Abstract
Objective
To analyse the long-term results of surgical removal of plaque-like diabetic foveal hard exudates.
Methods
In this retrospective, interventional, non-randomized, controlled study, vitrectomy and excision of submacular hard exudates was performed on 11 eyes of 11 patients with plaque-like hard exudates. The mean preoperative LogMAR best corrected visual acuity (BCVA) and exudate diameter were 1.35±0.3 and 1.19 disks (range, 0.5–1.8), respectively. In the control group, which consisted of 10 eyes of 7 patients who refused the surgery, baseline mean BCVA and exudate diameter were 1.06±0.2 and 1.2 disks (range, 0.7–2.0), respectively. Main outcome measures included BCVA, fundus photography, and surgical complications.
Results
The mean follow-up was 39.1±3.2 months for the study group and 32.5±3.6 months for the control group. In the study group, BCVA improved in 8 (73%) eyes at the final examination. The mean final BCVA was 1.08±0.4 (P=0.021). Macular oedema and exudates resolved completely in all eyes. In the control group, final LogMAR visual acuity declined to 1.53±0.1 (P=0.005). Macular pigment epithelium atrophy or scar formation was observed in 7 (64%) eyes in the study group and in all eyes in the control group.
Conclusion
Surgical excision of plaque-like foveal hard exudates resulted in better anatomical and functional outcome when compared to observation alone.
Similar content being viewed by others
Introduction
Exudative diabetic maculopathy is a frequent cause of visual deterioration in patients with diabetic retinopathy and represents macular oedema, which is derived from leaking retinal vessels.1, 2 As the fluid in the extracellular space of the retina is reabsorbed into the retinal capillaries, the deposition and accumulation of the large molecules within the exudative material causes a clinical picture of hard exudates.1, 2, 3, 4
Hard exudates are composed of lipid and proteinaceous material, and settle in the outer retinal layers. Plaque-like hard exudates may deposit in the foveal region and often cause visual loss. The visual loss may be severe and irreversible. Subfoveal hard exudates usually respond poorly to focal laser treatment, but may slowly resolve leaving behind a retinal pigment epithelium (RPE) scar and subfoveal fibrosis with associated poor visual acuity.3, 4, 5, 6, 7, 8, 9, 10 Unfavourable prognosis in eyes with subfoveal plaque-like hard exudates following a natural course encouraged the development of alternative treatment options, one of which is the surgical excision of plaque-like foveal exudate. Takagi et al11 and Sakuraba et al12 first performed excision of submacular hard exudates and reported promising short-term surgical outcomes with favourable prognoses. Takaya et al13 reported long-term results of surgical removal of hard exudates and noted that visual improvement could not be obtained.
In this study, we retrospectively reported an analysis of long-term results of surgical excision of subfoveal plaque-like hard exudates in patients with diabetic maculopathy.
Patients and methods
From April 2001 to April 2002, 11 consecutive eyes of 11 patients with chronic diabetic macular oedema with plaque-like hard exudates involving the fovea with or without diffuse macular thickening and visual acuity less than 6/38 that had undergone surgery were included in the study. All patients were under follow-up in the medical retina service for at least 2 years before election to the study. The study was approved by the local ethical committee of Uludag University and performed in accordance with the tenets of the Declaration of Helsinki. Written informed consent was obtained from each patient after explaining to them the natural process of the disease and the nature of the study. After starting the study, 10 eyes of seven patients with submacular plaque-like hard exudates who did not accept surgical excision of exudates or preferred to have laser treatment only were included in the control group. Patient characteristics and ocular features are given in Table 1.
In the study group, all eyes had received at least one session of focal or grid laser photocoagulation for treatment of exudative diabetic macular oedema before the current operation. In the control group, all eyes had been treated by focal and/or grid photocoagulation as well as partial panretinal photocoagulation in some of the patients with severe non-proliferative diabetic retinopathy. Patients with active neovascularization, fibrous proliferation, tractional retinal detachment, macular ischaemia, vitreomacular traction, and patients with a history of vitreous or subhyaloid haemorrhage were excluded.
