Abstract
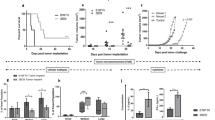
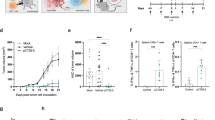
A mouse glioblastoma cell line, termed GL261, was shown to express high levels of proteins involved in melanin biosynthesis such as the tyrosinase-related protein-2 (TRP-2), which is commonly overexpressed in melanoma cells. Mice injected with GL261 cells developed a CD8+ T-cell response to TRP-2 and a DNA vaccine expressing human (h)TRP-2 induced CD8+ T cells that recognized TRP-2 expressed by GL261 cells indicating that this melanoma-associated antigen may be suited for active immunotherapy of glioblastoma. Mice vaccinated with a DNA vaccine expressing TRP-2 were partially protected against subcutaneous, intravenous, or intracerebral challenge with the glioblastoma cells. Vaccine-induced protection against intracerebral challenge required both CD4+ and CD8+ T cells. Vaccine efficacy was enhanced upon addition of IL-12 as a genetic adjuvant. These results indicate that this well-defined melanoma-associated antigen can induce an adaptive immune response, which limits the intracerebral progression of a glioblastoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Mahaley MS, Mettlin C, Natarajan N, et al. National survey of patterns of care for brain-tumor patients. J Neurosurg. 1989;71:826–836.
Soiffer R, Lynch T, Mihm M, et al. Vaccination with irradiated autologous melanoma cells engineered to secrete human granulocyte–macrophage colony-stimulating factor generates potent antitumor immunity in patients with metastatic melanoma. Proc Natl Acad Sci USA. 1998;95:13141–13146.
Gilboa E, Nair SK, Lyerly HK . Immunotherapy of cancer with dendritic-cell-based vaccines. Cancer Immunol Immunother. 1998;46:82–87.
Tuting T, Gambotto A, DeLeo A, et al. Induction of tumor antigen-specific immunity using plasmid DNA immunization in mice. Cancer Gene Ther. 1999;6:73–80.
Hawkins WG, Gold JS, Blachere NE, et al. Xenogeneic DNA immunization in melanoma models for minimal residual disease. J Surg Res. 2002;102:137–143.
Bronte V, Apolloni E, Ronca R, et al. Genetic vaccination with “self” tyrosinase-related protein 2 causes melanoma eradication but not vitiligo. Cancer Res. 2000;60:253–258.
Mendiratta SK, Thai G, Eslahi, NK, et al. Therapeutic tumor immunity induced by polyimmunization with melanoma antigens gp100 and TRP-2. Cancer Res. 2001;61:859–863.
Tanaka M, Kaneda Y, Fujii S, et al. Induction of a systemic immune response by a polyvalent melanoma-associated antigen DNA vaccine for prevention and treatment of malignant melanoma. Mol Ther. 2002;5:291–299.
O I, Ku G, Ertl HC, et al. A dendritic cell vaccine induces protective immunity to intracranial growth of glioma. Anticancer Res. 2002;22:613–621.
Kowalczyk D, Wlazlo AP, Giles-Davis W, et al. Staining of antigen activated lymphocytes (SAAL): a highly specific method for amplification of tumor-specific CD8+ T cells. J Immunol Methods 2000;241:131–139.
Kleinman HK, McGarvey ML, Liotta LA, et al. Isolation and characterization of type IV procollagen, laminin and heparan sulfate proteoglycan from the EHS sarcoma. Biochemistry. 1982;21: 6188–6193.
Parkhurst MR, Fitzgerald EB, Southwood S, et al. Identification of a shared HLA-A*0201-restricted T-cell epitope from the melanoma antigen tyrosinase-related protein 2 (TRP2). Cancer Res. 1998;58:4895–4901.
Yokoyama K, Suzuki H, Yasumoto K, et al. Molecular cloning and functional analysis of a cDNA coding for human DOPAchrome tautomerase/tyrosinase-related protein-2. Biochem Biophys Acta. 1994;1217:317–321.
He Z, Wlazlo AP, Kowalczyk DW, et al. Viral recombinant vaccines to the E6 and E7 antigens of HPV-16. Virology. 2000;270:146–161.
Overwijk WW, Restifo NP . Autoimmunity and the immunotherapy of cancer: targeting the “self” to destroy the “other”. Crit Rev Immunol. 2000;20:433–450.
