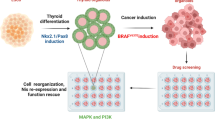
Abstract
Thyroid cancer affects between 10 000 and 15 000 people per year in the US. Typically, this disease can be controlled with surgical resection and radioiodide treatment. However, resistance to these conventional therapies is observed in some patients, who develop intractable anaplastic thyroid cancer (ATC), for which no effective therapies exist. Recently, a sizable fraction of undifferentiated or poorly differentiated thyroid cancers were shown to contain mutations in β-catenin, an oncogenic protein involved in the etiology of cancers of many tissues. We developed a conditionally replicative adenovirus (named ‘HILMI’) which, by virtue of TCF response elements drives E1A and E1B expression, replicates specifically in cells with an active Wnt/β-catenin pathway. We show that several thyroid cancer cell lines, derived from undifferentiated or anaplastic tissues and possessing an active Wnt/β-catenin pathway, are susceptible to cell killing by HILMI. Furthermore, viral replication in ATC cells as xenograft tumors in nude mice was observed, and prolonged survival of mice with ATC tumors was observed following administration of the HILMI therapeutic vector. The results warrant further development of this therapeutic approach for ATC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Nilsson O, Lindeberg J, Zedenius J, Ekman E, Tennvall J, Blomgren H et al. Anaplastic giant cell carcinoma of the thyroid gland: treatment and survival over a 25-year period. World J Surg 1998; 22: 725–730.
Kebebew E, Greenspan FS, Clark OH, Woeber KA, McMillan A . Anaplastic thyroid carcinoma. Treatment outcome and prognostic factors. Cancer 2005; 103: 1330–1335.
McIver B, Hay ID, Giuffrida DF, Dvorak CE, Grant CS, Thompson GB et al. Anaplastic thyroid carcinoma: a 50-year experience at a single institution. Surgery 2001; 130: 1028–1034.
Gilliland FD, Hunt WC, Morris DM, Key CR . Prognostic factors for thyroid carcinoma. A population-based study of 15, 698 cases from the Surveillance, Epidemiology and End Results (SEER) program 1973–1991. Cancer 1997; 79: 564–573.
Hundahl SA, Fleming ID, Fremgen AM, Menck HR . A National Cancer Data Base report on 53, 856 cases of thyroid carcinoma treated in the US, 1985–1995 (see commentn). Cancer 1998; 83: 2638–2648.
Tennvall J, Lundell G, Wahlberg P, Bergenfelz A, Grimelius L, Akerman M et al. Anaplastic thyroid carcinoma: three protocols combining doxorubicin, hyperfractionated radiotherapy and surgery. Br J Cancer 2002; 86: 1848–1853.
Veness MJ, Porter GS, Morgan GJ . Anaplastic thyroid carcinoma: dismal outcome despite current treatment approach. ANZ J Surg 2004; 74: 559–562.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science 1996; 274: 373–376.
Fueyo J, Gomez-Manzano C, Alemany R, Lee PS, McDonnell TJ, Mitlianga P et al. A mutant oncolytic adenovirus targeting the Rb pathway produces anti-glioma effect in vivo. Oncogene 2000; 19: 2–12.
Liu TC, Hallden G, Wang Y, Brooks G, Francis J, Lemoine N et al. An E1B-19 kDa gene deletion mutant adenovirus demonstrates tumor necrosis factor-enhanced cancer selectivity and enhanced oncolytic potency. Mol Ther 2004; 9: 786–803.
Heise C, Hermiston T, Johnson L, Brooks G, Sampson-Johannes A, Williams A et al. An adenovirus E1A mutant that demonstrates potent and selective systemic anti-tumoral efficacy. Nat Med 2000; 6: 1134–1139.
Hallenbeck PL, Chang YN, Hay C, Golightly D, Stewart D, Lin J et al. A novel tumor-specific replication-restricted adenoviral vector for gene therapy of hepatocellular carcinoma. Hum Gene Ther 1999; 10: 1721–1733.
Hernandez-Alcoceba R, Pihalja M, Wicha MS, Clarke MF . A novel, conditionally replicative adenovirus for the treatment of breast cancer that allows controlled replication of E1a-deleted adenoviral vectors. Hum Gene Ther 2000; 11: 2009–2024.
