Abstract
OBJECTIVE:
To determine the effectiveness of a 10-day subcutaneous erythropoietin (rHuEpo) course of 300 units per kg per dose plus oral iron compared to oral iron alone in anemic infants during their convalescent phase of illness.
STUDY DESIGN:
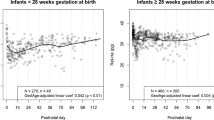
Prospective, randomized trial performed at a 40-bed, teaching, referral, level III, neonatal intensive care unit. Infants with a gestational age at birth of less than 32 weeks, hematocrit of less than or equal to 28% with a corrected reticulocyte count of less than or equal to 5%, postconceptual age of less than 48 weeks or 5 months chronological age, and a diagnosis of anemia of prematurity were considered for inclusion. Major outcome parameters included hematocrit, corrected reticulocyte count and red cell transfusion requirements.
RESULTS:
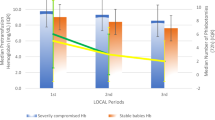
A total of 60 infants were enrolled (n=30 per group). Infants randomized to rHuEpo had a significantly higher post-treatment hematocrit and corrected reticulocyte count than infants in the iron only group (p<0.001). There was a trend towards fewer red cell requirements in the rHuEpo group.
CONCLUSIONS:
The rHuEpo regimen studied here was associated with an acute improvement in hematocrit and corrected reticulocyte counts. This study did not demonstrate a statistically significant decrease in transfusion therapy, in part related to increased subsequent use of rHuEpo in the control group. Taken together, these data demonstrate that this regimen can effectively treat anemia in convalescent premature infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Donato H, Vain N, Rendo P, et al. Effect of early versus late administration of human recombinant erythropoietin on transfusion requirements in premature infants: results of a randomized, placebo-controlled, multicenter trial. Pediatrics 2000;105:1066–1072.
Halperin DS, Felix M, Lacourt G, Babel JF, Wyss M . Recombinant human erythropoietin in the treatment of infants with anaemia of prematurity. Eur J Pediatr 1992;151:661–667.
Shannon KM, Mentzer WC, Abels RI, et al. Recombinant human erythropoietin in the anemia of prematurity: results of a placebocontrolled pilot study. J Pediatr 1991;118:949–955.
Meyer MP, Meyer JH, Commerford A, et al. Recombinant human erythropoietin in the treatment of the anemia of prematurity: results of a double-blind, placebo-controlled study. Pediatrics 1994;93:918–923.
Carnielli V, Montini G, Da Riol R, Dall’Amico, Cantarutti F . Effect of high doses of human recombinant erythropoietin on the need for blood transfusions in preterm infants. J Pediatr 1992;121:98–102.
Maier RF, Obladen M, Kattner E, et al. High-versus low-dose erythropoietin in extremely low birth weight infants. The European multicenter rhEPO study group. J Pediatr 1998;132 (5):866–870.
Ohls RK, Harcum J, Schibler KR, Christensen RD . The effect of erythropoietin on the transfusion requirements of preterm infants weighing 750 grams or less: a randomized, double-blind, placebo-controlled study. J Pediatr 1997;131:661–665.
Garcia MG, Hutson AD, Christensen RD . Effect of recombinant erythropoietin on “late” transfusions in the neonatal intensive care unit: a meta-analysis. J Perinatol 2002;22:108–111.
Pealer LN, Marfin AA, Petersen LR, et al. Transmission of West Nile Virus through blood transfusion in the United States in 2002. N Engl J Med 2003;349 (13):1236–1245.
Dodd RY . Emerging infections, transfusion safety, and epidemiology. N Engl J Med 2003;349 (13):1205–1206.
Ohls RK, Ehrenkranz RA, Wright LL, et al. Effects of early erythropoietin therapy on the transfusion requirements of preterm infants below 1250 grams birth weight: a multicenter, randomized, controlled trial. Pediatrics 2001;108:934–942.
Reiter PD, Rosenberg AA, Valuck RJ . Factors associated with successful epoetin alfa therapy in premature infants. Ann Pharmacother 2000;34 (4):433–439.
Author information
Authors and Affiliations
Additional information
There are no conflicts of interests with any of the authors.
This Research was supported by Grant Number M01 RR000069 General Clinical Research Centers Program National Centers for Research Resources NIH.
Rights and permissions
About this article
Cite this article
Reiter, P., Rosenberg, A., Valuck, R. et al. Effect of Short-Term Erythropoietin Therapy in Anemic Premature Infants. J Perinatol 25, 125–129 (2005). https://doi.org/10.1038/sj.jp.7211220
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7211220
This article is cited by
-
Single-Dose Darbepoetin Administration to Anemic Preterm Neonates
Journal of Perinatology (2005)