Abstract
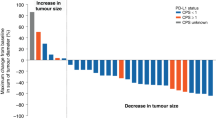
Despite recent improvements in the treatment of metastatic colorectal cancer, few patients are cured and the response rates to second-line treatments are poor. Onyx-015, an oncolytic virus, was administered to patients with metastatic colorectal cancer by hepatic artery infusion. No dose-limiting toxicities were observed in the phase I/II studies. Onyx-015 can kill tumor cells by mechanisms that are distinct from chemotherapeutic agents and may therefore have activity among patients who have failed first-line chemotherapy. The 24 patients included in this analysis had failed first-line therapy with 5-FU/leucovorin, 79% of the patients failed two or more regimens and 58% had failed treatment with Irinotecan. Despite the extensive prior therapy, the median survival of these patients was 10.7 months, 46% were alive at 1 year and two patients (8%) had partial responses. In all, 11 patients (46%) had stable disease at the completion of the four planned viral treatments (3 months). The median survival of this group of patients was 19 months, suggesting that stable disease may be an important predictor of benefit with oncolytic viruses. Eight of the 11 patients with stable disease at 3 months demonstrated a unique radiographic pattern of transient enlargement of tumor masses (10–48%) after the initial infusions of Onyx-015, followed by radiographic evidence of extensive tumor necrosis and regression. The initial enlargement and subsequent tumor necrosis resulted in a prolonged time to achieve objective tumor regression. In addition, the transient enlargement of the tumor masses may have resulted in premature removal of responding patients. Treatment of eight patients was stopped prior to completion of the planned four treatments due to presumed progression as defined by standard radiographic criteria (>25% increase in tumor size). Functional imaging, such as positron emission tomography (PET) scans, may help distinguish clinical responses from progressive disease following treatment with oncolytic viruses. Onyx-015 may benefit patients with refractory colorectal cancer and additional studies that include PET scans to assess clinical response are warranted.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Greenlee RT, Murray T, Bolden S, Wingo PA . Cancer statistics, 2000. CA Cancer J Clin. 2000;50:7–33.
Rothenberg ML . Efficacy of oxaliplatin in the treatment of colorectal cancer. Oncology (Huntingt). 2000;14:9–14.
Rothenberg ML, Oza AM, Bigelow RH, et al. Superiority of oxaliplatin and fluorouracil-leucovorin compared with either therapy alone in patients with progressive colorectal cancer after irinotecan and fluorouracil-leucovorin: interim results of a phase III trial. J Clin Oncol. 2003;21:2059–2069.
Saltz LB . The role of irinotecan in colorectal cancer. Curr Oncol Rep. 1999;1:155–160.
Zeuli M, Nardoni C, Pino MS, et al. Phase II study of capecitabine and oxaliplatin as first-line treatment in advanced colorectal cancer. Ann Oncol. 2003;14:1378–1382.
Kabbinavar F, Hurwitz HI, Fehrenbacher L, et al. Phase II, randomized trial comparing bevacizumab plus fluorouracil (FU)/leucovorin (LV) with FU/LV alone in patients with metastatic colorectal cancer. J Clin Oncol. 2003;21:60–65.
Lavery IC, Lopez-Kostner F, Pelley RJ, Fine RM . Treatment of colon and rectal cancer. Surg Clin North Am. 2000;80:535–569.
Saltz LB, Meropol NJ, Loehrer Sr PJ, Needle MN, Kopit J, Mayer RJ . Phase II trial of cetuximab in patients with refractory colorectal cancer that expresses the epidermal growth factor receptor. J Clin Oncol. 2004;22:1201–1208.
Motzer RJ, Amato R, Todd M, et al. Phase II trial of antiepidermal growth factor receptor antibody C225 in patients with advanced renal cell carcinoma. Invest New Drugs. 2003;21:99–101.
Rougier P, Van Cutsem E, Bajetta E, et al. Randomised trial of irinotecan versus fluorouracil by continuous infusion after fluorouracil failure in patients with metastatic colorectal cancer. Lancet. 1998;352:1407–1412.
Rougier P, Bugat R, Douillard JY, et al. Phase II study of irinotecan in the treatment of advanced colorectal cancer in chemotherapy-naive patients and patients pretreated with fluorouracil-based chemotherapy. J Clin Oncol. 1997;15:251–260.
Falcone A, Cianci C, Pfanner E, et al. Continuous-infusion 5-fluorouracil in metastatic colorectal cancer patients pretreated with bolus 5-fluorouracil: clinical evidence of incomplete cross-resistance. Ann Oncol. 1994;5:291.
Falcone A, Allegrini G, Lencioni M, et al. Protracted continuous infusion of 5-fluorouracil and low-dose leucovorin in patients with metastatic colorectal cancer resistant to 5-fluorouracil bolus-based chemotherapy: a phase II study. Cancer Chemother Pharmacol. 1999;44:159–163.
Mori A, Bertoglio S, Guglielmi A, et al. Activity of continuous-infusion 5-fluorouracil in patients with advanced colorectal cancer clinically resistant to bolus 5-fluorouracil. Cancer Chemother Pharmacol. 1993;33:179–180.
Cunningham D, Glimelius B . A phase III study of irinotecan (CPT-11) versus best supportive care in patients with metastatic colorectal cancer who have failed 5-fluorouracil therapy. V301 Study Group. Semin Oncol. 1999;26:6–12.
Sadahiro S, Suzuki T, Ishikawa K, et al. Recurrence patterns after curative resection of colorectal cancer in patients followed for a minimum of ten years. Hepatogastroenterology. 2003;50:1362–1366.
Altendorf-Hofmann A, Scheele J . A critical review of the major indicators of prognosis after resection of hepatic metastases from colorectal carcinoma. Surg Oncol Clin N Am. 2003;12:165–192.
