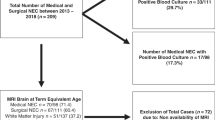
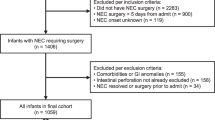
Abstract
BACKGROUND: Prior studies have identified individual risk factors that are associated with necrotizing enterocolitis (NEC); however, the small sample sizes of these previous studies have not allowed the analysis of potential interaction between multiple variables and NEC. Our purpose was to describe the incidence and risk factors for NEC in premature neonates admitted for intensive care.
METHODS: We identified neonates as having NEC if they met accepted diagnostic criterion for necrotizing enterocolitis. Using a national database, we assessed the association between NEC and a battery of risk factors previously reported in peer-reviewed literature.
RESULTS: There were 15,072 neonates that met inclusion criteria; 14,682 did not have NEC, while 390 (2.6%) met criterion for NEC. Multivariate analysis showed that low birth weight was the most important risk factor for NEC. Other factors that were associated with an increased risk of NEC were exposure to antenatal glucocorticoids, vaginal delivery, need for mechanical ventilator support, exposure to both glucocorticoids and indomethacin during the first week of life, absence of an umbilical arterial catheter, and low Apgar score at 5 minutes. Length of hospital stay and mortality were higher in neonates with NEC than in neonates without NEC.
CONCLUSIONS: NEC remains an important cause of morbidity and mortality in prematurely born neonates. In contrast to previous studies, we found that exposure to antenatal glucocorticoids was associated with an increased risk for NEC independent of birth weight.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Holman RC, Stoll BJ, Clarke MJ, Glass RI . The epidemiology of necrotizing enterocolitis infant mortality in the United States. Am J Public Health 1997;87:2026–2031.
Gordon P, Rutledge J, Sawin R, Thomas S, Woodrum D . Early postnatal dexamethasone increases the risk of focal small bowel perforation in extremely low birth weight infants. J Perinatol 1999;19:573–577.
Ghidini A, Espada RA, Spong CY . Does exposure to magnesium sulfate in utero decrease the risk of necrotizing enterocolitis in premature infants? Acta Obstet Gynecol Scand 2001;80:126–129.
Gordon PV, Marshall DD, Stiles AD, Price WA . The clinical, morphologic, and molecular changes in the ileum associated with early postnatal dexamethasone administration: from the baby's bowel to the researcher's bench. Mol Genet Metab 2001;72:91–103.
Bury RG, Tudehope D . Enteral antibiotics for preventing necrotising enterocolitis in low birthweight or preterm infants. Cochrane Database Syst Rev 2000;CD000405.
Chandler JC, Hebra A . Necrotizing enterocolitis in infants with very low birth weight. Semin Pediatr Surg 2000;9:63–72.
De Laet MH, Dassonville M, Johansson A, et al. Small-bowel perforation in very low birth weight neonates treated with high-dose dexamethasone. Eur J Pediatr Surg 2000;10:323–327.
Kennedy KA, Tyson JE, Chamnanvanakij S . Rapid versus slow rate of advancement of feedings for promoting growth and preventing necrotizing enterocolitis in parenterally fed low-birth-weight infants. Cochrane Database Syst Rev 2000;CD001241.
Ledbetter DJ, Juul SE . Erythropoietin and the incidence of necrotizing enterocolitis in infants with very low birth weight. J Pediatr Surg 2000;35:178–181.
Parilla BV, Grobman WA, Holtzman RB, Thomas HA, Dooley SL . Indomethacin tocolysis and risk of necrotizing enterocolitis. Obstet Gynecol 2000;96:120–123.
Hoyos AB . Reduced incidence of necrotizing enterocolitis associated with enteral administration of Lactobacillus acidophilus and Bifidobacterium infantis to neonates in an intensive care unit. Int J Infect Dis 1999;3:197–202.
Neu J, Weiss MD . Necrotizing enterocolitis: pathophysiology and prevention. J Parenter Enteral Nutr 1999;23:S13-S17.
Hung FC, Huang CB, Huang SC, Hsieh CS, Chuang JH . Necrotizing enterocolitis in newborn: nine years' experience. Changgeng Yi Xue Za Zhi 1997;20:29–33.
Uauy RD, Fanaroff AA, Korones SB, Phillips EA, Phillips JB, Wright LL . Necrotizing enterocolitis in very low birth weight infants: ideographic and clinical correlates. National Institute of Child Health and Human Development Neonatal Research Network. J Pediatr 1991;119:630–638.
Clark RH, Auten RL, Peabody J . A comparison of the outcomes of neonates treated with two different natural surfactants. J Pediatr 2001;139:828–831.
Stark AR, Carlo WA, Tyson JE et al. Adverse effects of early dexamethasone in extremely-low-birth-weight infants. National Institute of Child Health and Human Development Neonatal Research Network. N Engl J Med 2001;344:95–101.
Lemons JA, Bauer CR, Oh W et al. Very low birth weight outcomes of the National Institute of Child health and human development neonatal research network, January 1995 through December 1996. NICHD Neonatal Research Network. Pediatrics 2001;107:E1.
Stoll BJ . Epidemiology of necrotizing enterocolitis. Clin Perinatol 1994;21:205–218.
Ballard RA, Ballard PL . Antenatal hormone therapy for improving the outcome of the preterm infant. J Perinatol 1996;16:390–396.
