Abstract
Aim:
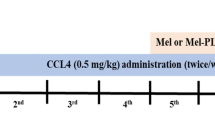
Melatonin-selenium nanoparticle (MT-Se), a novel complex, was synthesized by preparing selenium nanoparticles in a melatonin medium. The present investigation was designed to determine the protective effects of MT-Se against immunological liver injury in mice induced by bacillus Calmette-Guérin (BCG)/ lipopolysaccharide (LPS).
Methods:
The model of immunological liver injury in mice was prepared. The levels of alanine aminotransferase, aspartate aminotransferase, nitric oxide (NO) in serum, malondialdehyde content, superoxide dismutase (SOD), and glutathione peroxidase (GSH-px) activities in a liver homogenate were assayed by spectrophotometry. The content of tumor necrosis factor-α (TNF-α) and interleukin-1 (IL-1) were determined by ELISA. The splenocyte proliferation was assayed by 3-[4,5-dimethylthiazol-2-yl] -2,5-diphenyltetra-zolium bromide (MTT) dye reduction. Meanwhile, a hepatic pathological examination was observed.
Results:
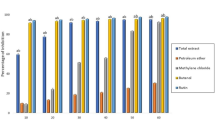
In the BCG/LPS-induced hepatic injury model, MT-Se administered at doses of 5, 10, or 20 mg/kg to the BCG/LPS-treated mice for 10 d significantly reduced the increase in serum aminotransferase, reduced the severe extent of hepatic cell damage and the immigration of inflammatory cells. It also attenuated the increase in the content of thiobarbituric acid-reactive substances and enhanced the decrease in activities of SOD and GSH-px. In contrast, the treatment with MT-Se suppressed the increase in NO level in both the serum and liver tissue. Furthermore, MT-Se significantly lowered an increase in TNF-α and IL-1α levels in the liver and inhibited the production of TNF-α and IL-1α by peritoneal macrophages. A downregulation effect of MT-Se on splenocyte proliferation was also observed.
Conclusion:
MT-Se showed a hepatic protective action on immunological liver injury in mice.
Similar content being viewed by others
Article PDF
References
Hoek JB, Pastorino JG . Ethanol, oxidative stress, and cytokine-induced liver cell injury. Alcohol 2002; 27: 63–8.
Wang GS, Liu GT . Influences of Kupffer cell stimulation and suppression on immunological liver injury in mice. Acta Pharmacol Sin 1997; 18: 173–6.
Guler R, Olleros ML, Vesin D, Parapanov R, Vesin C, Kantengwa S, et al. Inhibition of inducible nitric oxide synthase protects against liver injury induced by mycobacterial infection and endotoxins. J Hepatol 2004; 41: 773–81.
Nagakawa J, Hishinuma I, Hirota K, Miyamoto K, Yamanaka T, Tsukidate K, et al. Involvement of tumor necrosis factor-α in the pathogenesis of activated macrophage mediated hepatitis in mice. Gastroenterology 1990; 99: 758–65.
Tsuji H, Harada A, Mukaida N, Nakanuma Y, Bluethmann H, Kaneko S, et al. Tumor necrosis factor receptor p55 is essential for intrahepatic granuloma formation and hepatocellular apoptosis in a murine model of bacterium-induced fulminant hepatitis. Infect Immun 1997; 65: 1892–8.
Cuzzocrea S, Reiter RJ . Pharmacological actions of melatonin in acute and chronic inflammation. Curr Top Med Chem 2002; 2: 153–65.
Guerrero JM, Reiter RJ . Melatonin-immune system relationships. Curr Top Med Chem 2002; 2: 167–79.
Reiter RJ, Tan DX, Mayo JC, Sainz RM, Leon J, Czarnocki Z . Melatonin as an antioxidant: biochemical mechanisms and pathophysiological implications in humans. Acta Biochim Pol 2003; 50: 1129–46.
Rodriguez C, Mayo JC, Sainz RM, Antolin I, Herrera F, Martin V, et al. Regulation of antioxidant enzymes: a significant role for melatonin. J Pineal Res 2004; 36: 1–9.
Wang H, Wei W, Shen YX, Dong C, Zhang LL, Wang NP, et al. Protective effect of melatonin against liver injury in mice induced by Bacillus Calmette-Guerin plus lipopolysaccharide. World J Gastroenterol 2004; 10: 2690–6.
Bruck R, Aeed H, Avni Y, Shirin H, Matas Z, Shahmurov M, et al. Melatonin inhibits nuclear factor kappa B activation and oxidative stress and protects against thioacetamide induced liver damage in rats. J Hepatol 2004; 40: 86–93.
Jozanov-Stankov O, Demajo M, Djujic I, Mandic M . Selenium intake as a modulator of responsiveness to oxidative stress. J Environ Pathol Toxicol Oncol 1998; 17: 251–7.
Tomei FA, Barton LL, Semanski CL . Transformation of selenate and selenite to elemental selenium by Desulfovibrio desulfuricans. J Ind Microbiol 1995; 14: 329–36.
Zhang JS, Gao XY, Zhang LD, Bao YP . Biological effects of a nano red elemental selenium. Biofactors 2001; 15: 27–38.
