Abstract
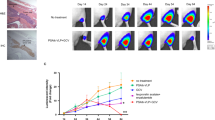
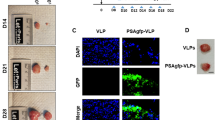
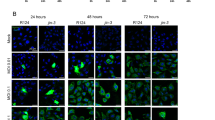
Conditionally replicating adenoviruses are developing as a complement to traditional cancer therapies. Ad[I/PPT-E1A] is an E1B/E3-deleted virus that replicates exclusively in prostate cells, since the expression of E1A is controlled by the recombinant 1.4 kb prostate-specific PPT promoter. The transcriptional integrity of PPT is maintained by the 3.0 kb mouse H19 insulator that was introduced directly upstream of the PPT sequence. In order to increase the cloning capacity to be able to reintroduce E3 sequences in the 35.7 kb Ad[I/PPT-E1A] genome, various shorter insulators were examined in a luciferase reporter gene assay. It was found that the 1.6 kb core H19 insulator (i) improves the activity of PPT, compared to the 3.0 kb full-length insulator, while still maintaining prostate cell specificity and releasing 1.4 kb of space for insertion of additional sequences. To improve the ability of the virus to efficiently lyse infected cells and persist in vivo, we inserted the adenovirus death protein (ADP) or the full-length adenovirus E3 region. The oncolytic activity of PPT-E1A-based viruses was studied using MTS, crystal violet and replication assays. The virus with the reintroduced full-length E3-region (Ad[i/PPT-E1A, E3]) showed the highest cytopathic effects in vitro. Furthermore, this virus suppressed the growth of aggressively growing prostate tumors in vivo. Therefore, we conclude that Ad[i/PPT-E1A, E3] is a prostate-specific oncolytic adenovirus with a high potential for treating localized prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gronberg H . Prostate cancer epidemiology. Lancet 2003; 361: 859–864.
Nelson WG, De Marzo AM, Isaacs WB . Prostate cancer. N Engl J Med 2003; 349: 366–381.
Kanerva A, Hemminki A . Modified adenoviruses for cancer gene therapy. Int J Cancer 2004; 110: 475–480.
Essand M . Gene therapy and immunotherapy of prostate cancer: adenoviral-based strategies. Acta Oncol 2005; 44: 610–627.
Russell WC . Update on adenovirus and its vectors. J Gen Virol 2000; 81 (Part 11): 2573–2604.
Cheng WS, Kraaij R, Nilsson B, van der Weel L, de Ridder CM, Tötterman TH et al. A novel TARP-promoter-based adenovirus against hormone-dependent and hormone-refractory prostate cancer. Mol Ther 2004; 10: 355–364.
Cheng WS, Dzojic H, Nilsson B, Totterman TH, Essand M . An oncolytic conditionally replicating adenovirus for hormone-dependent and hormone-independent prostate cancer. Cancer Gene Ther 2006; 13: 13–20.
Cleutjens KB, van der Korput HA, van Eekelen CC, van Rooij HC, Faber PW, Trapman J . An androgen response element in a far upstream enhancer region is essential for high, androgen-regulated activity of the prostate-specific antigen promoter. Mol Endocrinol 1997; 11: 148–161.
Watt F, Martorana A, Brookes DE, Ho T, Kingsley E, O’Keefe DS et al. A tissue-specific enhancer of the prostate-specific membrane antigen gene, FOLH1. Genomics 2001; 73: 243–254.
Cheng WS, Giandomenico V, Pastan I, Essand M . Characterization of the androgen-regulated prostate-specific T cell receptor gamma-chain alternate reading frame protein (TARP) promoter. Endocrinology 2003; 144: 3433–3440.
Chang SS, Gaudin PB, Reuter VE, Heston WD . Prostate-specific membrane antigen: present and future applications. Urology 2000; 55: 622–629.
Wolffe AP . Transcriptional control: imprinting insulation. Curr Biol 2000; 10: R463–R465.
Ginjala V, Holmgren C, Ulleras E, Kanduri C, Pant V, Lobanenkov V et al. Multiple cis elements within the Igf2/H19 insulator domain organize a distance-dependent silencer. A cautionary note. J Biol Chem 2002; 277: 5707–5710.
Hearing P, Shenk T . The adenovirus type 5 E1A transcriptional control region contains a duplicated enhancer element. Cell 1983; 33: 695–703.
Horwitz MS . Function of adenovirus E3 proteins and their interactions with immunoregulatory cell proteins. J Gene Med 2004; 6: 172–183.
