Abstract
OBJECTIVE: To assess the impact of highly active antiretroviral therapy, including a protease inhibitor (HAART/PI), on maternal glucose tolerance and fetal growth.
STUDY DESIGN: A retrospective chart review of pregnancy outcomes of human immunodeficiency virus–infected pregnant women receiving HAART/PI (n=41) or zidovudine monotherapy (n=23).
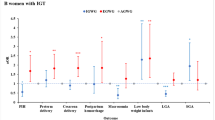
RESULTS: Abnormal 1-hour glucose tolerance tests (1hGTT) were observed in 30% of subjects receiving HAART/PI between 24 and 28 weeks' gestation. An elevated 1hGTT was associated with a significantly lower mean birth weight in subjects receiving HAART/PI compared to babies born to mothers with a normal 1hGTT (3.40±0.09 vs 3.00±0.18 kg, p<0.05, ANOVA).
CONCLUSION: HAART/PI therapy is associated with an increased rate of impaired glucose tolerance in pregnancy and impaired fetal growth. This finding merits further investigation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Connor EM, Sperling RS, Gelber R et al. Reduction of maternal–infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. Pediatric AIDS Clinical Trials Group Protocol 076 Study Group N Engl J Med 1994; 331: 1173–80
Flexner C . HIV-protease inhibitors N Engl J Med 1998; 338: 1281–92
Safrin S, Grunfeld C . Fat distribution and metabolic changes in patients with HIV infection AIDS 1999; 13: 2493–505
Carr A, Samaras K, Thorisdottir A, Kaufmann GR, Chisholm DJ, Cooper DA . Diagnosis, prediction, and natural course of HIV-1 protease-inhibitor–associated lipodystrophy, hyperlipidaemia, and diabetes mellitus: a cohort study Lancet 1999; 353: 2093–9
Murata H, Hruz PW, Mueckler M . The mechanism of insulin resistance caused by HIV protease inhibitor therapy J Biol Chem 2000; 275: 20251–4
Walli R, Herfort O, Michl GM et al. Treatment with protease inhibitors associated with peripheral insulin resistance and impaired oral glucose tolerance in HIV-1–infected patients AIDS 1998; 12: F167–73
Moore TR . Diabetes in pregnancy In: Creasy RK, Resnik R, editors Maternal Fetal Medicine Philadelphia: Saunders 1999
Carpenter MW, Coustan DR . Criteria for screening tests for gestational diabetes Am J Obstet Gynecol 1982; 144: 768–73
Alexander GR, Himes JH, Kaufman RB, Mor J, Kogan M . A United States national reference for fetal growth Obstet Gynecol 1996; 87: 163–8
Langer O, Brustman L, Anyaegbunam A, Mazze R . The significance of one abnormal glucose tolerance test value on adverse outcome in pregnancy Am J Obstet Gynecol 1987; 157: 758–63
Pedersen J . Diabetes mellitus and pregnancy: present status of the hyperglycaemia–hyperinsulinism theory and the weight of the newborn baby Postgrad Med J 1971; 66–7 (Supplement)
Katz EB, Stenbit AE, Hatton K, DePinho R, Charron MJ . Cardiac and adipose tissue abnormalities but not diabetes in mice deficient in GLUT4 Nature 1995; 377: 151–5
Casey BM, Bawdon RE . Placental transfer of ritonavir with zidovudine in the ex vivo placental perfusion model Am J Obstet Gynecol 1998; 179: 758–61
Bawdon RE . The ex vivo human placental transfer of the anti-HIV nucleoside inhibitor abacavir and the protease inhibitor amprenavir Infect Dis Obstet Gynecol 1998; 6: 244–6
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chmait, R., Franklin, P., Spector, S. et al. Protease Inhibitors and Decreased Birth Weight in HIV-Infected Pregnant Women With Impaired Glucose Tolerance. J Perinatol 22, 370–373 (2002). https://doi.org/10.1038/sj.jp.7210748
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7210748
This article is cited by
-
HIV Protease Inhibitors in Pregnancy
Drugs (2013)