Abstract
Aim:
To study the pharmacokinetics and accumulation of an Escherichia coli expressed His-tag fused recombinant human endostatin (rh-endostatin) in Rhesus monkeys.
Methods:
Rh-endostatin was iv or sc injected in Rhesus monkeys, and the rh-endostatin concentration in serum samples was determined by an enzyme immunoassay (EIA) method. The serum drug concentration-time data were analyzed by compartmental analysis using the practical pharmacokinetic program 3p97.
Results:
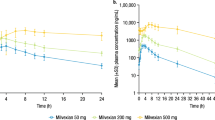
Following iv administration at a dose rate of 1.5, 4.5, and 13.5 mg/kg in rhesus monkeys, the concentration-time curves of rh-endostatin were best fitted to a three-compartment open model. AUC(0-∞) linearly increased with dose, while Cls exhibited no significant difference among different dose groups. The terminal half-lives (λ3) were 8±8, 3.1±1.4, and 20±14 h, respectively. After sc administration at a dose rate of 1.5 mg/kg, the concentration-time curve was best fitted to a two-compartment open model, with a terminal half-life (T1/2β) of 8±3 h. Bioavailability following sc injection was approximately 70%±3%. After consecutive iv injection of rh-endostatin at a dose rate of 1.5 mg·kg−1·d−1 for 7 d, the AUC(0-24 h) substantially increased from 22±13 mg·h·L−1 (d 1) to 50±29 mg·h·L−1 (d 7), with an accumulation factor of 2.3±0.6 (P < 0.05).
Conclusion:
The pharmacokinetic behavior of rh-endostatin in Rhesus monkeys complies with linear kinetics within the examined dose range. It tends to be accumulated in bodies after repeated administration at a dose level of 1.5 mg·kg−1·d−1 for more than 7 consecutive days.
Similar content being viewed by others
Article PDF
References
Sasaki T, Fukai N, Mann K, Gohring W, Olsen BR, Timpl R . Structure, function and tissue forms of the C-terminal globular domain of collagen XVIII containing the angiogenesis inhibitor endostatin. EMBO J 1998; 17: 4249–56.
O'Reilly MS, Boehm T, Shing Y, Fukai N, Vasios G, Lane WS, et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 1997; 88: 277–85.
Beohm T, Folkman J, Browder T, O'Reilly MS . Antiangiogenic therapy of experimental cancer does not induce acquired drug resistance. Nature 1997; 390: 404–7.
Zatterstrom UK, Felbor U, Fukai N, Olsen BR . Collagen XVIII/endostatin structure and functional role in angiogenesis. Cell Struct Funct 2000; 25: 97–101.
Iughetti P, Suzuki O, Godoi PH, Alves VA, Sertie AL, Zorick T, et al. A polymorphism in endostatin, an angiogenesis inhibitor, predisposes for the development of prostatic adenocarcinoma. Cancer Res 2001; 61: 7375–8.
Maio M, Altomonte M, Calabro L, Fonsatti E . Bioimmunotherapeutic targets on angiogenetic blood vessels in solid malignangies. Front Biosci 2001; 6: D776–84.
Ryan CJ, Wilding G . Angiogenesis inhibitors. New agents in cancer therapy. Drugs Aging 2000; 17: 249–55.
Mundhenke C, Thomas JP, Wilding G, Lee FT, Kelzc F, Chappell R, et al. Tissue examination to monitor antiangiogenic therapy: a phase I clinical trial with endostatin. Clin Cancer Res 2001; 7: 3366–74.
Huang X, Wong MK, Zhao Q, Zhu Z, Wang KZ, Huang N, et al. Soluble recombinant endostatin purified from Escherichia coli: antiangiogenic activity and antitumor effect. Cancer Res 2001; 61: 478–81.
Hengen P . Purification of His-Tag fusion proteins from Escherichia coli. Trends Biochem Sci 1995; 20: 285–6.
Tallet B, Astier-Gin T, Castroviejo M, Santarelli X . One-step chromatographic purification procedure of a His-tag recombinant carboxyl half part of the HTLV-I surface envelope glycoprotein overexpressed in Escherichia coli as a secreted form. J Chromatogr B Biomed Sci Appl 2001; 753: 17–22.
Gibert S, Bakalara N, Santarelli X . Three-step chromatographic purification procedure for the production of a his-tag recombinant kinesin overexpressed in E. coli. J Chromatogr B Biomed Sci Appl 2000; 737: 143–50.
Huang YW, Lu ML, Qi H, Lin SX . Membrane-bound human 3beta-hydroxysteroid dehydrogenase: overexpression with His-tag using a baculovirus system and single-step purification. Protein Expr Purif 2000; 18: 169–74.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China, No 39930180.
Rights and permissions
About this article
Cite this article
Song, Hf., Liu, Xw., Zhang, Hn. et al. Pharmacokinetics of His-tag recombinant human endostatin in Rhesus monkeys. Acta Pharmacol Sin 26, 124–128 (2005). https://doi.org/10.1111/j.1745-7254.2005.00009.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00009.x
Keywords
This article is cited by
-
A comparison of the efficacy of antiangiogenic agents combined with chemotherapy for the treatment of non-small cell lung cancer: a network meta-analysis
Cancer Cell International (2020)
-
Thoracic perfusion of recombinant human endostatin (Endostar) combined with chemotherapeutic agents versus chemotherapeutic agents alone for treating malignant pleural effusions: a systematic evaluation and meta-analysis
BMC Cancer (2016)
-
Pharmacokinetics of PEGylated recombinant human endostatin (M2ES) in rats
Acta Pharmacologica Sinica (2015)
-
Antiangiogenic agents combined with chemotherapy in non-small cell lung cancer
Oncology and Translational Medicine (2015)
-
Expression of liver-targeting peptide modified recombinant human endostatin and preliminary study of its biological activities
Applied Microbiology and Biotechnology (2014)