All patients had complete ophthalmic examinations including slit-lamp examination for anterior segment, slit-lamp binocular indirect ophthalmoscopy, and intraocular pressure measurement. Potential visual acuity was measured using Lotmar visometer (Haag-Streit, Bern, Switzerland). Colour fundus photography (CFF) and fundus fluorescein angiography (FFA) (Zeiss FF450 Plus Visupac 431) were routinely performed before inclusion into the study.
The operations were performed by the same surgeon (RA). Following a standard three-port total pars plana vitrectomy and removal of posterior hyaloid, a retinotomy was performed at the upper nasal (left eye) or temporal (right eye) site and approximately 2 disk diameters from the fovea using subretinal spatula (DORC 1295-1). A serous neurosensory detachment in the macula was created by injecting balanced salt solution using a subretinal cannula that was attached by a short flexible tube to a 5-ml syringe filled with balanced salt solution. Subretinal hard exudates were gently released using a subretinal spatula to mobilize the plaque exudates. Only subretinally localized hard exudates were removed using a subretinal fluid needle (DORC 1295-4). Any scattered exudates tightly attached to the retina or located in the sensory retina were not removed to avoid accidental tearing of the retina. Consequently, subretinal fluid was passively aspirated by fluid needle. Finally, fluid–air exchange was performed and 20% sulphur hexafluoride tamponade was added. The postoperative follow-up was maintained for at least 35 months (mean 39.1±3.2 months). The follow-up period was between 30 and 39 months (mean 32.5±3.6 months) in the control group. In both groups, visual acuity, CFF, and FFA were evaluated at 3 months, 1 year, and 3 years after surgery. The visual acuity was measured with best refractive correction by the ETDRS charts. BCVA was converted to the logarithm of the minimum angle of resolution (logMAR) and averaged. The visual changes were statistically analysed with the Wilcoxon rank sum tests. Between-groups comparisons were made by Mann–Whitney U test.
Results
Five men and six women were included in the study group. Mean age was 61.7±7.8 years. All patients had type II diabetes. The mean duration of diabetes was 14.6±4.1 years. Patients 3, 5, 6, and 11 were on insulin therapy whereas others were on treatment by oral anti-diabetics. In seven patients, accompanying diffuse oedema of varying degrees was observed. The mean duration of hard exudates was 5.7±2.1 months (Table 1).
The mean age of control patients was 62.3±8.2 years. The mean duration of diabetes was 13.1±8.3 years. All patients had type II diabetes and were on treatment with oral anti-diabetics. There was no difference between the study group and the control group with respect to age (P=0.86), size of plaque-like exudates (P=0.83), baseline BCVA (P=0.06), Lotmar potential visual acuity (P=0.55), fasting blood glucose level (P=0.84), cholesterol (P=0.97), blood urea level (P=0.90), and duration of diabetes mellitus (P=0.38).
Precise anatomical success was obtained in all patients. No intraoperative complication was observed in any patient. The only postoperative complication observed was cataract formation seen in five patients who had undergone phacoemulsification surgery during the period between 12 and 42 months. In three patients, there was progression in nuclear sclerosis that was thought to be visually insignificant. In all eyes, plaque-like hard exudates were removed totally with small pieces of intraretinal residue of exudates. In all eyes, diffuse exudates decreased at the 3-month follow-up and disappeared completely at 1-year follow-up. The recurrence of hard exudates was not observed throughout the follow-up period.
