Abstract
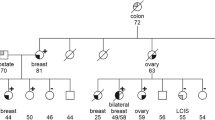
Mutations of the BRCA1 or BRCA2 genes have been shown to strongly predispose towards the development of contralateral breast cancer in patients from large multi-case families. In order to test the hypothesis that BRCA1 and BRCA2 mutations are more frequent in patients with bilateral breast cancer, we have investigated a hospital-based series of 75 consecutive patients with bilateral breast cancer and a comparison group of 75 patients with unilateral breast cancer, pairwise matched by age and family history, for mutations in the BRCA1 and BRCA2 genes. Five frameshift deletions (517delGT in BRCA1; 4772delA, 5946delCT, 6174delT and 8138del5 in BRCA2) were identified in patients with bilateral disease. No further mutations, apart from polymorphisms and 3 rare unclassified variants, were found after scanning the whole BRCA1 and BRCA2 coding sequence. Three pathogenic BRCA1 mutations (Cys61Gly, 3814del5, 5382insC) were identified in the group of patients with unilateral breast cancer. The frequencies of common BRCA1 and BRCA2 missense variants were not different between the 2 groups. In summary, we did not find a significantly increased prevalence of BRCA1 and BRCA2 mutations in a hospital-based cohort of German patients with bilateral breast cancer. We conclude that bilaterality of breast cancer on its own is not strongly associated with BRCA1 and BRCA2 mutations when adjusted for age and family history. The high frequency of bilateral disease in multi-case breast cancer families may be due to a familial aggregation of additional susceptibility factors modifying the penetrance of BRCA1 and BRCA2 mutations. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Adami HO, Bergstrom R and Hansen J (1985) Age at first primary as a determinant of the incidence of bilateral breast cancer. Cumulative and relative risks in a population-based case–control study. Cancer 55: 643–647
Anderson DE (1971) Some characteristics of familial breast cancer. Cancer 28: 1500–1505
Barker DF, Almeida ERA, Casey G, Fain PR, Liao S-Y, Masunaka I, Noble B, Kurosaki T and Anton-Culver H (1996) BRCA1 R841W: A strong candidate for a common mutation with moderate phenotype. Genet Epidemiol 13: 595–604
Beckmann MW, Niederacher D, Goecke TO, Bodden-Heinrich R, Schnürch H-G and Bender HG (1997) Hochrisikofamilien mit Mamma-und Ovarialkarzinomen. Möglichkeiten der Beratung, genetischen Analyse und Früherkennung. Dt Ärztebl 94: A161–167
Bernstein JL, Thompson WD, Risch N and Holford TR (1992) The genetic epidemiology of second primary breast cancer. Am J Epidemiol 136: 937–948
Broeks A, Urbanus JHM, Floore AN, Dahler EC, Klijn JGM, Rutgers EJT, Devilee P, Russell NS, van Leeuwen FE and van’t Veer L (2000) ATM-heterozygous germline mutations contribute to breast cancer-susceptibility. Am J Hum Genet 66: 494–500
Dunning AM, Chiano M, Smith NR, Dearden J, Gore M, Oakes S, Wilson C, Stratton M, Peto J, Easton D, Clayton C and Ponder BAJ (1996) Common BRCA1 variants and susceptibility to breast and ovarian cancer in the general population. Hum Mol Genet 6: 285–289
Durocher F, Shattuck-Eidens D, McClure M, Labrie F, Skolnick MH, Goldgar DE and Simard J (1996) Comparison of BRCA1 polymorphisms, rare sequence variants and/or missense mutations in unaffected and breast/ovarian cancer populations. Hum Mol Genet 5: 835–842
Easton DF, Ford D and Bishop DT the Breast Cancer Linkage Consortium (1995) Breast and ovarian cancer incidence in BRCA1-mutation carriers. Am J Hum Genet 56: 265–271
Eccles DM, Englefield P, Soulby MA and Campbell IG (1998) BRCA1 mutations in southern England. Br J Cancer 77: 2199–2203