Steitz J, Bruck J, Steinbrink K, et al. Genetic immunization of mice with human tyrosinase-related protein 2: implications for the immunotherapy of melanoma. Int J Cancer. 2000;86:89–94.
Overwijk WW, Lee DS, Surman DR, et al. Vaccination with a recombinant vaccinia virus encoding a “self” antigen induces autoimmune vitiligo and tumor cell destruction in mice: requirement for CD4(+) T lymphocytes. Proc Acad Natl Sci USA. 1999;96:2982–2987.
Pertmer TM, Oran AE, Madorin CA, et al. Th1 genetic adjuvants modulate immune responses in neonates. Vaccine. 2001;19:1764–1771.
Deng H, Kowalczyk D, O I, et al. A modified DNA vaccine to p53 induces protective immunity to challenge with a chemically induced sarcoma cell line. Cell Immunol. 2002;215:20–31.
Pisetsky DS . Mechanisms of immune stimulation by bacterial DNA. Springer Sem Immunopathol 2000;22:21–33.
Krieg AM . CpG motifs in bacterial DNA and their immune effects. Annu Rev Immunol. 2002;20:709–760.
Tuting T, Gambotto A, DeLeo A, et al. Induction of tumor antigen-specific immunity using plasmid DNA immunization in mice. Cancer Gene Ther. 1999;6:73–80.
Le TP, Coonan KM, Hedstrom RC, et al. Safety, tolerability and humoral immune responses after intramuscular administration of a malaria DNA vaccine to healthy adult volunteers. Vaccine. 2000;18:1893–1901.
Wang R, Doolan, DL, Le TP, et al. Induction of antigen-specific cytotoxic T lymphocytes in humans by a malaria DNA vaccine. Science. 1998;282:476–480.
MacGregor RR, Boyer JD, Ugen KE, et al. First human trial of a DNA-based vaccine for treatment of human immunodeficiency virus type 1 infection: safety and host response. J Infect Dis. 1998;178:92–100.
Ulmer JB, Donnelly JJ, Parker SE, et al. Heterologous protection against influenza by injection of DNA encoding a viral protein. Science. 1993;259:1745–1749.
Xiang ZQ, Ertl HCJ . Manipulation of the immune response to a plasmid-encoded viral antigen by coinoculation with plasmid expressing cytokines. Immunity. 1995;2:129–136.
Barouch DH, Craiu A, Santra S, et al. Elicitation of high-frequency cytotoxic T-lymphocyte responses against both dominant and subdominant simian–human immunodeficiency virus epitopes by DNA vaccination of rhesus monkeys. J Virol. 2001;75:2462–2467.
Xiang ZQ, Pasquini S, Ertl HCJ . Induction of genital immunity by DNA priming and intranasal booster immunization with a replication-defective adenoviral recombinant. J Immunol. 1999;162:6716–6723.
Huang SK, Okamoto T, Morton DL, et al. Antibody responses in melanoma/melanocytes autoantigens in melanoma patients. J Int Dermatol. 1998;111:662–667.
Reynolds SR, Celis E, Sette A, et al. HLA-independent heterogeneity of CD8+ T cell responses to MAGE-3, Melan-A/MART-1, gp100, tyrosinase, MC1R and TRP-2 in vaccine-treated melanoma patients. J Immunol. 1998;161:6970–6976.
Sgadari C, Angiolillo AL, Tosato G . Inhibition of angiogenesis by interleukin-12 is mediated by the interferon-inducible protein 10. Blood. 1996;87:3877–3882.
Rubin LL, Staddon JM . The cell biology of the blood–brain barrier. Annu Rev Neurosci. 1999;2:11–28.
Joly E, Mucke L, Oldstone M . Viral persistence in neurons explained by lack of major histocompatibility class I expression. Science. 1991;253:1283–1285.
Frei K, Fontana A . Antigen presentation in the CNS. Mol Psychol. 1997;2:96–98.
De Micco C . Immunology of central nervous system tumors. J Neuroimmunol. 1989;25:93–108.
De Micco C . Immunology of tumors of the central nervous system. Bull Cancer. 1989;76:17–31.
Saas P, Walker PR, Hahne M, et al. Fas ligand expression by astrocytoma in vivo: maintaining immune privilege in the brain? J Clin Invest. 1997;99:1173–1178.
Weller M, Fontana A . The failure of current immunotherapy for malignant glioma. Tumor-derived TGF-beta, T-cell apoptosis, and the immune privilege of the brain. Brain Res Rev. 1995;21:128–151.