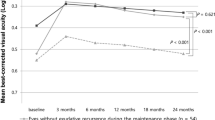
Preoperative Lotmar potential visual acuity (LogMAR) was 0.79±0.3 in the study group and 0.69±0.2 in the control group. Before the operation, BCVAs were less than 6/60 in nine eyes in the study group (Table 1). Visual acuity improved after surgery in 10 eyes at 1-year follow-up. After surgery, visual acuities better than 6/60 were obtained in five eyes at the final examination. Six eyes gained three or more lines of ETDRS visual acuity. Visual acuity did not change or decreased in three eyes at the final follow-up. The best BCVA obtained during the entire follow-up period was 6/15. There was a statistically significant improvement of visual acuity by ETDRS charts at the 3-month, 1-year, and 3-year follow-up examinations compared to preoperative visual acuity measurements. Mean preoperative LogMAR visual acuity of 1.35±0.3 increased to 1.15±0.4 at 3-month (P=0.016), 1.28±0.4 at 1-year (P=0.010), and 1.08±0.5 at 3-year follow-up (P=0.021).
In the control group, BCVA measured at the time of inclusion in the study ranged between 6/30 and 6/150 (Table 1). BCVA changed between 6/120 and 6/240 at the final examination. There was a visual acuity loss varying from 1 ETDRS chart line to 8 ETDRS chart lines in all control eyes. The mean baseline LogMAR visual acuity of 1.06±0.2 decreased to 1.34±0.15 at year 1 and to 1.53±0.09 at the final examination (P=0.012 and P=0.005, respectively).
There was no sign of active diabetic maculopathy in any eye in the study group at the final examination. Diabetic retinopathy subsided in all eyes. Fluorescein angiography showed no significant leakage at the final follow-up. However, there was an RPE scar in two eyes and RPE atrophy, which was localized beneath the old plaque exudates in four eyes. Subretinal fibrosis developed in one eye. There was no apparent abnormality in four eyes (Table 1). In the control group, subfoveal plaque exudates resolved leaving behind an RPE atrophy or scar at the fovea in five eyes, while plaque exudates did not resolve completely in five eyes during the follow-up period with the treatment of laser photocoagulation only (Table 1).
Discussion
In our study, we presented long-term results of removal of subretinal plaque-like hard exudates in patients with exudative diabetic maculopathy. No serious intraocular complications occurred. All of the eyes benefited anatomically from surgery, and all except three eyes gained two or more lines visual acuity.
Although retinal hard exudate usually accompanies diabetic macular oedema, increasing amounts of exudate appear to be independently associated with increased risk of visual impairment.14 The success of the surgical removal of the subfoveal hard exudates largely depends on the anatomical localization of exudates. Although OCT can be used to localize the hard exudates, we did not use it on our patients. Hard exudates are assumed to appear primarily in the outer retinal layer. They can replace almost the entire retinal layer and continue to the subretinal space after longstanding and massive deposition.11 In cases in which foveolar massive hard exudates accumulate in the outer retinal layer, surgical removal has been claimed to result in damage to the neural retina, therefore functional success in these eyes will be very unlikely.11 On the other hand, subretinal hard exudates seem to block interaction between the neurosensory retina and retinal pigment epithelium, and will cause deterioration of visual acuity unless they are extracted or reabsorbed before the time that irreversible damage can occur.15 Therefore, the necessity of removal of subretinal hard exudates to restore the interaction may justify surgical excision of exudates.
Recently, Takaya et al13 reported their long-term results of vitrectomy for removal of submacular hard exudates in patients with diabetic maculopathy. In their series, visual acuity improved in 7 of 13 eyes 1 year after surgery, but improvement of visual acuity was observed in only 5 eyes 3 years after surgery. They compared the visual results with those of eight eyes that underwent vitrectomy without extraction of hard exudates, and concluded that removing submacular hard exudates was not superior to vitrectomy alone. However, in the treatment group, they were confronted with complications of iatrogenic macular hole in three eyes, postoperative submacular haemorrhage in one eye, and optic atrophy in one eye at the final examination, which worsened final visual acuity results. They did not provide durations of hard exudates, which seem to be affecting the results of surgical success. In their control group, there was no statistically significant improvement in visual acuity at the end of the follow-up.