Ford D, Easton DF, Bishop DT, Narod SA and Goldgar DE the Breast Cancer Linkage Consortium (1994) Risks of cancer in BRCA1 – mutation carriers. Lancet 343: 692–695
Ford D and Easton D et al (1998) Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. Am J Hum Genet 62: 676–689
Gershoni-Baruch R, Dagan E, Fried G, Kepten I and Robinson E (1999) BRCA1 and BRCA2 founder mutations in patients with bilateral breast cancer. Eur J Hum Genet 7: 833–836
Goss PE and Sierra S (1998) Current perspectives on radiation-induced breast cancer. J Clin Oncol 16: 338–347
Haber D (2000) Roads leading to breast cancer. N Engl J Med 343: 1566–1568
Healey CS, Dunning AM, Dawn Teare M, Chase D, Parker L, Burn J, Chang-Claude J, Mannermaa A, Kataja V, Huntsman DG, Pharoah PDP, Luben RN, Easton DF and Ponder BAJ (2000) A common variant in BRCA2 is associated with both breast cancer risk and prenatal viability. Nature Genet 26: 362–364
Healey EA, Cook EF, Orav EJ, Schnitt SJ, Connolly JL and Harris JR (1993) Contralateral breast cancer: clinical characteristics and impact on prognosis. J Clin Oncol 11: 1545–1552
Hislop TG, Elwood JM, Coldmann AJ, Spinelli JJ, Wort JJ and Ellison LG (1984) Second primary cancer of the breast: incidence and risk factors. Br J Cancer 49: 70–85
Knudson AG Jr (1971) Mutation and cancer: Statistical study of retinoblastoma. Proc Natl Acad Sci USA 68: 820–823
Kote-Jarai Z and Eeles RA (1999) BRCA1, BRCA2 and their possible function in DNA damage response. Br J Cancer 81: 1099–1102
Lamlum H, Tassan NA, Jaeger E, Frayling I, Sieber O, Bin Reza F, Eckert M, Rowan A, Barclay E, Atkin W, Williams C, Gilbert J, Cheadle J, Bell J, Houlston R, Bodmer W, Sampson J and Tomlinson I (2000) Germline APC variants in patients with multiple colorectal adenomas, with evidence for the particular importance of E1317Q. Hum Mol Genet 9: 2215–2221
Larson GP, Zhang G, Ding S, Foldenauer KF, Udar N, Gatti RA, Neuberg D, Lunetta KL, Ruckdeschel JC, Longmate J, Flanagan S and Krontiris TG (1998) An allelic variant at the ATM locus is implicated in breast cancer susceptibility. Genet Testing 1: 165–170
Martin A-M and Weber BL (2000) Genetic and hormonal risk factors in breast cancer. J Natl Cancer Inst 92: 1126–1135
Mose S, Adamietz IA, Thilmann C, Saran F, Pahnke R and Böttcher HD (1995) Die Prognose des bilateralen Mammakarzinoms im Vergleich zum unilateralen Mammatumor. Strahlenther Onkol 171: 207–213
Olsen JH, Seersholm N, Boice JD, Kruger Kjaer S and Fraumeni JF (1999) Cancer risk in close relatives of women with early-onset breast cancer – a population-based incidence study. Br J Cancer 79: 673–679
Otterson GA, Modi S, Nguyen K, Coxon AB and Kaye FJ (1999) Temperature-sensitive RB mutations linked to incomplete penetrance of familial retinoblastoma in 12 families. Am J Hum Genet 65: 1040–1046
Peto J and Mack TM (2000) High constant incidence in twins and other relatives of women with breast cancer. Nature Genet 26: 411–414
Phelan CM, Rebbeck TR, Weber BL, Devilee P, Ruttledge MH, Lynch HT, Lenoir GM, Stratton MR, Easton DF, Ponder BAJ, Cannon-Albright L, Larsson C, Goldgar DE and Narod SA (1996) Ovarian cancer risk in BRCA1 carriers is modified by the HRAS1 variable number of tandem repeat (VNTR) locus. Nature Genet 12: 309–311
Phillips KA, Glendon G and Knight JA (1999) Putting the risk of breast cancer in perspective. N Engl J Med 340: 141–144
Rebbeck TR, Kantoff PW, Krithivas K, Neuhausen S, Blackwood MA, Godwin AK, Daly MB, Narod SA, Garber JE, Lynch HT, Weber BL and Brown M (1999) Modification of BRCA1 – associated breast cancer risk by the polymorphic androgen-receptor CAG repeat. Am J Hum Genet 64: 1371–1377