Bauer CR, Morrison JC, Poole WK, et al. A decreased incidence of necrotizing enterocolitis after prenatal glucocorticoid therapy. Pediatrics 1984;73:682–688.
Halac E, Halac J, Begue EF, et al. Prenatal and postnatal corticosteroid therapy to prevent neonatal necrotizing enterocolitis: a controlled trial. J Pediatr 1990;117:132–138.
Smith LM, Qureshi N, Chao CR . Effects of single and multiple courses of antenatal glucocorticoids in preterm newborns less than 30 weeks' gestation. J Matern Fetal Med 2000;9:131–135.
Lawrence D, Brewer D, Hornung R, Mersmann M, Donovan D . Antenatal glucocorticoid use, not perinatal antibiotics, may result in increased risk of necrotizing enterocolitis in very-low-birthweight Infants. Pediatr Res 2001;49:Abstract 1798.
Kamitsuka MD, Horton MK, Williams MA . The incidence of necrotizing enterocolitis after introducing standardized feeding schedules for infants between 1250 and 2500 grams and less than 35 weeks of gestation. Pediatrics 2000;105:379–384.
Gordon PV, Price WA, Stiles AD . Dexamethasone administration to newborn mice alters mucosal and muscular morphology in the ileum and modulates IGF-I localization. Pediatr Res 2001;49:93–100.
Lucas A, Cole TJ . Breast milk and neonatal necrotising enterocolitis. Lancet 1990;336:1519–1523.
Schanler RJ, Shulman RJ, Lau C . Feeding strategies for premature infants: beneficial outcomes of feeding fortified human milk versus preterm formula. Pediatrics 1999;103:1150–1157.
Acknowledgements
In addition to the authors, the following physicians participated in this study: Harrisburg, PA — K. Lorah; Utica, NY — M. Siriwardena; Wichita, KS — E. Otero, C. Smart; Ft. Lauderdale, FL — S. Haskins; Boca Raton, FL — F. Miller; Coral Springs, FL — G. Melnick; Boynton Beach, FL — L. Whetstine; Denver, CO — E. Berman, D. Eichorst, J. Toney, P. Honeyfield; Houston, TX — R. Rivas, H. Pierantoni, E. O'Donnell; Englewood, CO — K. Zarlengo; West Palm Beach, FL — D. Kanter; Virginia Beach, VA — R. Balcom, E. Bollerup; Frederickburg, VA — J. Amin; Spartanburg, SC — V. Iskersky; Watertown, NY — K. Komar; Tarzana, CA — J. Banks; Ventura, CA — J. van Houten; Hoboken, NJ — M. Dyan; Stratford, NJ — J. Coleman; Trenton, NJ — K. Weiss, R. Axelrod; Fountain Valley, CA — A. Esparza; Covina, CA — V. Chundu, G. Martin; San Luis Obispo, CA — J. Martin; Newport Beach, CA — L. Wickham, B. Hannam; Riverside, CA — M. Leitner; Las Vegas, NV — M. Kaneta; Alexandria, VA — M. Dwyer, L. Goldberg; Albuquerque, NM — R. Nederhoff, S. Swetnam; Aurora, CO — M. Brown; Phoenix, AZ — M. McQueen; Dallas, TX — J. Whitfield, T. Brannon; Roanoke, VA — R. Allen; Dayton, OH — N. Kantor; Ogden, UT — N. Harper; Columbia, SC — S. Ellis; Panama City, FL — D. Sprague; Pensacola, FL — J. Nagel; Reno, NV — G. Yup; Tacoma, WA — J. Mulligan, G. Jordan, R. Knudson; Ponce, PR — E. Ochoa; Barrington, IL — F. Uraizee; Fort Worth, TX — R. Sidebottom, D. Turbeville, M. Stanley; Charleston, WV — S. Maxwell; San Juan, PR — A. Rivera, M. Ortega; Austin, TX — J. Courtney, D. Wermer, J. Scharnberg; San Jose, CA — E. Alderete; Rock Hill, SC — A. Payne; South Bend, IN — R. White; Kansas City, MO — B. Heimes, J. Anderson; Huntington, CA — R. Liberman; Elmira, NY — W. Helmuth, J. Felix.
Author information
Authors and Affiliations
Additional information
From The Pediatrix-Obstetrix Center for Research and Education.
Rights and permissions
About this article
Cite this article
Guthrie, S., Gordon, P., Thomas, V. et al. Necrotizing Enterocolitis Among Neonates in the United States. J Perinatol 23, 278–285 (2003). https://doi.org/10.1038/sj.jp.7210892
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7210892
This article is cited by
-
Cholestasis is associated with a higher rate of complications in both medical and surgical necrotizing enterocolitis
Journal of Perinatology (2024)
-
Novel pathogenic GATA6 variant associated with congenital heart disease, diabetes mellitus and necrotizing enterocolitis
Pediatric Research (2024)
-
Necrotizing enterocolitis: current understanding of the prevention and management
Pediatric Surgery International (2024)
-
Impact of concomitant necrotizing enterocolitis on mortality in very low birth weight infants with intraventricular hemorrhage
Journal of Perinatology (2023)
-
Necrotizing enterocolitis: recent advances in treatment with translational potential
Pediatric Surgery International (2023)