Vitrectomy and removal of the normal or thickened posterior hyaloid has considerable effects on diabetic macular oedema.16, 17 Yang18 reported that in his series of vitrectomy alone, obvious reduction of hard exudates was observed after approximately 3 months and marked resolution occurred within 12 months. Although visual acuity improved in 11 eyes and decreased in 2 eyes, none of the eyes improved more than 6/60. The advantage of surgical removal of the plaque exudates may be early cleaning of the exudates and protecting the fovea from harmful effects of exudates. In our cases, the eyes with duration of subfoveal hard exudates shorter than 6 months (patients 2, 3, 4, 7, 9, and 10) resulted in an increase of visual acuity with the ETDRS chart lines more than the eyes with longer period of time of hard exudates. Visual impairment was observed in two eyes (patients 6 and 8) of our series. The scar or atrophy of retinal pigment epithelium and subsequent visual acuity loss may result from the surgical damage to the retinal pigment epithelium or neurosensorial injury caused by long-term plaque hard exudates. There was no central or peripheral macular ischaemia that would cause dramatic visual loss in our patients. Therefore, visual impairment could be mainly attributed to the impact of long-term effects of plaque exudates. Although intraocular macular hole developed in some eyes in the other series, no such complication was observed in this study. An effort was made not to touch the neurosensory retina; thus, forceps were not used to nudge and draw the plaque-like exudates. Instead of forceps, we used a subretinal fluid needle for atraumatic or at least, less traumatic removal of the exudates (Figure 1).
(a) Fundus photographs of a patient (case 7 in Table 1) in the study group. Preoperative fundus photograph shows plaque hard exudate deposited in foveolar area with the size of 1.8 disk area together with circinate-shaped scattered hard exudates. Diffuse macular oedema is also present. Visual acuity was 6/60 (top left). Subfoveal plaque hard exudate was removed with some residue. (b) In the fundus photograph taken 12 months after surgery, a few residual hard exudates are shown. Scattered exudates are shown to be decreased. Diffuse macular oedema has resolved. Visual acuity was 6/30 (upper middle left). (c, d) In the fundus photograph and fluorescein angiogram taken 39 months after surgery, there is neither any residual hard exudate nor diffuse oedema. Visual acuity was 6/15 (lower middle left and top right). (e) Contralateral eye of the same patient did not undergo surgery and was followed (case 6 in the control group). Baseline photograph showed subfoveal plaque hard exudates similar to fellow eye with the size of 1.2 disk area. Visual acuity was 6/75 (upper middle right). (f, g) In the photograph taken after 39 months, subfoveal exudate is shown to be decreased (lower middle right), but became scared tissue that stained with fluorescein (bottom). Visual acuity was 6/190.
The size of the subfoveal hard exudates is also an important prognostic factor for the success of the surgery. In the series of Takagi et al,11 the extent of hard exudates ranged from 1 to 5 disk areas with the mean of 2.35 disk areas. Takaya et al13 performed surgery in eyes with exudates from 0.5 to 3 disk areas with the mean of 1.25 disk areas. In our study, none of the exudates extended diffusely and all the exudates had ‘plaque-like’ characteristics. The mean size of plaque exudates in our study eyes was 1.19 disk areas with the range of 0.5–1.8 disk areas. In this study, we performed quantitative assessment of visual capacity using Lotmar visometer in all cases.19 All eyes included in the study group had shown an increase in potential visual acuity with Lotmar visometer before surgery. The increased visual acuity by the ETDRS lines obtained long after the operation was concordant with preoperative Lotmar visometer results. However, the BCVA did not reach Lotmar visometer levels in any of the eyes. Lotmar visometer probably predicted potential visual acuity better as reported in macular pathologies in earlier studies.19
In conclusion, the results of surgery appear to be better than the natural history. Although the surgical removal of subfoveal plaque hard exudates is not yet a widespread treatment option, it may be considered in cases resistant to alternative therapies before damage has occurred. Prospective, randomized, controlled studies are needed to enhance our knowledge on the management of subfoveal hard exudates.
References
Bresnick GH . Diabetic macular edema. A review. Ophthalmology 1986; 93: 989–997.
Tachi N . Surgical management of macular edema. Semin Ophthalmol 1998; 13: 20–30.