Rodriguez R, Schuur ER, Lim HY, Henderson GA, Simons JW, Henderson DR . Prostate attenuated replication competent adenovirus (ARCA) CN706: a selective cytotoxic for prostate-specific antigen-positive prostate cancer cells. Cancer Res 1997; 57: 2559–2563.
Khuri FR, Nemunaitis J, Ganly I, Arseneau J, Tannock IF, Romel L et al. A controlled trial of intratumoral ONYX-015, a selectively-replicating adenovirus, in combination with cisplatin and 5-fluorouracil in patients with recurrent head and neck cancer. Nat Med 2000; 6: 879–885.
DeWeese TL, van der Poel H, Li S, Mikhak B, Drew R, Goemann M et al. A phase I trial of CV706, a replication-competent, PSA selective oncolytic adenovirus, for the treatment of locally recurrent prostate cancer following radiation therapy. Cancer Res 2001; 61: 7464–7472.
Morin PJ . Beta-catenin signaling and cancer. Bioessays 1999; 21: 1021–1030.
Jamieson CHM, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL et al. Granulocyte-Macrophage Progenitors as Candidate Leukemic Stem Cells in Blast-Crisis CML. N Engl J Med 2004; 351: 657–667.
Hrafnkelsson J, Tulinius H, Kjeld M, Sigvaldason H, Jonasson JG . Serum thyroglobulin as a risk factor for thyroid carcinoma. Acta Oncol 2000; 39: 973–977.
Wu R, Zhai Y, Fearon ER, Cho KR . Diverse mechanisms of beta-catenin deregulation in ovarian endometrioid adenocarcinomas. Cancer Res 2001; 61: 8247–8255.
Polakis P . Wnt signaling and cancer. Genes Dev 2000; 14: 1837–1851.
Polakis P . More than one way to skin a catenin. Cell 2001; 105: 563–566.
Satoh S, Daigo Y, Furukawa Y, Kato T, Miwa N, Nishiwaki T et al. AXIN1 mutations in hepatocellular carcinomas, and growth suppression in cancer cells by virus-mediated transfer of AXIN1. Nat Genet 2000; 24: 245–250.
Korinek V, Barker N, Morin PJ, van Wichen D, de Weger R, Kinzler KW et al. Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC−/− colon carcinoma. Science 1997; 275: 1784–1787.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B et al. Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science 1997; 275: 1787–1790.
Rubinfeld B, Robbins P, El-Gamil M, Albert I, Porfiri E, Polakis P . Stabilization of beta-catenin by genetic defects in melanoma cell lines. Science 1997; 275: 1790–1792.
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT et al. Identification of c-MYC as a target of the APC pathway. Science 1998; 281: 1509–1512.
Hussein SM, Duff EK, Sirard C . Smad4 and beta-catenin co-activators functionally interact with lymphoid-enhancing factor to regulate graded expression of Msx2. J Biol Chem 2003; 278: 48805–48814.
Schwartz DR, Wu R, Kardia SL, Levin AM, Huang CC, Shedden KA et al. Novel candidate targets of beta-catenin/T-cell factor signaling identified by gene expression profiling of ovarian endometrioid adenocarcinomas. Cancer Res 2003; 63: 2913–2922.
Tetsu O, McCormick F . Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature 1999; 398: 422–426.
Takahashi M, Tsunoda T, Seiki M, Nakamura Y, Furukawa Y . Identification of membrane-type matrix metalloproteinase-1 as a target of the beta-catenin/Tcf4 complex in human colorectal cancers. Oncogene 2002; 21: 5861–5867.
Takeichi M, Hirano S, Matsuyoshi N, Fujimori T . Cytoplasmic control of cadherin-mediated cell-cell adhesion. Cold Spring Harb Symp Quant Biol 1992; 57: 327–334.
Garcia-Rostan G, Camp RL, Herrero A, Carcangiu ML, Rimm DL, Tallini G . Beta-catenin dysregulation in thyroid neoplasms: down-regulation, aberrant nuclear expression, and CTNNB1 exon 3 mutations are markers for aggressive tumor phenotypes and poor prognosis. Am J Pathol 2001; 158: 987–996.
Garcia-Rostan G, Tallini G, Herrero A, D'Aquila TG, Carcangiu ML, Rimm DL . Frequent mutation and nuclear localization of beta-catenin in anaplastic thyroid carcinoma. Cancer Res 1999; 59: 1811–1815.
