Abstract
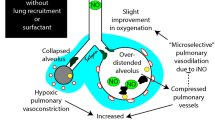
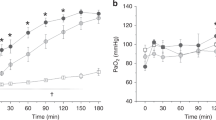
Many different surfactant preparations derived from animal sources, as well as synthetic surfactants, are available for the treatment of preterm infants with respiratory distress syndrome (RDS). Natural, modified surfactants containing surfactant-associated proteins appear to be more effective than non-protein-containing synthetic surfactants. Comparative trials with poractant alfa at a higher initial dose of 200 mg/kg appear to be associated with rapid weaning of FiO2, less need for additional doses, and decreased mortality in infants <32 weeks gestation when compared with beractant. Early rescue (<30 min of age) surfactant therapy is an effective method to minimize over treatment of some preterm infants who may not develop RDS. Surfactant therapy followed by rapid extubation to nasal ventilation appears to be more beneficial than continued mechanical ventilation. In near-term or term newborns with acute RDS, surfactant therapy has been shown to be 70% effective in improving respiratory failure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Martin JA, Hamilton BE, Sutton PD, Menacker F, Munson ML Births: Final Data for 2002. National Center for Health Statistics. Vital Health Stat Series vol. 52, no. 10, 2003.
Lemons JA, Bauer CR, Oh W, Korones S, Papile LA, Stoll BJ et al. Very low-birth weight infant (VLBW) outcomes of the NICHD neonatal research network. Pediatrics 2001; 107: 1–8.
Crowley PA . Antenatal corticosteroid therapy: a meta-analysis of the randomized trials. Am J Obstet Gynecol 1995; 173: 322–335.
NIH Consensus Development Panel. Effect of corticosteroids for fetal maturation on perinatal outcomes. JAMA 1995; 273: 413–418.
Soll RF . Synthetic surfactant for respiratory distress syndrome in preterm infants (Cochrane Review). In: The Cochrane Library, Issue 4, 2004. John Wiley & Sons, Ltd: Chichester, UK.
Soll RF, Blanco F . Natural surfactant extract versus synthetic surfactant for neonatal respiratory distress syndrome. (Cochrane Review). In: The Cochrane Library, Issue 4, 2004. John Wiley & Sons, Ltd: Chichester, UK.
Horbar JD, Wright LL, Soll RF, Wright EC, Fanaroff AA, Korones SB et al. A multicenter randomized trial comparing two surfactants for the treatment of neonatal respiratory distress syndrome. J Pediatr 1993; 123: 757–766.
Sehgal SS, Ewing CK, Richards T, Taeusch W . Modified bovine surfactant (survanta) versus a protein free surfactant (exosurf) in the treatment of respiratory distress syndrome in preterm infants: a pilot study. J Natl Med Assoc 1994; 86: 46–52.
The Vermont Oxford Neonatal Network. A multicenter randomized trial comparing synthetic surfactant with modified bovine surfactant extract in the treatment of neonatal respiratory distress syndrome. Pediatrics 1996; 97: 1–6.
Hudak ML, Farrell EE, Rosenberg AA, Jung AL, Auten RL, Durand DJ et al. A multicenter randomized masked comparison trial of natural versus synthetic surfactant for the treatment of respiratory distress syndrome. J Pediatr 1996; 128: 396–406.
Hudak ML, Martin DJ, Egan EA, Matteson EJ, Cummings J, Jung AL et al. A multicenter randomized masked comparison trial of synthetic surfactant versus calf lung surfactant extract in the prevention of neonatal respiratory distress syndrome. Pediatrics 1997; 100: 39–50.
Rollins M, Jenkins J, Tubman R, Corkey C, Wilson D . Comparison of clinical responses to natural and synthetic surfactants. J Perinat Med 1993; 21: 341–347.
Alvarado M, Hingre R, Hakason D, Gross S . Clinical trial of survanta versus exosurf in infants <1500 g with respiratory distress syndrome. Pediatr Res 1993; 33: 314A.
Pearlman SA, Leef KH, Stefano JL, Spear ML, Esterly KL . A randomized trial comparing exosurf versus survanta in the treatment of neonatal RDS. Pediatr Res 1993; 33: 340A.