Rebbeck TR, Walker AH, Zeigler-Johnson C, Weisburg S, Martin A-M, Nathanson KL, Wein AJ and Malkowicz SB (2000) Association of HPC2/ELAC2 genotypes and prostate cancer. Am J Hum Genet 67: 1014–1019
Roberts SA, Spreadborough AR, Bulman B, Barber JBP, Evans DGR and Scott D (1999) Heritability of cellular radiosensitivity: a marker of low-penetrance predisposition genes in breast cancer?. Am J Hum Genet 65: 784–794
Robson M, Gilewki T and Haas B (1998) BRCA-associated breast cancer in young women. J Clin Oncol 16: 1642–1649
Schrag D, Kuntz KM, Garber JE and Weeks JC (2000) Life expectancy gains from cancer prevention strategies for women with breast cancer and BRCA1 or BRCA2 mutations. JAMA 283: 617–624
Southey MC, Tesoriero AA, Anderson CR, Jennings KM, Brown SM, Dite GS, Jenkins MA, Osborne RH, Maskiell JA, Porter L, Giles GG, McCredie MRE, Hopper JL and Venter DJ (1999) BRCA1 mutations and other sequence variants in a population-based sample of Australian women with breast cancer. Br J Cancer 79: 34–39
Varley JM, McGown G, Thorncroft M, James LA, Margison GP, Forster G, Evans DGR, Harris M, Kelsey AM and Birch JM (1999) Are there low-penetrance p53 alleles? Evidence from childhood adrenocortical tumors. Am J Hum Genet 65: 995–1006
Verhoog LC, Brekelmans CTM and Seynaeve C (1998) Survival and tumour characteristics of breast-cancer patients with germline mutations of BRCA1. Lancet 251: 316–321
Verhoog LC, Brekelmans CTM, Seynaeve C, Dahmen G, vanGeel AN, Bartels CCM, Tilanus-Linthorst MMA, Wagner A, Devilee P, Halley DJJ, vandenOuweland AMW, Meijers-Heijboer EJ and Klijn JGM (1999) Survival in hereditary breast cancer associated with germline mutations of BRCA2. J Clin Oncol 17: 3396–3402
Verhoog LC, Brekelmans CTM, Seynaeve C, Meijers-Heijboer EJ and Klijn JGM (2000) Contralateral breast cancer risk is influenced by the age at onset in BRCA1-associated breast cancer. Br J Cancer 83: 384–386
Wagner TMU, Hirtenlehner K, Shen P, Moeslinger R, Muhr D, Fleischmann E, Concin H, Doeller W, Haid A, Lang AH, Mayer P, Petru E, Ropp E, Langbauer G, Kubista E, Scheiner O, Underhill P, Mountain J, Stierer M, Zielinski C and Oefner P (1999) Global sequence diversity of BRCA2: analysis of 71 breast cancer families and 95 control individuals of worldwide populations. Hum Molec Genet 8: 413–423
Welcsh PL, Owens KN and King M-C (2000) Insights into the functions of BRCA1 and BRCA2. Trends Genet 16: 69–74
Zheng L, Pan H, Li S, Flesken-Nikitin A, Chen P-L, Boyer TG and Lee W-H (2000) Sequence-specific transcriptional corepressor function for BRCA1 through a novel zinc finger protein, ZBRK1. Molecular Cell 6: 757–768
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Steinmann, D., Bremer, M., Rades, D. et al. Mutations of the BRCA1 and BRCA2 genes in patients with bilateral breast cancer. Br J Cancer 85, 850–858 (2001). https://doi.org/10.1054/bjoc.2001.2016
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2016
Keywords
This article is cited by
-
Survival analysis of patients with unilateral and bilateral primary breast cancer in Northeast China
Breast Cancer (2015)
-
Comparison of clinicopathological characteristics and prognoses between bilateral and unilateral breast cancer
Journal of Cancer Research and Clinical Oncology (2012)
-
PALB2 mutations in German and Russian patients with bilateral breast cancer
Breast Cancer Research and Treatment (2011)
-
Breast-Conserving Therapy in Women with BRCA1/BRCA2-Associated Breast Cancer
Current Breast Cancer Reports (2010)
-
Combined effects of single nucleotide polymorphisms TP53 R72P and MDM2 SNP309, and p53 expression on survival of breast cancer patients
Breast Cancer Research (2009)