Mees DR, Pysto W, Tarcha PJ . Formation of selenium colloids using sodium ascorbate as the reducing agent. J Colloid Interface Sci 1995; 170: 254–90.
Shechter I, Fogelman AM, Haberland ME, Seager J, Hokom M, Edwards PA . Effect of polyunsaturated fatty acids and antioxidants on lipid peroxidation in tissue cultures. J Lipid Res 1981; 22: 63–9.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ . Protein measurement with the Folin-phenol reagent. J Biol Chem 1951; 193: 265–73.
Sykes JA, McCormack FX Jr, O'Brien TJ . A preliminary study of the superoxide dismutase content of some human tumors. Cancer Res 1978; 38: 2759–62.
Paglia DE, Valentine WN . Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 1967; 70: 58–169.
Kiechle FL, Malinski T . Nitric oxide. Biochemistry, pathophysiology, and detection. Am J Clin Pathol 1993; 100: 567–75.
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG . Selenium: biochemical role as a component of glutathione peroxidase. Science 1973; 179: 588–90.
Sakaguchi S, Iizuka Y, Furusawa S, Tanaka Y, Takayanagi M, Takayanagi Y . Roles of selenium in endotoxin-induced lipid peroxidation in the rats liver and in nitric oxide production in J774A.1cells. Toxicol Lett 2000; 118: 69–77.
Ozdil S, Bolkent S, Yanardag R, Arda-Pirincci P . Protective effects of ascorbic acid, dl-alpha-tocopherol acetate, and sodium selenate on ethanol-induced liver damage of rats. Biol Trace Elem Res 2004; 97: 149–62.
Kono H, Arteel GE, Rusyn I, Sies H, Thurman RG . Ebselen prevents early alcohol-induced liver injury in rats. Free Radic Biol Med 2001; 30: 403–11.
Yu MW, Horng IS, Hsu KH, Chiang YC, Liaw YF, Chen CJ . Plasma selenium levels and the risk of hepatocellular carcinoma among men with chronic hepatitis virus infection. Am J Epidemiol 1999; 150: 367–74.
Rayman MP . The importance of selenium to human health. Lancet 2000; 356: 233–41.
Calvo JR, Reiter RJ, Garcia JJ, Ortiz GG, Tan DX, Karbownik M . Characterization of the protective effects of melatonin and related indoles against alpha-naphthylisothiocyanate-induced liver injury in rats. J Cell Biochem 2001; 80: 461–70.
Ohta Y, Kongo-Nishimura M, Matsura T, Yamada K, Kitagawa A, Kishikawa T . Melatonin prevents disruption of hepatic reactive oxygen species metabolism in rats treated with carbon tetrachloride. J Pineal Res 2004; 36: 10–7.
Okatani Y, Wakatsuki A, Reiter RJ, Enzan H, Miyahara Y . Protective effect of melatonin against mitochondrial injury induced by ischemia and reperfusion of rat liver. Eur J Pharmacol 2003; 469: 145–52.
Sener G, Tosun O, Sehirli AO, Kacmaz A, Arbak S, Ersoy Y, et al. Melatonin and N-acetylcysteine have beneficial effects during hepatic ischemia and reperfusion. Life Sci 2003; 72: 2707–18.
Ohta Y, Kongo M, Kishikawa T . Melatonin exerts a therapeutic effect on cholestatic liver injury in rats with bile duct ligation. J Pineal Res 2003; 34: 119–26.
Sener G, Sehirli AO, Ayanoglu-Dulger G . Protective effects of melatonin, vitamin E and N-acetylcysteine against acetaminophen toxicity in mice: a comparative study. J Pineal Res 2003; 35: 61–8.
Shin S, Hur GH, Kim YB, Yeon GB, Park KJ, Park YM, et al. Dehydroepiandrosterone and melatonin prevent Bacillus anthracis lethal toxin-induced TNF production in macrophages. Cell Biology Toxicol 2000; 16: 165–74.
Sacco S, Aquilini L, Ghezzi P, Pinza M, Guglielmotti A . Mechanism of the inhibitory effect of melatonin on tumor necrosis factor production in vivo and in vitro. Eur J Pharmacol 1998; 343: 249–55.
Maestroni G.J . Melatonin as a therapeutic agent in experimental endotoxic shock. J Pineal Res 1996; 20: 84–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Key Project of the Natural Science Foundation of the Department of Education, Anhui Province (No 98JL0130Z).
Rights and permissions
About this article
Cite this article
Wang, H., Wei, W., Zhang, Sy. et al. Melatonin-selenium nanoparticles protects liver against immunological injury induced by bacillus Calmette-Guérin and lipopolysaccharide. Acta Pharmacol Sin 26, 745–752 (2005). https://doi.org/10.1111/j.1745-7254.2005.00745.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00745.x
Keywords
This article is cited by
-
Nanotechnology-based advances in the efficient delivery of melatonin
Cancer Cell International (2022)
-
Pre-Clinical Study for the Antidiabetic Potential of Selenium Nanoparticles
Biological Trace Element Research (2017)