Abstract
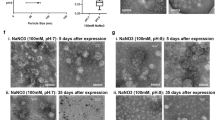
By means of a simple mixing procedure, we have constructed cationic Sendai virosomes consisting of fusogenic viral F/HN proteins and cationic lipids. Sendai virus F/HN proteins were purified by Triton X-100 treatment and sequential centrifugation, and then quantitatively added to cationic liposomes. The presence of HN proteins is essential for hemolytic activity of Sendai virus as well as efficient gene transfection. The amount of detergent added for purification of F/HN proteins was also crucial for hemolytic activity. The relevance of F/HN proteins in the gene-transfer capability of the cationic Sendai F/HN virosomes (CSVs) was verified by heat inactivation of the F/HN proteins, and cell-binding competition. DNA condensation by protamine sulfate was able to further enhance the transfection efficiency and serum resistance of CSV. The enhanced transfection efficiency of protamine-condensed DNA-encapsulating cationic Sendai F/HN virosomes (PCSVs) may result from specific and efficient cell binding mediated by F/HN proteins and efficient DNA encapsulation by protamine. The DNA condensation by protamine was crucial for systemic in vivo gene transfer by CSVs. The PCSVs exhibited a higher gene expression in various organs, especially the liver, compared to DOTAP/Chol lipoplexes. These results demonstrate the potential for the use of PCSV as gene delivery vehicles for systemic gene transfer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Marshall E . Gene therapy death prompts review of adenovirus vector. Science 1999; 286: 2244–2245.
Felgner PL, Gadek TR, Holm M, Roman R, Chan HW, Wenz M et al. Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. Proc Natl Acad Sci USA 1987; 84: 7413–7417.
Hofland HE, Shephard L, Sullivan SM . Formation of stable cationic lipid/DNA complexes for gene transfer. Proc Natl Acad Sci USA 1996; 93: 7305–7309.
Miao CH, Thompson AR, Loeb K, Ye X . Long-term and therapeutic-level hepatic gene expression of human factor IX after naked plasmid transfer in vivo. Mol Ther 2001; 3: 947–957.
Miao CH, Ohashi K, Patijn GA, Meuse L, Ye X, Thompson AR et al. Inclusion of the hepatic locus control region, an intron, and untranslated region increases and stabilizes hepatic factor IX gene expression in vivo but not in vitro. Mol Ther 2000; 1: 522–532.
Ehrhardt A, Xu H, Huang Z, Engler JA, Kay MA . A direct comparison of two nonviral gene therapy vectors for somatic integration: in vivo evaluation of the bacteriophage integrase phiC31 and the Sleeping Beauty transposase. Mol Ther 2005; 11: 695–706.
Nakanishi M, Uchida T, Kim J, Okada Y . Glycoproteins of Sendai virus (HVJ) have a critical ratio for fusion between virus envelopes and cell membranes. Exp Cell Res 1982; 142: 95–101.
Tomasi M, Pasti C, Manfrinato C, Dallocchio F, Bellini T . Peptides derived from the heptad repeat region near the C-terminal of Sendai virus F protein bind the hemagglutinin-neuraminidase ectodomain. FEBS Lett 2003; 536: 56–60.
Hsu MC, Scheidd A, Choppin PW . Fusion of Sendai virus with liposomes: dependence on the viral fusion protein (F) and the lipid composition of liposomes. Virology 1983; 126: 361–369.
Kaneda Y, Saeki Y, Morishita R . Gene therapy using HVJ-liposomes: the best of both worlds? Mol Med Today 1999; 5: 298–303.
Hirano T, Fujimoto J, Ueki T, Yamamoto H, Iwasaki T, Morisita R et al. Persistent gene expression in rat liver in vivo by repetitive transfections using HVJ-liposome. Gene Therapy 1998; 5: 459–464.
Tomita M, Morishita R, Higaki J, Tomita S, Aoki M, Ogihara T et al. In vivo gene transfer of insulin gene into neonatal rats by the HVJ-liposome method resulted in sustained transgene expression. Gene Therapy 1996; 3: 477–482.
Dzau VJ, Mann MJ, Morishita R, Kaneda Y . Fusigenic viral liposome for gene therapy in cardiovascular disease. Proc Natl Acad Sci USA 1996; 93: 11421–11425.
Kaneda Y, Nakajima T, Nishikawa T, Yamamoto S, Ikegami H, Suzuki N et al. Hemagglutinating virus of Japan (HVJ) envelope vector as a versatile gene delivery system. Mol Ther 2002; 6: 219–226.
Kim HS, Park YS . Gene transfection by quantitatively reconstituted Sendai envelope proteins into liposomes. Cancer Gene Ther 2002; 9: 173–177.
Tomasi M, Loyter A . Selective extraction of biologically active F-glycoprotein from dithiothreitol reduced Sendai virus particles. FEBS Lett 1981; 131: 381–385.
Bagai S, Puri A, Blumenthal R, Sarkar DP . Hemagglutinin–neuraminidase enhances is F protein-mediated membrane fusion of reconstituted Sendai virus envelopes with cells. J Virol 1993; 67: 3312–3318.
