Abstract
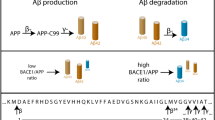
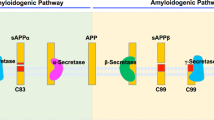
Proteolytic processing of the amyloid precursor protein (APP) generates amyloid β (Aβ) peptide, which is thought to be causal for the pathology and subsequent cognitive decline in Alzheimer's disease. Cleavage by β-secretase at the amino terminus of the Aβ peptide sequence, between residues 671 and 672 of APP, leads to the generation and extracellular release of β-cleaved soluble APP1, and a corresponding cell-associated carboxy-terminal fragment. Cleavage of the C-terminal fragment by γ-secretase(s) leads to the formation of Aβ. The pathogenic mutation K670M671 → N670L671 at the β-secretase cleavage site in APP2, which was discovered in a Swedish family with familial Alzheimer's disease, leads to increased β-secretase cleavage of the mutant substrate3. Here we describe a membrane-bound enzyme activity that cleaves full-length APP at the β-secretase cleavage site, and find it to be the predominant β-cleavage activity in human brain. We have purified this enzyme activity to homogeneity from human brain using a new substrate analogue inhibitor of the enzyme activity, and show that the purified enzyme has all the properties predicted for β-secretase. Cloning and expression of the enzyme reveals that human brain β-secretase is a new membrane-bound aspartic proteinase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Seubert,P. et al. Secretion of beta-amyloid precursor protein cleaved at the amino terminus of the beta-amyloid peptide. Nature 361, 260–263 (1993).
Mullan,M. et al. A pathogenic mutation for probably Alzheimer's disease in the APP gene at the N-terminus of beta-amyloid. Nature Genet. 1, 345–347 (1992).
Citron,M. et al. Mutation of the beta-amyloid precursor protein in familial Alzheimer's disease increases beta-protein secretion. Nature 360, 672–674 (1992).
Knops,J. et al. Cell-type and amyloid protein-type specific inhibition of Ab release by bafilomycin A1, a selective inhibitor of vacuolar ATPases. J. Biol. Chem. 270, 2419–2422 (1995).
Valdes,F., Munoz,C., Feria-Velasco,A. & Orrego,F. Subcellular distribution of rat brain cortex high-affinity, sodium-dependent, glycine transport sites. Brain Res. 122, 95–112 (1977).
Narutaki,S., Dunn,B. M. & Oda,K. Subsite preference of pepstatin-insensitive carboxyl proteinases from bacteria. J. Biochem. (Tokyo) 125, 75–81 (1999).
Schechter,I. & Berger,A. On the size of the active site in proteinases. I. Papain. Biochem. Biophys. Res. Commun. 27, 157–162 (1967).
Rich,D. H. in Proteinase Inhibitors (eds Barrett, A. J. & Salvesen, G.) (Elsevier Science, Amsterdam, 1986).
Johnson-Wood,K. et al. Amyloid precursor protein processing and Aβ42 deposition in a transgenic mouse model of Alzheimer disease. Proc. Natl Acad. Sci. USA 94, 1550–1555 (1997).
Vassar,R. et al. β-secretase cleavage of the Alzheimer's amyloid precursor protein by the transmembrane aspartic protease BACE. Science 286, 735–741 (1999).
Lin,J. H., Ostovic,D. & Vacca,J. P. The integration of medicinal chemistry, drug metabolism, and pharmaceutical research and development. The story of Crixivan, an HIV proteinase inhibitor. Pharm. Biotechnol. 11, 233–255 (1998).
Knops,J. et al. Isolation of Baculovirus-derived secreted and full-length β-amyloid precursor protein. J. Biol. Chem. 266, 7285–7290 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sinha, S., Anderson, J., Barbour, R. et al. Purification and cloning of amyloid precursor protein β-secretase from human brain. Nature 402, 537–540 (1999). https://doi.org/10.1038/990114
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/990114
This article is cited by
-
The Alzheimer’s disease-linked protease BACE1 modulates neuronal IL-6 signaling through shedding of the receptor gp130
Molecular Neurodegeneration (2023)
-
Mechanisms of amyloid-β34 generation indicate a pivotal role for BACE1 in amyloid homeostasis
Scientific Reports (2023)
-
Amyloid β-based therapy for Alzheimer’s disease: challenges, successes and future
Signal Transduction and Targeted Therapy (2023)
-
BACE1 in PV interneuron tunes hippocampal CA1 local circuits and resets priming of fear memory extinction
Molecular Psychiatry (2023)
-
The US9-Derived Protein gPTB9TM Modulates APP Processing Without Targeting Secretase Activities
Molecular Neurobiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.