Abstract
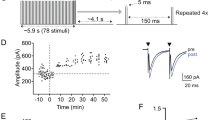
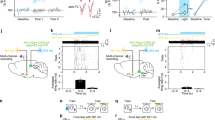
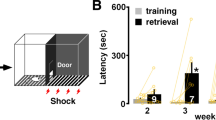
Long-term potentiation (LTP), the increase in synaptic strength evoked by high-frequency stimulation, is often considered to be a cellular model for learning and memory. The validity of this model depends on the assumptions that physiological stimuli can induce LTP in vivo and that the resulting synaptic modifications correlate with behavioural changes. However, modifiable synapses are generally embedded deep in complex circuits. In contrast, the goldfish Mauthner (M)-cell and its afferent synapses are easily accessible for electrophysiological studies, and firing of this neuron is sufficient to trigger fast escape behaviour in response to sudden stimuli1,2. We have previously shown that tetanic stimulation can induce LTP of the feedforward inhibitory synapses that control the excitability of the M-cell3,4. Here we report that natural sensory stimulation can induce potentiation of this inhibitory connection that resembles the LTP induced by afferent tetanization. Furthermore, comparable acoustic stimulation produced a parallel decrease in the probability of the sound-evoked escape reflex. Thus we demonstrate for the first time, to our knowledge, a behavioural role for the long-term synaptic strengthening of inhibitory synapses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zottoli, S. J. Correlation of the startle reflex and Mauthner cell auditory responses in unrestrained goldfish. J. Exp. Biol. 66, 243–254 (1977).
Faber, D. S. & Korn, H. Neurobiology of the Mauthner Cell (Raven, New York, (1978)).
Korn, H., Oda, Y. & Faber, D. S. Long-term potentiation of inhibitory circuits and synapses in the CNS. Proc. Natl Acad. Sci. USA 89, 440–443 (1992).
Oda, Y., Charpier, S., Murayama, Y., Suma, C. & Korn, H. Long-term potentiation of glycinergic inhibitory synaptic transmission. J. Neurophysiol. 74, 1056–1074 (1995).
Fetcho, J. R. Spinal network of the Mauthner cell. Brain Behav. Evol. 37, 298–316 (1991).
Zottoli, S. J. & Faber, D. S. An identifiable class of statoacoustic interneurons with bilateral projections in the goldfish medulla. Neuroscience 5, 1287–1302 (1980).
Faber, D. S. & Korn, H. Transmission at a central inhibitory synapse. I. Magnitude of unitary postsynaptic conductance change and kinetics of channel activation. J. Neurophysiol. 48, 654–678 (1982).
Faber, D. S. & Korn, H. Unitary conductance changes at teleost Mauthner cell glycinergic synapses: a voltage-clamp and pharmacological analysis. J. Neurophysiol. 60, 1982–1999 (1988).
Furukawa, T. & Ishii, Y. Neurophysiological studies on hearing in goldfish. J. Neurophysiol. 30, 1377–1403 (1967).
Fay, R. R. Physiology of primary saccular afferents of goldfish: Implications for Mauthner cell response. Brain Behav. Evol. 46, 141–150 (1995).
Furukawa, T. & Furshpan, E. J. Two inhibitory mechanisms in the Mauthner neurons of the goldfish. J.Neurophysiol. 26, 140–176 (1963).
Eaton, R. C., DiDomenico, R. & Nissanov, J. Flexible body dynamics of the goldfish C-start: Implications for reticulospinal command mechanisms. J. Neurosci. 8, 2758–2768 (1988).
Eaton, R. C. & Emberley, D. S. How stimulus direction determines the trajectory of the Mauthner-initiated escape response in a teleost fish. J. Exp. Biol. 161, 469–487 (1991).
Rotenberg, A., Mayford, M., Hawkins, R. D., Kandel, E. R. & Muller, R. U. Mice expressing activated CaMKII lack low frequency LTP and do not form stable place cells in the CA1 region of the hippocampus. Cell 87, 1351–1361 (1996).
Wilson, M. A. & Tonegawa, S. Synaptic plasticity, place cells and spatial memory: study with second generation knockouts. Trends Neurosci. 20, 102–106 (1997).
Rogan, M. T., Stäubli, U. V. & LeDoux, J. E. Fear conditioning induces associative long-term potentiation in the amygdala. Nature 390, 604–607 (1997).
McKernan, M. G. & Shinnick-Gallagher, P. Fear conditioning induces a lasting potentiation of synaptic currents in vitro. Nature 390, 607–611 (1997).
Yang, X. D., Korn, H. & Faber, D. S. Long-term potentiation of electrotonic coupling at mixed synapses. Nature 348, 542–545 (1990).
Yang, X. D. & Faber, D. S. Initial synaptic efficacy influences induction and expression of long-term changes in transmission. Proc. Natl Acad. Sci. USA 88, 4299–4303 (1991).
Vu, E. T., Lee, S. C. & Krasne, F. B. The mechanism of tonic inhibition of crayfish escape behavior: Distal inhibition and its functional significance. J. Neurosci. 13, 4379–4393 (1993).
Fisher, T. M. & Carew, T. J. Cutaneous activation of the inhibitory L30 interneurons provides a mechanism for regulating adaptive gain control in the siphon withdrawal reflex of Aplysia. J. Neurosci. 15, 762–773 (1995).
Faber, D. S. & Korn, H. Inputs from the posterior lateral line nerves upon the goldfish Mauthner cell. II. Evidence that the inhibitory components are mediated by interneurons of the recurrent collateral network. Brain Res. 96, 349–356 (1975).
Rodgers, W. L., Melzack, R. & Segal, J. R. “Tail flip response” in goldfish. J. Comp. Physiol. Psychol. 56, 917–923 (1963).
Aljure, E., Day, J. W. & Bennett, M. V. L. Postsynaptic depression of Mauthner cell-mediated startle reflex, a possible contributor to habituation. Brain Res. 188, 261–268 (1980).
Groves, P. M. & Thompson, R. F. Habituation: a dual-process theory. Psychol. Rev. 77, 419–450 (1970).
Kandel, E. R. Acell-biological approach to learning. Grass Lecture Monogr. 1 (Soc. Neurosci., Bethesda, (1978)).
Blaxter, J. H. S. in Sensory Biology of Aquatic Animals (eds Atema, J., Fay, R. R., Popper, A. N. & Tavolga, W. N.) 203–232 (Springer, New York, (1988)).
Canfield, J. G. & Rose, G. J. Hierarchical sensory guidance of Mauthner-mediated escape responses in goldfish (Carassius auratus) and Cichlids (Haplochromis burtoni). Brain Behav. Evol. 48, 137–156 (1996).
Acknowledgements
We thank D. S. Faber, R. Miles, E. S. Ruthazer, F. Murakami, N. Yamamoto and W.-J. Song for comments. This work was supported by Grant-in-Aid for Scientific Research on Priority Areas of Japan to Y.O.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oda, Y., Kawasaki, K., Morita, M. et al. Inhibitory long-term potentiation underlies auditory conditioning of goldfish escape behaviour. Nature 394, 182–185 (1998). https://doi.org/10.1038/28172
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/28172
This article is cited by
-
Rotational stimulation-related changes of the motor asymmetry in the goldfish
Neurophysiology (2005)
-
Asymmetry of Motor Behavior of the Goldfish in a Narrow Channel
Neurophysiology (2005)
-
Activity-dependent modification of inhibitory synapses in models of rhythmic neural networks
Nature Neuroscience (2001)
-
Visual input induces long-term potentiation of developing retinotectal synapses
Nature Neuroscience (2000)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.