. Mech Ageing Dev 2001; 122: 1757–70.
Seasholtz TM, Zhang T, Morissette MR, Howes AL, Yang AH, Brown JH . Increased expression and activity of RhoA are associated with increased DNA synthesis and reduced p27(Kip1) expression in the vasculature of hypertensive rats. Circ Res 2001; 89: 488–95.
Massey AR, Miao L, Smith BN, Liu J, Kusaka I, Zhang JH, et al. Increased RhoA translocation in renal cortex of diabetic rats. Life Sci 2003; 72: 2943–52.
Sauzeau V, Rolli-Derkinderen M, Marionneau C, Loirand G, Pacaud P . RhoA expression is controlled by nitric oxide through cGMP-dependent protein kinase activation. J Biol Chem 2003; 278: 9472–80.
Singh LP, Andy J, Anyamale V, Greene K, Alexander M, Crook ED . Hexosamine-induced fibronectin protein synthesis in mesangial cells is associated with increases in cAMP responsive element binding (CREB) phosphorylation and nuclear CREB: the involvement of protein kinases A and C. Diabetes 2001; 50: 2355–62.
Ichiki T, Tokunou T, Fukuyama K, Iino N, Masuda S, Takeshita A . Cyclic AMP response element-binding protein mediates reactive oxygen species-induced c-fos expression. Hypertension 2003; 42: 177–83.
Cachero TG, Morielli AD, Peralta EG . The small GTP-binding protein RhoA regulates a delayed rectifier potassium channel. Cell 1998; 93: 1077–85.
Shimoni Y, Firek L, Severson D, Giles W . Short-term diabetes alters K+ currents in rat ventricular myocytes. Circ Res 1994; 74: 620–8.
Norby FL, Aberle NS 2nd, Kajstura J, Anversa P, Ren J . Transgenic overexpression of insulin-like growth factor I prevents streptozotocin-induced cardiac contractile dysfunction and beta-adrenergic response in ventricular myocytes. J Endocrinol 2004; 180: 175–82.
Kajstura J, Fiordaliso F, Andreoli AM, Li B, Chimenti S, Medow MS, et al. IGF-1 overexpression inhibits the development of diabetic cardiomyopathy and angiotensin II-mediated oxidative stress. Diabetes 2001; 50: 1414–24.
Norby FL, Wold LE, Duan J, Hintz KK, Ren J . IGF-I attenuates diabetes-induced cardiac contractile dysfunction in ventricular myocytes. Am J Physiol Endocrinol Metab 2002; 283: E658–66.
Ren J, Duan J, Hintz KK, Ren BH . High glucose induces cardiac insulin-like growth factor I resistance in ventricular myocytes: role of Akt and ERK activation. Cardiovasc Res 2003; 57: 738–48.
Mohri M, Shimokawa H, Hirakawa Y, Masumoto A, Takeshita A . Rho-kinase inhibition with intracoronary fasudil prevents myocardial ischemia in patients with coronary microvascular spasm. J Am Coll Cardiol 2003; 41: 15–9.
Bao W, Hu E, Tao L, Boyce R, Mirabile R, Thudium DT, et al. Inhibition of Rho-kinase protects the heart against ischemia/reperfusion injury. Cardiovasc Res 2004; 61: 548–58.
Mozzicato S, Joshi BV, Jacobson KA, Liang BT . Role of direct RhoA-phospholipase D1 interaction in mediating adenosine-induced protection from cardiac ischemia. FASEB J 2004; 18: 406–8.
Shimokawa H . Rho-kinase as a novel therapeutic target in treatment of cardiovascular diseases. J Cardiovasc Pharmacol 2002; 39: 319–27.
Glyn MC, Lawrenson JG, Ward BJ . A Rho-associated kinase mitigates reperfusion-induced change in the shape of cardiac capillary endothelial cells in situ. Cardiovasc Res 2003; 57: 195–206.
Sanada S, Asanuma H, Tsukamoto O, Minamino T, Node K, Takashima S, et al. Protein kinase A as another mediator of ischemic preconditioning independent of protein kinase C. Circulation 2004; 110: 51–7.
Hattori T, Shimokawa H, Higashi M, Hiroki J, Mukai Y, Tsutsui H, et al. Long-term inhibition of Rho-kinase suppresses left ventricular remodeling after myocardial infarction in mice. Circulation 2004; 109: 2234–9.
Ishizaki T, Morishima Y, Okamoto M, Furuyashiki T, Kato T, Narumiya S . Coordination of microtubules and actin cytoskeleton by the Rho effector mDial. Nature Cell Biol 2001; 3: 8–14.
Somlyo AV . New roads leading to Ca2+ sensitization. Circ Res 2002; 91: 83–4.
Glotzer M . Cytokinesis: progress on all fronts. Curr Opin Cell Biol 2003; 15: 684–90.
McBeath R, Pirone DM, Nelson CM, Bhadriraju K, Chen CS . Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell 2004; 6: 483–95.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, J., Fang, C. Small guanine nucleotide-binding protein Rho and myocardial function. Acta Pharmacol Sin 26, 279–285 (2005). https://doi.org/10.1111/j.1745-7254.2005.00059.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00059.x
Keywords
This article is cited by
-
The 2A2 protein of Duck hepatitis A virus type 1 induces apoptosis in primary cell culture
Virus Genes (2016)
-
Microdomain heterogeneity in 3D affects the mechanics of neonatal cardiac myocyte contraction
Biomechanics and Modeling in Mechanobiology (2013)
-
Long-term administration of rosuvastatin prevents contractile and electrical remodelling of diabetic rat heart
Journal of Bioenergetics and Biomembranes (2013)