Hamilton AMP, Ulbig MW, Polkinghorne P . Management of Diabetic Retinopathy. London: BMJ Publishing Group, 1996. 115–128.
Sigurdsson R, Begg IS . Organised macular plaques in exudative diabetic maculopathy. Br J Ophthalmol 1980; 64: 392–397.
Toussaint D, Cogan DG, Kuwabara T . Extravascular lesions of diabetic retinopathy. Arch Ophthalmol 1962; 67: 42–47.
Murata T, Ishibashi T, Inomata H . Immunohistochemical detection of extravasated fibrinogen (fibrin) in human diabetic retina. Graefes Arch Clin Exp Ophthalmol 1992; 30: 428–431.
Vinores SA, Gadegbeku C, Campochiaro PA, Green WR . Immunohistochemical localization of blood–retinal barrier breakdown in human diabetes. Am J Pathol 1989; 134: 231–235.
Early Treatment of Diabetic Retinopathy Study Group. Photocoagulation for diabetic macular edema. Early treatment of diabetic retinopathy study report number 1. Arch Ophthalmol 1985; 103: 1796–1806.
Ferris FL, Patz A . Macular edema: a complication of diabetic retinopathy. Surv Ophthalmol 1984; 28: 452–461.
Fong DS, Segal PP, Myers F, Ferris FL, Hubbard LD, Davis MD . Subretinal fibrosis in diabetic macular edema. ETDRS report no. 23. Arch Ophthalmol 1997; 115: 873–877.
Takagi H, Otani A, Kiryu J, Ogura Y . New surgical approach for removing massive foveal hard exudates in diabetic macular edema. Ophthalmology 1999; 106: 249–256.
Sakuraba T, Suzuki Y, Mizutani H, Nakazawa M . Visual improvement after removal of submacular exudates in patients with diabetic maculopathy. Ophthalmic Surg Lasers 2000; 31: 287–291.
Takaya K, Suzuki Y, Mizutani H, Sakuraba T, Nakazawa M . Long-term results of vitrectomy for removal of submacular hard exudates in patients with diabetic maculopathy. Retina 2004; 24: 23–29.
Chew EY, Klein ML, Ferris FL, Remaley NA, Murphy RP, Chantry K et al., for the ETDRS research group. ETDRS Report 22. Association of elevated serum lipid levels with retinal hard exudate in diabetic retinopathy. Arch Ophthalmol 1996; 114: 1079–1084.
Otani T, Kishi S . Tomographic findings of foveal hard exudates in diabetic macular edema. Am J Ophthalmol 2001; 131: 50–54.
Harbour JW, Smiddy WE, Flynn HW, Rubsamen PE . Vitrectomy for diabetic macular edema associated with a thickened and taut posterior hyaloid membrane. Am J Ophthalmol 1996; 121: 405–413.
Avci R, Kaderli B, Avci B, Simsek S, Baykara M, Kahveci Z et al. Pars plana vitrectomy and removal of the internal limiting membrane in the treatment of chronic macular oedema. Graefes Arch Clin Exp Ophthalmol 2004; 242: 845–852.
Yang CM . Surgical treatment for severe diabetic macular edema with massive hard exudates. Retina 2000; 20: 121–125.
Yazici B, Gelisken O, Avci R, Yucel A . Prediction of visual outcome after retinal detachment surgery using the Lotmar visometer. Br J Ophthalmol 2002; 86: 278–281.
Author information
Authors and Affiliations
Corresponding author
Additional information
None of the authors has any proprietary interest in any subject mentioned in this study
Rights and permissions
About this article
Cite this article
Avci, R., Inan, Ü. & Kaderli, B. Long-term results of excision of plaque-like foveal hard exudates in patients with chronic diabetic macular oedema. Eye 22, 1099–1104 (2008). https://doi.org/10.1038/sj.eye.6702877
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6702877
Keywords
This article is cited by
-
Removal of foveal hard exudates by subretinal balanced salt solution injection using 38-gauge needle in diabetic patients
Graefe's Archive for Clinical and Experimental Ophthalmology (2020)