Modanlou HD, Beharry K, Padilla G, Norris K, Safvati S, Aranda JV . Comparative efficacy of exosurf and survanta surfactants on early clinical course of respiratory distress syndrome and complications of prematurity. J Perinatol 1997; 17: 455–460.
da Costa DE, Pai MG, Al Khusabiby SM . Comparative trial of artificial and natural surfactants in the treatment of respiratory distress syndrome of prematurity: experience in a developing country. Pediatr Pulmonol 1999; 27: 303–304.
Kukkonen AK, Virtanen M, Jarvenpaa AL, Pokela ML, Ikonen S, Fellman V . Randomized trial comparing natural and synthetic surfactant: increased infection rate after natural surfactant? Acta Pediatr 2000; 89: 556–561.
Ainsworth SB, Beresford MW, Milligan DWA, Shaw NJ, Matthews JNS, Fenton AC et al. Pumactant and poractant alfa for treatment of respiratory distress syndrome in neonates born at 25–29 weeks’ gestation: a randomised trial. Lancet 2000; 355: 1387–1392.
Sinha SK, Lacaze-Masmonteil T, Soler A, Wiswell TE, Gadzinowski J, Hajdu J et al. A multicenter, randomized, controlled trial of lucinactant versus poractant alfa among very premature infants at high risk for respiratory distress syndrome. Pediatrics 2005; 115: 1030–1038.
Moya FR, Gadzinowski J, Bancalari E, Salinas V, Kopelman B, Bancalari A et al. A multicenter, randomized, masked, comparison trial of lucinactant, colfosceril palmitate, and beractant for the prevention of respiratory distress syndrome among very preterm infants. Pediatrics 2005; 115: 1018–1029.
Notter RH . Lung biology in health and disease. Lung Surfactants: Basic Science and Clinical Applications, vol 149. Marcel Dekker Inc.: New York, 2000. pp 281–298.
Bloom BT, Kattwinkel J, Hall RT, Delmore PM, Egan EA, Trout JR et al. Comparison of infasurf (calf lung surfactant extract) to survanta in the treatment and prevention of respiratory distress syndrome. Pediatrics 1997; 100: 31–38.
Speer CP, Gefeller O, Groneck P, Laufkotter E, Roll C, Hanssler L et al. Randomised clinical trial of two treatment regimens of natural surfactant preparations in neonatal respiratory distress syndrome. Arch Dis Child Fetal Neonatal Ed 1995; 72: F8–F13.
Baroutis G, Kaleyias J, Liarou T, Papathoma E, Hatzistamatiou Z, Costalos C . Comparison of three treatment regimens of natural surfactant preparations in neonatal respiratory distress syndrome. Eur J Pediatr 2003; 162: 476–480.
Ramanathan R, Rasmussen MR, Gerstmann DR, Finer N, Sekar K, North American Study Group. A randomized, multicenter masked comparison trial of poractant alfa (curosurf) versus beractant (survanta) in the treatment of respiratory distress syndrome in preterm infants. Am J Perinatol 2004; 21: 109–119.
Malloy CA, Nicoski P, Muraskas JM . A randomized trial comparing beractant and poractant treatment in neonatal respiratory distress syndrome. Acta Paediatr 2005; 94: 779–784.
Bloom BT, Clark RH . Comparison of infasurf (calfactant) and survanta (beractant) in the prevention and treatment of respiratory distress syndrome. Pediatrics 2005; 116: 392–399.
Marsh W, Smeeding J, York JM, Ramanathan R, Sekar K . A cost minimization comparison of two surfactants, beractant and poractant alfa, based upon prospectively designed, comparative clinical trial data. J Pediatr Pharmacol Ther 2004; 9: 113–121.
Verder H, Robertson B, Greisen G, Ebbesen F, Albertsen P, Lundstrom K et al. Surfcatant therapy and nasal continuous positive airway pressure for newborns with respiratory distress syndrome. N Engl J Med 1994; 331: 1051–1055.