Vainstein A, Hershkovitz M, Israel S, Rabin S, Loyter A . A new method for reconstitution of highly fusogenic Sendai virus envelopes. Biochem Biophys Acta 1984; 773: 184–188.
Bitzer M, Lauer U, Baumann C, Spiegel M, Gregor M, Neubert WJ . Sendai virus efficiently infects cells via the asialoglycoprotein receptor and requires the presence of cleaved F0 precursor proteins for this alternative route of cell entry. J Virol 1997; 71: 5481–5486.
Zhang G, Gao X, Song YK, Vollmer R, Stolz DB, Gasiorowski JZ et al. Hydroporation as the mechanism of hydrodynamic delivery. Gene Therapy 2004; 11: 675–682.
Zhang G, Budker V, Wolff JA . High levels of foreign gene expression in hepatocytes after tail vein injections of naked plasmid DNA. Hum Gene Ther 1999; 10: 1735–1737.
Ohlfest JR, Frandsen JL, Fritz S, Lobitz PD, Perkinson SG, Clark KJ et al. Phenotypic correction and long-term expression of factor VIII in hemophilic mice by immunotolerization and nonviral gene transfer using the Sleeping Beauty transposon system. Blood 2005; 105: 2691–2698.
Held PK, Olivares EC, Aguilar CP, Finegold M, Calos MP, Grompe M . In vivo correction of murine hereditary tyrosinemia type I by phiC31 integrase-mediated gene delivery. Mol Ther 2005; 11: 399–408.
Takimoto T, Taylor GL, Connaris HC, Crennell SJ, Portner A . Role of hemagglutinin–neuraminidase protein in the mechanism of paramyxovirus-cell membrane fusion. J Virol 2002; 76: 13028–13033.
Rousso I, Mixon MB, Chen BK, Kim PS . Palmitoylation of the HIV-1 envelope glycoprotein is critical for viral infectivity. Proc Natl Acad Sci USA 2000; 97: 13523–13525.
Bavari S, Bosio CM, Wiegand E, Ruthel G, Will AB, Geisbert TW et al. Lipid rafts microdomains: a gateway for compartmentalized trafficking of Ebola and Marburg viruses. J Exp Med 2002; 195: 593–602.
Gao X, Huang L . Potentiation of cationic liposome-mediated gene delivery by polycations. Biochemistry 1996; 35: 1027–1036.
Bagai S, Sarkar DP . Fusion-mediated microinjection of lysozyme into HepG2 cells through hemagglutinin neuraminidase-depleted Sendai virus envelopes. J Biol Chem 1994; 269: 1966–1972.
Li S, Huang L . In vivo gene transfer via intravenous administration of cationic lipid–protamine–DNA (LPD) complexes. Gene Therapy 1997; 4: 891–900.
Morishita N, Nakagami H, Morishita R, Takeda S, Mishima F, Terazono B et al. Magnetic nanoparticles with surface modification enhanced gene delivery of HVJ-E vector. Biochem Biophys Res Commun 2005; 334: 1121–1126.
Sorgi FL, Bhattacharya S, Huang L . Protamine sulfate enhances lipid-mediated gene transfer. Gene Therapy 1997; 4: 961–968.
Ramesh R, Saeki T, Templeton NS, Ji L, Stephens LC, Ito I et al. Successful treatment of primary and disseminated human lung cancers by systemic delivery of tumor suppressor genes using an improved liposome vector. Mol Ther 2001; 3: 337–350.
Uyechi LS, Gagne L, Thurston G, Szoka FC . Mechanism of lipoplex gene delivery in mouse lung: binding and internalization of fluorescent lipid and DNA components. Gene Therapy 2001; 8: 828–836.
Delepine P, Guillaume C, Montier T, Clément JC, Yaouanc JJ, Des Abbayes H et al. Biodistribution study of phosphonolipids: a class of nonviral vectors efficient in mice lung directed gene transfer. J Gene Med 2003; 5: 600–608.
Nchinda G, Zschornig O, Uberla K . Increased non-viral gene transfer levels in mice by concentration of cationic lipid DNA complexes formed under optimized conditions. J Gene Med 2003; 5: 712–722.
Kim SH, Song IH, Kim JC, Kim EJ, Jang DO, Park YS . In vitro and in vivo gene-transferring characteristics of novel cationic lipids, DMKD (O,O′-dimyristyl-N-lysyl aspartate) and DMKE (O,O′-dimyristyl-N-lysyl glutamate). J Control Rel 2005; 115: 234–241.
Acknowledgements
This work was funded by the Korea Research Foundation, 2006.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H., Kim, J., Lee, Y. et al. An efficient liposomal gene delivery vehicle using Sendai F/HN proteins and protamine. Cancer Gene Ther 15, 214–224 (2008). https://doi.org/10.1038/sj.cgt.7701121
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701121
Keywords
This article is cited by
-
Hyaluronic acid pretreatment for Sendai virus-mediated cochlear gene transfer
Gene Therapy (2016)