Lichtenstein DL, Toth K, Doronin K, Tollefson AE, Wold WS . Functions and mechanisms of action of the adenovirus E3 proteins. Int Rev Immunol 2004; 23: 75–111.
Tollefson AE, Scaria A, Hermiston TW, Ryerse JS, Wold LJ, Wold WS . The adenovirus death protein (E3-11.6K) is required at very late stages of infection for efficient cell lysis and release of adenovirus from infected cells. J Virol 1996; 70: 2296–2306.
Bett AJ, Prevec L, Graham FL . Packaging capacity and stability of human adenovirus type 5 vectors. J Virol 1993; 67: 5911–5921.
Chung JH, Whiteley M, Felsenfeld G . A 5′ element of the chicken beta-globin domain serves as an insulator in human erythroid cells and protects against position effect in Drosophila. Cell 1993; 74: 505–514.
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Tollefson AE, Scaria A, Saha SK, Wold WS . The 11,600-MW protein encoded by region E3 of adenovirus is expressed early but is greatly amplified at late stages of infection. J Virol 1992; 66: 3633–3642.
Yun CO, Kim E, Koo T, Kim H, Lee YS, Kim JH . ADP-overexpressing adenovirus elicits enhanced cytopathic effect by induction of apoptosis. Cancer Gene Ther 2005; 12: 61–71.
Barton KN, Paielli D, Zhang Y, Koul S, Brown SL, Lu M et al. Second-generation replication-competent oncolytic adenovirus armed with improved suicide genes and ADP gene demonstrates greater efficacy without increased toxicity. Mol Ther 2006; 13: 347–356.
Suzuki K, Alemany R, Yamamoto M, Curiel DT . The presence of the adenovirus E3 region improves the oncolytic potency of conditionally replicative adenoviruses. Clin Cancer Res 2002; 8: 3348–3359.
Yu DC, Chen Y, Seng M, Dilley J, Henderson DR . The addition of adenovirus type 5 region E3 enables calydon virus 787 to eliminate distant prostate tumor xenografts. Cancer Res 1999; 59: 4200–4203.
Toth K, Doronin K, Kuppuswamy M, Ward P, Tollefson AE, Wold WS . Adenovirus immunoregulatory E3 proteins prolong transplants of human cells in immunocompetent mice. Virus Res 2005; 108: 149–159.
Edinger M, Sweeney TJ, Tucker AA, Olomu AB, Negrin RS, Contag CH . Noninvasive assessment of tumor cell proliferation in animal models. Neoplasia 1999; 1: 303–310.
Rehemtulla A, Stegman LD, Cardozo SJ, Gupta S, Hall DE, Contag CH et al. Rapid and quantitative assessment of cancer treatment response using in vivo bioluminescence imaging. Neoplasia 2000; 2: 491–495.
Acknowledgements
This work was supported by funding from the Swedish Cancer Society (grant 4419-B05-06XBC), Swedish Research Council (grant K2005-31X-15270-01A), Knut and Alice Wallenberg Foundation and European Community on behalf of GIANT (grant LSHB-CT-2004-512087). M Essand is a recipient of the Göran Gustafsson Foundation Award.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure
The authors have no conflicting financial interests.
Rights and permissions
About this article
Cite this article
Danielsson, A., Dzojic, H., Nilsson, B. et al. Increased therapeutic efficacy of the prostate-specific oncolytic adenovirus Ad[I/PPT-E1A] by reduction of the insulator size and introduction of the full-length E3 region. Cancer Gene Ther 15, 203–213 (2008). https://doi.org/10.1038/sj.cgt.7701117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701117
Keywords
This article is cited by
-
Immunostimulatory oncolytic virotherapy for multiple myeloma targeting 4-1BB and/or CD40
Cancer Gene Therapy (2020)
-
Activation of myeloid and endothelial cells by CD40L gene therapy supports T-cell expansion and migration into the tumor microenvironment
Gene Therapy (2017)
-
An Infection-enhanced Oncolytic Adenovirus Secreting H. pylori Neutrophil-activating Protein with Therapeutic Effects on Neuroendocrine Tumors
Molecular Therapy (2013)
-
Oncolytic adenovirus modified with somatostatin motifs for selective infection of neuroendocrine tumor cells
Gene Therapy (2011)
-
An ex vivo loop system models the toxicity and efficacy of PEGylated and unmodified adenovirus serotype 5 in whole human blood
Gene Therapy (2010)