Verder H, Albertsen P, Ebbesen F, Greisen G, Robertson B, Bertelsen A et al. Nasal continuous positive airway pressure and early surfactant therapy for respiratory distress syndrome in newborns of less than 30 weeks’ gestation. Pediatrics 1999; 103: e24.
Haberman B, Shankaran S, Stevenson DK, Papile LA, Stark A, Korones S et al. Does surfactant and immediate extubation to nasal continuous positive airway pressure reduce use of mechanical ventilation? Pediatr Res 2002; 51: 349A.
D’Angio CT, Khalak R, Stevens TP, Reininger A, Reubens L, Kendig JW et al. Intratracheal surfactant administration by transient intubation in infants 29–35 weeks’ gestation with RDS requiring nasal CPAP decreases the likelihood of later mechanical ventilation: a randomized controlled trial. Pediatr Res 2003; 53: 367A.
Soll RF, Conner JM, Howard D, the investigators of the early surfactant replacement study. Early surfactant replacement in spontaneously breathing premature infants with RDS. Pediatr Res 2003; Late breaker abstract 12, PAS 2003 meeting.
Dani C, Bertini G, Pezzati M, Cecchi A, Caviglioli C, Rubaltelli FF . Early extubation and nasal continuous positive airway pressure after surfactant treatment for respiratory distress syndrome among preterm infants <30 weeks’ gestation. Pediatrics 2004; 113: e560–e563.
The Texas Neonatal Research Group. Early surfactant for neonates with mild to moderate respiratory distress syndrome: a multicenter, randomized trial. J Pediatr 2004; 144: 804–808.
Sahni R, Ammari A, Suri MS, Milisavljevic V, Ohira-Kist K, Wung JT et al. Is the new definition of bronchopulmonary dysplasia more useful? J Perinatol 2005; 25: 41–46.
Lewis JF, Brackenbury A . Role of exogenous surfactant in acute lung injury. Crit Care Med 2003; 31: S324–S328.
Herting E, Gefeller O, Land M, Sonderen L, Harms K, Robertson B, Members of the Collaborative European Multicenter Study Group. Surfactant treatment of neonates with respiratory failure and Group B streptoloccal infection. Pediatrics 2000; 106: 957–964.
Findley RD, Taeusch W, Walther FJ . Surfactant replacement therapy for meconium aspiration syndrome. Pediatrics 1996; 97: 48–52.
Lotze A, Mitchell BR, Bulas DI . Multicenter study of surfactant (beractant) use in the treatment of term infants with severe respiratory failure. J Pediatr 1998; 132: 40–47.
Marks SD, Nicholl RM . The reduction in the need for ECMO by using surfactant for meconium aspiration syndrome. J Pediatr 1999; 135: 267–268.
Wiswell TE, Knight GR, Finer NN, Donn SM, Desai H, Walsh WF et al. A multicenter, randomized controlled trial comparing surfaxin (lucinactant) lavage with standard care for treatment of meconium aspiration syndrome. Pediatrics 2002; 109: 1081–1087.
Anderson JM and the Congenital Diaphragmatic Hernia Study Group. Is surfactant therapy beneficial in the management of congenital diaphragmatic hernia? Pediatr Res 2003; Late breaker abstract 13, PAS 2003 meeting.
Luchetti M, Ferraro F, Gallini C, Natale A, Pigna A, Tortorolo L et al. Multicenter, randomized, controlled study of porcine surfactant in severe respiratory syncytial virus-induced respiratory failure. Pediatr Crit Care Med 2002; 3: 261–268.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramanathan, R. Surfactant therapy in preterm infants with respiratory distress syndrome and in near-term or term newborns with acute RDS. J Perinatol 26 (Suppl 1), S51–S56 (2006). https://doi.org/10.1038/sj.jp.7211474
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7211474
Keywords
This article is cited by
-
Poractant alfa in respiratory distress syndrome in preterm infants: a profile of its use
Drugs & Therapy Perspectives (2017)
-
The role of surfactant treatment in preterm infants and term newborns with acute respiratory distress syndrome
Journal of Perinatology (2009)
-
Early surfactant therapy and noninvasive ventilation
Journal of Perinatology (2007)