Fontana A, Bodmer S, Frei K, et al. Expression of TGF-beta 2 in human glioblastoma: a role in resistance to immune rejection? Ciba Found Symp. 1991;157:232–241.
Weller M, Constam DB, Malipiero U, et al. Transforming growth factor-beta 2 induces apoptosis of murine T cell clones without down-regulating bcl-2 mRNA expression. Eur J Immunol. 1994;24:1293–1300.
Zou JP, Morford LA, Chougnet C, et al. Human glioma-induced immunosuppression involves soluble factor(s) that alters monocyte cytokine profile and surface markers. J Immunol. 1999;162:4882–4892.
Pazmany T, Kosa JP, Tomasi TB, et al. Effect of transforming growth factor-beta1 on microglial MHC-class II expression. J Neuroimmunol. 2000;103:122–130.
Aloisi F, De Simone R, Columba-Cabezas S, et al. Opposite effects of interferon-gamma and prostaglandin E2 on tumor necrosis factor and interleukin-10 production in microglia: a regulatory loop controlling microglia pro- and anti-inflammatory activities. J Neurosci Res. 1999;56:571–580.
Janabi N, Hau I, Tardieu M . Negative feedback between prostaglandin and alpha- and beta-chemokine synthesis in human microglial cells and astrocytes. J Immunol. 1999;162:1701–1706.
Hishii M, Nitta T, Ishida H, et al. Human glioma-derived interleukin-10 inhibits antitumor immune responses in vitro. Neurosurgery. 1995;37:1160–1166.
Knolle PA, Uhrig A, Hegenbarth S, et al. IL-10 down-regulates T cell activation by antigen-presenting liver sinusoidal endothelial cells through decreased antigen uptake via the mannose receptor and lowered surface expression of accessory molecules. Clin Exp Immunol. 1998;114:427–433.
Frei K, Lins H, Schwerdel C, et al. Antigen presentation in the central nervous system. The inhibitory effect of IL-10 on MHC class II expression and production of cytokines depends on the inducing signals and the type of cell analyzed. J Immunol. 1994;152:2720–2728.
Allegretta M, Albertini RJ, Howell MD, et al. Homologies between T cell receptor junctional sequences unique to multiple sclerosis and T cells mediating experimental allergic encephalomyelitis. J Clin Invest. 1994;94:105–109.
Soderstrom M, Link H, Sun JB, et al. Autoimmune T cell repertoire in optic neuritis and multiple sclerosis: T cells recognizing multiple myelin proteins are accumulated in cerebrospinal fluid. J Neurol Neurosurg Psychiatry. 1994;55:544–551.
Kalman B, Knobler RL, Lublin FD . Preferential but not exclusive T cell receptor V beta chain utilization of myelin basic protein and peptide-specific T cell clones in mice. Cell Immunol. 1994;153:206–213.
Dranoff G, Jaffee E, Lazenby A, et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte–macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA. 1993;90:3539–3543.
Wakimoto H, Yoshida Y, Aoyagi M, et al. Efficient retrovirus-mediated cytokine-gene transduction of primary-cultured human glioma cells for tumor vaccination therapy. J Cancer Res. 1997;88:296–305.
Acknowledgements
We would like to thank Ms W Giles-Davis for technical assistance and Ms Cole for preparation of the manuscript. This work was supported by a grant from the Katie's Kids Brain Tumor Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
O, I., Blaszczyk-Thurin, M., Shen, C. et al. A DNA vaccine expressing tyrosinase-related protein-2 induces T-cell-mediated protection against mouse glioblastoma. Cancer Gene Ther 10, 678–688 (2003). https://doi.org/10.1038/sj.cgt.7700620
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700620
Keywords
This article is cited by
-
Surface display of glycosylated Tyrosinase related protein-2 (TRP-2) tumour antigen on Lactococcus lactis
BMC Biotechnology (2015)
-
Incomplete tumour control following DNA vaccination against rat gliomas expressing a model antigen
Acta Neurochirurgica (2013)
-
Downregulation of KIF23 suppresses glioma proliferation
Journal of Neuro-Oncology (2012)
-
In vivo bioluminescence imaging in an experimental mouse model for dendritic cell based immunotherapy against malignant glioma
Journal of Neuro-Oncology (2009)
-
DNA vaccines for cancer too
Cancer Immunology, Immunotherapy (2006)