Ishigaki K, Namba H, Nakashima M, Nakayama T, Mitsutake N, Hayashi T et al. Aberrant localization of beta-catenin correlates with overexpression of its target gene in human papillary thyroid cancer. J Clin Endocrinol Metab 2002; 87: 3433–3440.
Rao AS, Kremenevskaja N, Resch J, Brabant G . Lithium stimulates proliferation in cultured thyrocytes by activating Wnt/beta-catenin signalling. Eur J Endocrinol 2005; 153: 929–938.
Kesmodel S, Prabakaran I, Canter R, Menon C, Molnar-Kimber K, Fraker D . Virus-mediated oncolysis of thyroid cancer by a replication-selective adenovirus driven by a thyroglobulin promoter-enhancer region. J Clin Endocrinol Metab 2005; 90: 3440–3448.
Kobayashi T, Asakawa H, Umeshita K, Takeda T, Maruyama H, Matsuzuka F et al. Treatment of 37 patients with anaplastic carcinoma of the thyroid. Head Neck 1996; 18: 36–41.
Phiel CJ, Zhang F, Huang EY, Guenther MG, Lazar MA, Klein PS . Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. J Biol Chem 2001; 276: 36734–36741.
Shimura H, Suzuki H, Miyazaki A, Furuya F, Ohta K, Haraguchi K et al. Transcriptional activation of the thyroglobulin promoter directing suicide gene expression by thyroid transcription factor-1 in thyroid cancer cells. Cancer Res 2001; 61: 3640–3646.
Kitazono M, Chuman Y, Aikou T, Fojo T . Adenovirus HSV-TK construct with thyroid-specific promoter: enhancement of activity and specificity with histone deacetylase inhibitors and agents modulating the camp pathway. Int J Cancer 2002; 99: 453–459.
Portella G, Scala S, Vitagliano D, Vecchio G, Fusco A . ONYX-015, an E1B gene-defective adenovirus, induces cell death in human anaplastic thyroid carcinoma cell lines. J Clin Endocrinol Metab 2002; 87: 2525–2531.
O'Shea CC, Johnson L, Bagus B, Choi S, Nicholas C, Shen A et al. Late viral RNA export, rather than p53 inactivation, determines ONYX-015 tumor selectivity. Cancer Cell 2004; 6: 611–623.
Abbosh PH, Nephew KP . Multiple signaling pathways converge on β-catenin in thyroid cancer. Thyroid 2005; 11: 555–565.
Portella G, Pacelli R, Libertini S, Cella L, Vecchio G, Salvatore M et al. ONYX-015 enhances radiation-induced death of human anaplastic thyroid carcinoma cells. J Clin Endocrinol Metab 2003; 88: 5027–5032.
Curiel DT . Strategies to adapt adenoviral vectors for targeted delivery. Ann NY Acad Sci 1999; 886: 158–171.
Glasgow JN, Bauerschmitz GJ, Curiel DT, Hemminki A . Transductional and transcriptional targeting of adenovirus for clinical applications. Curr Gene Ther 2004; 4: 1–14.
Acknowledgements
We acknowledge Sue Childress, Mark Braun, and Yanping Zhang for their technical support with microscopy studies. The authors gratefully acknowledge the following agencies for supporting this work: Thyroid Research and Advisory Council (TRAC)/Abbott Laboratories (Grant SYN-1201–07) and National Cancer Institute grant R01 CA-85289. This work is dedicated to Hilmi Abbosh who passed away during the preparation of this manuscript due to anaplastic thyroid cancer.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abbosh, P., Li, X., Li, L. et al. A conditionally replicative, Wnt/β-catenin pathway-based adenovirus therapy for anaplastic thyroid cancer. Cancer Gene Ther 14, 399–408 (2007). https://doi.org/10.1038/sj.cgt.7701024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701024
Keywords
This article is cited by
-
Preclinical efficacy of the oncolytic measles virus expressing the sodium iodide symporter in iodine non-avid anaplastic thyroid cancer: a novel therapeutic agent allowing noninvasive imaging and radioiodine therapy
Cancer Gene Therapy (2012)
-
ONYX-411, a conditionally replicative oncolytic adenovirus, induces cell death in anaplastic thyroid carcinoma cell lines and suppresses the growth of xenograft tumors in nude mice
Cancer Gene Therapy (2008)