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH . Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;230:309–318 discussion 318–321.
Kemeny N, Huang Y, Cohen AM, et al. Hepatic arterial infusion of chemotherapy after resection of hepatic metastases from colorectal cancer. N Engl J Med. 1999;341:2039–2048.
Kemeny N, Jarnagin W, Gonen M, et al. Phase I/II study of hepatic arterial therapy with floxuridine and dexamethasone in combination with intravenous irinotecan as adjuvant treatment after resection of hepatic metastases from colorectal cancer. J Clin Oncol. 2003;21:3303–3309.
Cohen AD, Kemeny NE . An update on hepatic arterial infusion chemotherapy for colorectal cancer. Oncologist. 2003;8:553–566.
Reid T, Galanis E, Abbruzzese J, et al. Hepatic arterial infusion of a replication-selective oncolytic adenovirus (dl1520): phase II viral, immunologic, and clinical endpoints. Cancer Res. 2002;62:6070–6079.
Reid T, Galanis E, Abbruzzese J, et al. Intra-arterial administration of a replication-selective adenovirus (dl1520) in patients with colorectal carcinoma metastatic to the liver: a phase I trial. Gene Therapy. 2001;8:1618–1626.
Heise C, Sampson-Johannes A, Williams A, McCormick F, Von Hoff DD, Kirn DH . ONYX-015, an E1B gene-attenuated adenovirus, causes tumor-specific cytolysis and antitumoral efficacy that can be augmented by standard chemotherapeutic agents. Nat Med. 1997;3:639–645.
McCormick F . ONYX-015 selectivity and the p14ARF pathway. Oncogene. 2000;19:6670–6672.
McCormick F . Cancer-specific viruses and the development of ONYX-015. Cancer Biol Ther. 2003;2:S157–S160.
Ries SJ, Brandts CH, Chung AS, et al. Loss of p14ARF in tumor cells facilitates replication of the adenovirus mutant dl1520 (ONYX-015). Nat Med. 2000;6:1128–1133.
Harada JN, Berk AJ . p53-Independent and -dependent requirements for E1B-55K in adenovirus type 5 replication. J Virol. 1999;73:5333–5344.
Martin ME, Berk AJ . Adenovirus E1B 55K represses p53 activation in vitro. J Virol. 1998;72:3146–3154.
Yew PR, Berk AJ . Inhibition of p53 transactivation required for transformation by adenovirus early 1B protein. Nature. 1992;357:82–85.
Kao CC, Yew PR, Berk AJ . Domains required for in vitro association between the cellular p53 and the adenovirus 2 E1B 55K proteins. Virology. 1990;179:806–814.
Yew PR, Kao CC, Berk AJ . Dissection of functional domains in the adenovirus 2 early 1B 55K polypeptide by suppressor-linker insertional mutagenesis. Virology. 1990;179:795–805.
Sze DY, Freeman SM, Slonim SM, et al. Dr Gary J Becker Young Investigator Award: intraarterial adenovirus for metastatic gastrointestinal cancer: activity, radiographic response, and survival. J Vasc Interv Radiol. 2003;14:279–290.
Reid TR, Sze DY . Developments in medical oncology and their implications for interventional radiology. Tech Vasc Interv Radiol. 2002;5:177–181.
Whiteford MH, Whiteford HM, Yee LF, et al. Usefulness of FDG-PET scan in the assessment of suspected metastatic or recurrent adenocarcinoma of the colon and rectum. Dis Colon Rectum. 2000;43:759–767 discussion 767–770.
Spaepen K, Stroobants S, Dupont P, et al. Early restaging positron emission tomography with (18)F-fluorodeoxyglucose predicts outcome in patients with aggressive non-Hodgkin's lymphoma. Ann Oncol. 2002;13:1356–1363.
Cerfolio RJ, Ojha B, Mukherjee S, Pask AH, Bass CS, Kotholi CR . Positron emission tomography scanning with 2-fluoro-2-deoxy-d-glucose as a predictor of response of neoadjuvant treatment for non-small cell carcinoma. J Thorac Cardiovasc Surg. 2003;125:938–944.
Libutti SK, Alexander Jr HR, Choyke P, et al. A prospective study of 2-[18F] fluoro-2-deoxy-D-glucose/positron emission tomography scan, 99mTc-labeled arcitumomab (CEA-scan), and blind second-look laparotomy for detecting colon cancer recurrence in patients with increasing carcinoembryonic antigen levels. Ann Surg Oncol. 2001;8:779–786.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reid, T., Freeman, S., Post, L. et al. Effects of Onyx-015 among metastatic colorectal cancer patients that have failed prior treatment with 5-FU/leucovorin. Cancer Gene Ther 12, 673–681 (2005). https://doi.org/10.1038/sj.cgt.7700819
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700819
Keywords
This article is cited by
-
Biodistribution Analysis of Oncolytic Adenoviruses in Patient Autopsy Samples Reveals Vascular Transduction of Noninjected Tumors and Tissues
Molecular Therapy (2015)
-
REO-001: A phase I trial of percutaneous intralesional administration of reovirus type 3 dearing (Reolysin®) in patients with advanced solid tumors
Investigational New Drugs (2013)
-
Ad3-hTERT-E1A, a Fully Serotype 3 Oncolytic Adenovirus, in Patients With Chemotherapy Refractory Cancer
Molecular Therapy (2012)
-
Deletion analysis of Ad5 E1a transcriptional control region: impact on tumor-selective expression of E1a and E1b
Cancer Gene Therapy (2011)
-
Adenoviruses with an αvβ integrin targeting moiety in the fiber shaft or the HI-loop increase tumor specificity without compromising antitumor efficacy in magnetic resonance imaging of colorectal cancer metastases
Journal of Translational Medicine (2010)